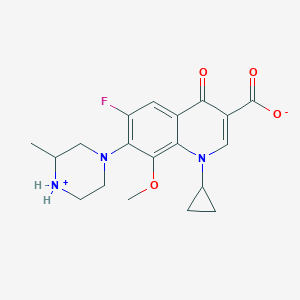
Gatifloxacin
Overview
Description
- Quetiapine Fumarate, also known by its brand names Seroquel and Seroquel XR, belongs to the class of medications called atypical antipsychotics.
- It is primarily used to treat several mental and mood disorders, including schizophrenia, bipolar disorder, and major depressive disorder.
- Quetiapine works by modulating the activity of certain neurotransmitters in the brain, specifically serotonin and dopamine .
Preparation Methods
- Quetiapine Fumarate can be synthesized through various routes, but one common method involves the reaction of quetiapine base with fumaric acid.
- Industrial production methods typically involve large-scale chemical synthesis, ensuring high purity and yield.
Chemical Reactions Analysis
pH-Dependent Solubility and Stability
Gatifloxacin exhibits pH-dependent solubility due to its zwitterionic nature:
- Solubility :
Degradation Pathways :
- Hydrolysis : The ester and amide bonds in prodrug derivatives are susceptible to enzymatic or acidic hydrolysis .
- Oxidation : The piperazinyl group undergoes oxidation under strong oxidative conditions, forming N-oxide derivatives .
Table 2: Stability in Aqueous Solutions
| pH | Half-Life (h) | Major Degradation Product | Reference |
|---|---|---|---|
| 2.5 | 48 | None detected | |
| 7.4 | 24 | Desmethyl-gatifloxacin |
Metal Ion Interactions
This compound forms chelates with divalent cations (e.g., Al³⁺, Mg²⁺), reducing its bioavailability:
- Mechanism : The keto-carboxyl group at position 4 and the piperazinyl nitrogen coordinate with metal ions .
- Example : Co-administration with aluminum hydroxide decreases serum concentrations by 30–40% due to chelation .
Table 3: Chelation Effects
| Metal Ion | Binding Constant (log K) | Bioavailability Reduction (%) | Reference |
|---|---|---|---|
| Al³⁺ | 8.2 | 35 | |
| Mg²⁺ | 6.7 | 25 |
Prodrug Design and Reactivity
To enhance solubility and target specificity, this compound has been modified into prodrugs:
- Ester Prodrugs :
Reaction Mechanism :
- Step 1 : Antioxidant + chloroacetyl chloride → Intermediate (chloroacetate ester).
- Step 2 : Intermediate + this compound → Ester prodrug (nucleophilic substitution) .
Table 4: Prodrug Efficacy
| Prodrug | Antibacterial Activity (MIC₉₀, μg/mL) | Solubility (mg/mL) | Reference |
|---|---|---|---|
| This compound-menthol | 0.25 vs. S. aureus | 45 | |
| This compound-vanillin | 0.5 vs. E. coli | 38 |
Photochemical Reactions
This compound undergoes photodegradation under UV light:
- Primary Products : Decarboxylated and defluorinated derivatives.
- Mechanism : Radical formation at the C-8 methoxy group leads to cleavage .
Experimental Findings :
Scientific Research Applications
- In chemistry, quetiapine serves as a valuable tool for studying neurotransmitter interactions and receptor binding.
- In biology, it aids in understanding brain function and the role of neurotransmitters in mental health.
- In medicine, it is crucial for managing schizophrenia, bipolar disorder, and depression.
- In industry, quetiapine contributes to the development of antipsychotic medications.
Mechanism of Action
- Quetiapine’s effects result from its interaction with specific receptors, including serotonin (5-HT2A and 5-HT2C) and dopamine (D2) receptors.
- By modulating these receptors, it helps restore neurotransmitter balance and alleviate symptoms of mental disorders.
Comparison with Similar Compounds
- Quetiapine stands out due to its dual action on serotonin and dopamine receptors.
- Similar compounds include other atypical antipsychotics like risperidone and olanzapine.
Q & A
Basic Research Questions
Q. What standardized methods are used to determine the minimum inhibitory concentration (MIC) of gatifloxacin against bacterial strains?
To determine MIC values, researchers typically employ broth microdilution or agar dilution assays. For example, in studies on canine periodontopathic bacteria, MICs were quantified by serial dilution of this compound in bacterial cultures, followed by incubation and visual turbidity assessment. Statistical validation using one-way ANOVA and post-hoc tests (e.g., Tukey’s test) ensures reproducibility, with p < 0.05 indicating significance .
Q. How can the physicochemical properties of this compound be characterized for drug formulation studies?
Key methods include:
- Melting point analysis (capillary method, 182–185°C) to confirm purity .
- UV spectroscopy (λmax 285 nm in pH 7.4 buffer) to validate absorption profiles .
- Fourier-transform infrared (FTIR) spectroscopy and differential scanning calorimetry (DSC) to identify functional groups and thermal stability .
Q. What in vitro models are suitable for evaluating this compound’s efficacy against intracellular pathogens?
Macrophage infection models, such as THP-1 cell lines infected with Mycobacterium tuberculosis, are used. Researchers measure bacterial survival rates post-treatment via colony-forming unit (CFU) counts or fluorescence-based assays. Dose-response curves and time-kill kinetics are analyzed to assess bactericidal activity .
Q. How should researchers design pharmacokinetic studies for this compound in animal models?
Protocols include:
- Administering this compound orally/intravenously at therapeutic doses.
- Collecting serial blood/tissue samples over 24–48 hours.
- Using high-performance liquid chromatography (HPLC) or liquid chromatography–mass spectrometry (LC-MS) to quantify drug concentrations .
Q. What criteria define this compound’s in vitro bactericidal activity in time-kill assays?
A ≥3-log reduction in CFU/mL over 24 hours compared to baseline is considered bactericidal. Studies often compare this compound to control antibiotics (e.g., moxifloxacin) and use statistical models to account for variability .
Advanced Research Questions
Q. How can contradictory outcomes in this compound clinical trials (e.g., TB treatment) be systematically analyzed?
Meta-analyses or individual participant data (IPD) reviews are critical. For instance, the REMoxTB trial compared this compound-based regimens to standard therapy, with discrepancies attributed to bacterial load heterogeneity or regional resistance patterns. Researchers should stratify data by covariates like baseline CFU counts and apply mixed-effects models to resolve contradictions .
Q. What advanced spectroscopic techniques validate this compound’s structural integrity in complex matrices (e.g., biological fluids)?
Raman spectroscopy and density functional theory (DFT) simulations are used to map vibrational modes and predict spectral peaks. This approach detects drug residues in meat products by matching experimental spectra to computational models, achieving sensitivities <1 ppm .
Q. How do this compound’s stereoisomers (e.g., 8-hydroxy metabolites) influence antimicrobial activity and toxicity profiles?
Chiral HPLC separates isomers for individual testing. For example, 8-hydroxy this compound (retention time 8.3 min) shows reduced potency compared to the parent compound. Toxicity is assessed via cell viability assays (e.g., MTT), with isomer-specific effects linked to DNA gyrase binding affinities .
Q. What experimental frameworks address this compound resistance mechanisms in gram-negative bacteria?
- Whole-genome sequencing identifies mutations in gyrA/parC genes.
- Efflux pump inhibition assays (using phenylalanine-arginine β-naphthylamide) quantify resistance reversal.
- Transcriptomic analysis (RNA-seq) reveals upregulated resistance genes under sub-MIC exposure .
Q. How can this compound be integrated into combination therapies to mitigate resistance emergence?
Synergy studies use checkerboard assays to calculate fractional inhibitory concentration indices (FICIs). For example, this compound + amikacin (FICI ≤0.5) shows additive effects against multidrug-resistant Pseudomonas aeruginosa. Mechanistic studies (e.g., time-lapse microscopy) further elucidate cooperative bactericidal pathways .
Q. Methodological Notes
- Statistical rigor : Always apply corrections for multiple comparisons (e.g., Bonferroni) in ANOVA-based analyses .
- Spectroscopic validation : Cross-reference FTIR/DSC data with pharmacopeial standards (e.g., USP) to confirm drug purity .
- Ethical compliance : Adhere to institutional guidelines for animal/human studies, including informed consent and sample size justification .
Properties
IUPAC Name |
1-cyclopropyl-6-fluoro-8-methoxy-7-(3-methylpiperazin-4-ium-1-yl)-4-oxoquinoline-3-carboxylate | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C19H22FN3O4/c1-10-8-22(6-5-21-10)16-14(20)7-12-15(18(16)27-2)23(11-3-4-11)9-13(17(12)24)19(25)26/h7,9-11,21H,3-6,8H2,1-2H3,(H,25,26) | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XUBOMFCQGDBHNK-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1CN(CC[NH2+]1)C2=C(C=C3C(=C2OC)N(C=C(C3=O)C(=O)[O-])C4CC4)F | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C19H22FN3O4 | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Weight |
375.4 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Gatifloxacin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015178 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
6.31e-01 g/L | |
| Record name | Gatifloxacin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015178 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
CAS No. |
112811-59-3 | |
| Record name | Gatifloxacin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=112811-59-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Gatifloxacin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015178 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
182 - 185 °C | |
| Record name | Gatifloxacin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015178 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















