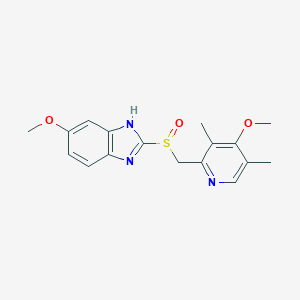
Omeprazole
Overview
Description
Omeprazole is a proton pump inhibitor used to treat various conditions related to excessive stomach acid production, such as gastroesophageal reflux disease (GERD), peptic ulcer disease, and Zollinger-Ellison syndrome . It works by reducing the amount of acid produced in the stomach, providing relief from symptoms and promoting healing of the affected tissues .
Preparation Methods
Synthetic Routes and Reaction Conditions: Omeprazole is synthesized through a multi-step process. One common method involves the reaction of 5-methoxy-2-[(4-methoxy-3,5-dimethyl-2-pyridinyl)methylthio]-1H-benzimidazole with peroxyacetic acid in a two-phase water and chlorinated organic solvent medium under alkaline conditions . The reaction mixture is then separated into water and organic phases, and this compound is isolated from the organic phase .
Industrial Production Methods: In industrial settings, this compound is produced using similar synthetic routes but on a larger scale. The process involves the use of sodium sulfite and sodium hydroxide in an aqueous solution, followed by the addition of acetic acid and this compound seed crystals . The mixture is stirred, centrifuged, washed, and dried to obtain the final product .
Chemical Reactions Analysis
Types of Reactions: Omeprazole undergoes various chemical reactions, including oxidation, reduction, and substitution. For example, it can be oxidized to form hydroxythis compound and the corresponding carboxylic acid .
Common Reagents and Conditions: Common reagents used in the reactions involving this compound include peroxyacetic acid for oxidation and sodium hydroxide for maintaining alkaline conditions . The reactions are typically carried out in organic solvents such as chlorinated solvents .
Major Products Formed: The major products formed from the reactions of this compound include hydroxythis compound and its carboxylic acid derivative .
Scientific Research Applications
Gastroesophageal Reflux Disease (GERD)
Omeprazole is primarily indicated for the treatment of GERD, a condition characterized by the backward flow of stomach acid into the esophagus, causing heartburn and potential esophageal injury. Clinical studies have demonstrated that this compound effectively alleviates GERD symptoms and promotes esophageal healing.
- Efficacy : In a study involving patients with GERD, this compound showed healing rates of 70-80% after 8 weeks of treatment .
- Dosage : The typical dosage ranges from 20 mg to 40 mg daily.
Peptic Ulcer Disease
This compound is extensively utilized in treating peptic ulcers, including gastric and duodenal ulcers. Its ability to suppress gastric acid secretion facilitates ulcer healing.
- Healing Rates : Research indicates that this compound achieves cumulative healing rates of up to 100% after 4–8 weeks at doses of 20–40 mg once daily .
- Combination Therapy : It is often prescribed alongside antibiotics for Helicobacter pylori eradication, enhancing ulcer healing rates .
Zollinger-Ellison Syndrome
This rare condition is characterized by excessive gastric acid production due to gastrin-secreting tumors. This compound is considered the drug of choice due to its potent and long-lasting antisecretory effects.
- Clinical Outcomes : Studies have shown that this compound can effectively manage acid hypersecretion in patients with Zollinger-Ellison syndrome, often requiring higher doses (up to 60 mg daily) for optimal control .
Erosive Esophagitis
Erosive esophagitis, resulting from prolonged exposure to stomach acid, can be effectively treated with this compound.
- Treatment Protocol : Patients typically receive 20 mg to 40 mg daily until healing is confirmed through endoscopy .
Prevention of Upper Gastrointestinal Bleeding
This compound is also used prophylactically in high-risk patients (e.g., those on NSAIDs) to prevent upper gastrointestinal bleeding.
- Effectiveness : A meta-analysis indicated significant reductions in bleeding events among patients receiving PPIs compared to those not receiving them .
Novel Applications and Research Insights
Recent studies have explored innovative applications of this compound beyond traditional uses:
- Anti-Cancer Properties : this compound has been shown to reduce breast cancer cell invasion and metastasis in vitro and in vivo models by downregulating prometastatic genes such as CXCR4 . This suggests potential roles in adjunct cancer therapies.
- Microbial Infections : New formulations combining this compound with chitosan have demonstrated antimicrobial effects, indicating its potential use in treating skin infections .
Data Table: Summary of this compound Applications
| Application | Indication | Typical Dosage | Healing Rate |
|---|---|---|---|
| Gastroesophageal Reflux Disease | GERD | 20-40 mg daily | 70-80% after 8 weeks |
| Peptic Ulcer Disease | Gastric/Duodenal Ulcers | 20-40 mg daily | Up to 100% after 4–8 weeks |
| Zollinger-Ellison Syndrome | Excessive gastric acid production | Up to 60 mg daily | Effective control |
| Erosive Esophagitis | Damage from acid reflux | 20-40 mg daily | Confirmed via endoscopy |
| Prevention of GI Bleeding | High-risk patients (e.g., NSAID users) | Varies | Significant reduction |
Case Studies
-
Case Study on GERD Treatment :
A cohort study involving 150 patients with GERD showed that those treated with this compound experienced significant symptom relief and esophageal healing compared to those receiving placebo . -
Zollinger-Ellison Syndrome Management :
A long-term study on patients with Zollinger-Ellison syndrome demonstrated that those on high-dose this compound maintained normal gastric pH levels, effectively controlling symptoms associated with excessive acid secretion . -
Breast Cancer Metastasis Reduction :
A preclinical trial indicated that this compound significantly decreased metastasis in breast cancer models by inhibiting CXCR4 expression, suggesting a novel application in oncology .
Mechanism of Action
Comparison with Similar Compounds
While all these compounds share a similar mechanism of action, omeprazole is unique in its specific chemical structure and pharmacokinetic properties . For instance, esthis compound is the S-isomer of this compound and is considered to have a more consistent pharmacokinetic profile . Pantoprazole and lansoprazole differ in their chemical structures and are used for slightly different clinical indications .
Conclusion
This compound is a widely used proton pump inhibitor with significant therapeutic benefits for acid-related disorders. Its synthesis involves complex chemical reactions, and it undergoes various transformations to form active metabolites. This compound’s mechanism of action and its applications in scientific research make it a valuable compound in both medicine and industry. Compared to other proton pump inhibitors, this compound stands out due to its unique chemical properties and clinical efficacy.
Biological Activity
Omeprazole is a widely used proton pump inhibitor (PPI) that primarily functions to reduce gastric acid secretion. This article explores the biological activity of this compound, detailing its pharmacological effects, mechanisms of action, and clinical efficacy based on diverse research findings.
This compound exerts its therapeutic effects by irreversibly binding to the H+/K+ ATPase enzyme (proton pump) located in the parietal cells of the stomach. This binding inhibits the final step of gastric acid production, leading to decreased acidity in the gastric lumen. The activation of this compound occurs in an acidic environment, where it transforms into its active sulfenamide form, which then covalently attaches to the proton pump .
Key Mechanisms:
- Proton Pump Inhibition : this compound inhibits acid secretion by blocking the proton pump, effectively reducing gastric acidity.
- Antioxidant Activity : It has been shown to possess antioxidant properties that may protect gastric and intestinal tissues from oxidative damage .
- Anti-inflammatory Effects : this compound reduces inflammation in gastric tissues, contributing to its gastroprotective effects .
Pharmacological Effects
This compound has demonstrated a variety of biological activities beyond its primary role as an acid reducer.
Table 1: Summary of this compound's Biological Activities
| Activity Type | Description |
|---|---|
| Gastroprotective | Reduces gastric lesions and promotes healing through increased prostaglandin synthesis. |
| Antibacterial | Exhibits antibacterial activity against Helicobacter pylori and other bacteria at low pH. |
| Antioxidant | Reduces oxidative stress in gastric tissues, potentially lowering the risk of lesions. |
| Anti-inflammatory | Decreases pro-inflammatory cytokines in various cell types, including microglial cells. |
Clinical Efficacy
Numerous studies have evaluated the effectiveness of this compound for treating acid peptic diseases (APD). A recent observational study involving 96 patients reported significant symptom reduction after four weeks of this compound therapy. The study measured symptom severity using the Patient Assessment of Gastrointestinal Disorder Symptom Severity Index (PAGI-SYM) and patient satisfaction through the Treatment Satisfaction Questionnaire for Medication (TSQM).
Study Findings:
- Symptom Reduction : The proportion of patients with symptoms decreased from 38.54% at baseline to 8.33% by day 28.
- PAGI-SYM Scores : Initial scores averaged 41.32, dropping to 20.86 on day 14 and 8.93 on day 28 (p < 0.0001).
- Satisfaction Scores : TSQM scores improved significantly from 34.69 on day 14 to 36.67 on day 28 .
Case Studies
A case study highlighted the potential hepatotoxicity associated with this compound use. In this study, reactive metabolites were identified that correlated with liver injury in both rat models and human liver microsomal systems . These findings underscore the importance of monitoring liver function in patients undergoing long-term this compound therapy.
Safety Profile
While this compound is generally well-tolerated, some adverse effects have been reported:
Q & A
Basic Research Questions
Q. What analytical methods are recommended for assessing omeprazole purity in compliance with pharmacopeial standards?
High-performance liquid chromatography (HPLC) with UV detection is the primary method for quantifying this compound and its related substances, as outlined in the European Pharmacopoeia 6.0. Critical parameters include mobile phase composition (e.g., phosphate buffer and acetonitrile gradients) and column selection (C18 stationary phase). Validation must include specificity, linearity, and precision testing to distinguish this compound from impurities like this compound sulfone and desmethyl this compound .
Q. How should stability studies for this compound formulations be designed to evaluate degradation under varying conditions?
Stability protocols should follow ICH guidelines (Q1A-R2), testing accelerated (40°C/75% RH) and long-term (25°C/60% RH) conditions over 6–24 months. Key degradation pathways include oxidation (e.g., sulfone formation) and pH-dependent hydrolysis. Analytical methods like forced degradation studies under acidic/alkaline/oxidative stress, coupled with mass spectrometry, help identify degradation products .
Q. What literature review strategies are effective for identifying knowledge gaps in this compound research?
Systematic searches across MEDLINE, EMBASE, and SCOPUS using MeSH terms (e.g., "this compound/pharmacokinetics," "proton pump inhibitors/adverse effects") and Boolean operators. Exclude non-peer-reviewed sources (e.g., theses, editorials) and prioritize meta-analyses for clinical data synthesis. Tools like PRISMA flow diagrams enhance reproducibility .
Advanced Research Questions
Q. How can chemometric models optimize chromatographic separation of this compound and its metabolites?
A face-centered central composite design (FCCD) evaluates factors like buffer pH (7.0–9.0), column temperature (20–40°C), and voltage in capillary electrophoresis. Responses (retention time, resolution) are modeled using regression analysis, with MODDE software removing insignificant coefficients (p > 0.05). This approach reduces analysis time to <10 minutes while maintaining baseline resolution between this compound and lansoprazole .
Q. What methodologies address contradictory data in this compound pharmacovigilance studies (e.g., hypertension risk)?
Subgroup analysis of WHO VigiBase reports can isolate cases where this compound is the sole suspected agent. Apply Austin Bradford-Hill criteria to assess causality: temporal relationship (time-to-onset), biological plausibility (e.g., endothelial dysfunction), and rechallenge/dechallenge outcomes. Exclude patients with pre-existing hypertension and adjust for confounders (e.g., NSAID co-administration) .
Q. How are adsorption isotherm models applied to study this compound retention in reversed-phase liquid chromatography (RP-LC)?
Combine factorial design (e.g., full factorial with center points) and adsorption isotherm measurements (e.g., Langmuir model) to predict retention behavior. Variables include mobile phase pH and temperature. Responses like tailing factor and resolution are optimized using iterative refinement, validated via ANOVA (p < 0.05) .
Q. What strategies ensure robust impurity profiling in generic this compound formulations?
Method validation per ICH Q2(R1) guidelines includes specificity (spiking studies with impurities like this compound sulfone), linearity (R² ≥ 0.995), and accuracy (98–102% recovery). Use reference standards (e.g., this compound Impurity 29) for quantification, with LC-MS/MS confirming structural identity. Cross-validate against pharmacopeial monographs .
Q. How can environmental risk assessments (ERA) for this compound be conducted to meet EMA guidelines?
ERA requires data on predicted environmental concentrations (PEC) in aquatic systems, derived from excretion rates and wastewater removal efficiency. Chronic toxicity tests on Daphnia magna and algae (OECD 211/201) determine PNEC (predicted no-effect concentration). A risk quotient (PEC/PNEC) >1 triggers mitigation strategies (e.g., improved wastewater treatment) .
Q. Data Management and Reporting
Q. What best practices ensure reproducibility in this compound research data?
- Metadata: Document experimental conditions (e.g., HPLC column lot, mobile phase pH).
- Storage: Use FAIR-aligned repositories (e.g., Zenodo) with DOI assignment.
- Tables/Figures: Follow journal guidelines (e.g., Roman numeral labeling, self-explanatory captions). Include raw data in supplementary files .
Q. How to design ethical clinical studies on this compound’s off-label uses?
Submit protocols to institutional review boards (IRBs) detailing inclusion/exclusion criteria (e.g., age, renal/hepatic function). Use stratified randomization to balance covariates (e.g., CYP2C19 genotype). Report adverse events per CONSORT guidelines, with causality assessed via Naranjo scale .
Properties
IUPAC Name |
6-methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl)methylsulfinyl]-1H-benzimidazole | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C17H19N3O3S/c1-10-8-18-15(11(2)16(10)23-4)9-24(21)17-19-13-6-5-12(22-3)7-14(13)20-17/h5-8H,9H2,1-4H3,(H,19,20) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
SUBDBMMJDZJVOS-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=CN=C(C(=C1OC)C)CS(=O)C2=NC3=C(N2)C=C(C=C3)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C17H19N3O3S | |
| Record name | omeprazole | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Omeprazole | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID6021080 | |
| Record name | Omeprazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6021080 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
345.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Omeprazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001913 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
35.4 [ug/mL] (The mean of the results at pH 7.4), Freely soluble in ethanol and methanol, and slightly soluble in acetone and isopropanol and very slightly soluble in water., In water, 82.3 mg/L at 25 °C /Estimated/, 0.5 mg/mL | |
| Record name | SID56422106 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Omeprazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00338 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | OMEPRAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3575 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Omeprazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001913 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Vapor Pressure |
9.2X10-13 mm Hg at 25 °C /Estimated/ | |
| Record name | OMEPRAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3575 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Hydrochloric acid (HCl) secretion into the gastric lumen is a process regulated mainly by the H(+)/K(+)-ATPase of the proton pump, expressed in high quantities by the parietal cells of the stomach. ATPase is an enzyme on the parietal cell membrane that facilitates hydrogen and potassium exchange through the cell, which normally results in the extrusion of potassium and formation of HCl (gastric acid). Omeprazole is a member of a class of antisecretory compounds, the substituted _benzimidazoles_, that stop gastric acid secretion by selective inhibition of the _H+/K+ ATPase_ enzyme system. Proton-pump inhibitors such as omeprazole bind covalently to cysteine residues via disulfide bridges on the alpha subunit of the _H+/K+ ATPase_ pump, inhibiting gastric acid secretion for up to 36 hours. This antisecretory effect is dose-related and leads to the inhibition of both basal and stimulated acid secretion, regardless of the stimulus. **Mechanism of H. pylori eradication** Peptic ulcer disease (PUD) is frequently associated with _Helicobacter pylori_ bacterial infection (NSAIDs). The treatment of H. pylori infection may include the addition of omeprazole or other proton pump inhibitors as part of the treatment regimen,. _H. pylori_ replicates most effectively at a neutral pH. Acid inhibition in H. pylori eradication therapy, including proton-pump inhibitors such as omeprazole, raises gastric pH, discouraging the growth of H.pylori. It is generally believed that proton pump inhibitors inhibit the _urease_ enzyme, which increases the pathogenesis of H. pylori in gastric-acid related conditions., Omeprazole is a selective and irreversible proton pump inhibitor. Omeprazole suppresses gastric acid secretion by specific inhibition of the hydrogen-potassium adenosinetriphosphatase (H+, K+-ATPase) enzyme system found at the secretory surface of parietal cells. It inhibits the final transport of hydrogen ions (via exchange with potassium ions) into the gastric lumen. Since the H+/K+ ATPase enzyme system is regarded as the acid (proton) pump of the gastric mucosa, omeprazole is known as a gastric acid pump inhibitor. Omeprazole inhibits both basal and stimulated acid secretion irrespective of the stimulus., After oral administration, the onset of the antisecretory effect of omeprazole occurs within one hour, with the maximum effect occurring within two hours. Inhibition of secretion is about 50% of maximum at 24 hours and the duration of inhibition lasts up to 72 hours. The antisecretory effect thus lasts far longer than would be expected from the very short (less than one hour) plasma half-life, apparently due to prolonged binding to the parietal H + /K + ATPase enzyme. When the drug is discontinued, secretory activity returns gradually, over 3 to 5 days. The inhibitory effect of omeprazole on acid secretion increases with repeated once-daily dosing, reaching a plateau after four days. | |
| Record name | Omeprazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00338 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | OMEPRAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3575 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from acetonitrile, White to off-white crystalline powder | |
CAS No. |
73590-58-6 | |
| Record name | Omeprazole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=73590-58-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Omeprazole [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0073590586 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Omeprazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00338 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | omeprazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759192 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | omeprazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=751450 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Omeprazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6021080 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 6-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.122.967 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | OMEPRAZOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/KG60484QX9 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | OMEPRAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3575 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Omeprazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001913 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
156 °C | |
| Record name | Omeprazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00338 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | OMEPRAZOLE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3575 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Omeprazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001913 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details






Synthesis routes and methods III
Procedure details






Synthesis routes and methods IV
Procedure details






Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














