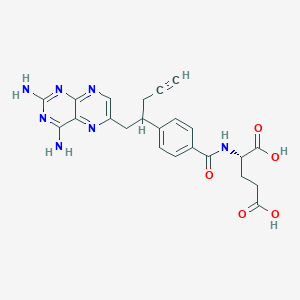
Pralatrexate
Overview
Description
Pralatrexate is a novel antifolate compound used primarily for the treatment of relapsed or refractory peripheral T-cell lymphoma. It was approved by the U.S. Food and Drug Administration in 2009. This compound is designed to have a high affinity for the reduced folate carrier, which allows it to be efficiently internalized by tumor cells .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Pralatrexate involves several key steps. One method starts with 10-propargyl-10-methoxycarbonyl-4-deoxy-4-amino-10-deaza pteroic acid methyl ester as the starting material. This compound undergoes a saponification reaction to yield 4-(2-carboxy-1-(2,4-diaminopteridine-6-yl)pent-4-yn-2-yl)benzoic acid. This intermediate is then decarboxylated to produce 4-(1-(2,4-diaminopteridine-6-yl)pent-4-yn-2-yl)benzoic acid, which reacts with L-diethyl glutamate to form 10-propargyl-10-deaza aminopterin diethyl ester. Finally, a saponification reaction is performed to obtain this compound .
Industrial Production Methods: The industrial production of this compound follows similar synthetic routes but is optimized for higher yields and purity. The process involves the use of polar solvents like dehydrated alcohol and basic solutions such as sodium hydroxide to facilitate the reactions .
Chemical Reactions Analysis
Types of Reactions: Pralatrexate undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under specific conditions to form different derivatives.
Reduction: Reduction reactions can modify the pteridine ring system.
Substitution: Substitution reactions can occur at the amino groups or the pteridine ring.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents like sodium borohydride are used.
Substitution: Reagents such as alkyl halides and acyl chlorides are employed.
Major Products: The major products formed from these reactions include various derivatives of this compound that can have different pharmacological properties .
Scientific Research Applications
Pralatrexate has a wide range of scientific research applications:
Chemistry: Used as a model compound to study antifolate mechanisms and develop new antifolate drugs.
Biology: Helps in understanding folate metabolism and transport in cells.
Medicine: Primarily used in the treatment of peripheral T-cell lymphoma. .
Industry: this compound’s synthesis and production methods are studied to improve industrial processes and reduce costs
Mechanism of Action
Pralatrexate is a folate analog metabolic inhibitor that competitively inhibits dihydrofolate reductase (DHFR). This inhibition prevents the reduction of dihydrofolate to tetrahydrofolate, a critical step in the synthesis of thymidylate, purine nucleotides, and certain amino acids. By inhibiting DHFR, this compound disrupts DNA, RNA, and protein synthesis, leading to cell death. The compound selectively targets cancer cells overexpressing the reduced folate carrier protein-1 (RFC-1), enhancing its therapeutic window .
Comparison with Similar Compounds
Methotrexate: An older antifolate used in various cancers and autoimmune diseases.
Pemetrexed: Another antifolate used in the treatment of non-small-cell lung cancer and mesothelioma.
Raltitrexed: Used in the treatment of colorectal cancer.
Lometrexol: An investigational antifolate with potential anticancer properties.
Uniqueness of Pralatrexate: this compound is unique due to its high affinity for the reduced folate carrier, which allows for efficient internalization and retention in tumor cells. This property makes it more effective in targeting cancer cells compared to other antifolates .
Biological Activity
Pralatrexate, a novel antifolate drug, has emerged as a significant therapeutic agent in the treatment of various malignancies, particularly peripheral T-cell lymphoma (PTCL). Its mechanism of action, efficacy, and safety profile have been extensively studied, providing insights into its biological activity.
This compound acts primarily as an inhibitor of dihydrofolate reductase (DHFR) and is known to interfere with folate metabolism. This inhibition leads to a reduction in the synthesis of nucleotides necessary for DNA replication and cell division. The drug is transported into cells via the Reduced Folate Carrier (RFC), and its efficacy is closely linked to the expression levels of RFC in different cancer cell lines .
Overview of Clinical Trials
This compound has been evaluated in several clinical trials, notably the PROPEL study, which focused on patients with relapsed or refractory PTCL. This study enrolled 115 patients and reported a response rate of 29%, with 11% achieving complete responses and 18% partial responses. The median duration of response was 10.1 months, and the median progression-free survival (PFS) was 3.5 months .
Case Studies
-
PROPEL Study :
- Population : 115 patients with relapsed/refractory PTCL.
- Dosage : this compound administered at 30 mg/m² weekly for six weeks.
- Results :
- Combination Therapy with Romidepsin :
Biological Activity in Preclinical Models
This compound has demonstrated potent anti-tumor activity in various preclinical models. Studies have shown that it exhibits concentration- and time-dependent cytotoxicity against T-cell lymphoma lines, often showing synergistic effects when combined with other agents such as bortezomib. In vitro studies indicated that this compound induced apoptosis through caspase activation pathways, particularly in sensitive cell lines .
Table: Summary of Key Findings from Preclinical Studies
| Study Type | Cell Lines Tested | Efficacy Observed | Mechanism of Action |
|---|---|---|---|
| In Vitro | T-cell lymphoma lines | Synergistic effects with bortezomib | Induction of apoptosis via caspases |
| In Vivo | Mouse xenograft models | Significant tumor regression | Inhibition of DHFR and RFC uptake |
| Combination Therapy | Various malignancies | Enhanced response rates | Modulation of growth/survival pathways |
Safety Profile
The safety profile of this compound has been characterized by common adverse effects including myelosuppression, mucositis, and fatigue. In the PROPEL study, severe adverse events were reported but were generally manageable . The addition of vitamin B12 and folic acid has been recommended to mitigate some side effects associated with antifolate therapy .
Q & A
Basic Research Questions
Q. What is the mechanism of action of pralatrexate, and how does it differ from other antifolate agents?
this compound is a folate analogue metabolic inhibitor that selectively targets dihydrofolate reductase (DHFR), a critical enzyme in nucleotide synthesis. Unlike methotrexate, this compound exhibits higher affinity for the reduced folate carrier type 1 (RFC-1), enhancing cellular uptake in tumors with overexpressed RFC-1 receptors. This selectivity improves cytotoxicity in malignant cells while potentially sparing normal tissues . Methodologically, its efficacy is validated via in vitro assays measuring DHFR inhibition and competitive binding studies comparing RFC-1 affinity across antifolates.
Q. What clinical trial designs have been used to evaluate this compound’s efficacy in relapsed/refractory lymphomas?
The pivotal PROPEL study (PDX-008) was a non-randomized, single-arm, open-label Phase II trial that established this compound’s monotherapy efficacy in peripheral T-cell lymphoma (PTCL). Key endpoints included overall response rate (ORR: 29% per central review) and duration of response (median 10.1 months). Limitations of this design (e.g., lack of comparator arm) were addressed in follow-up studies like PDX-009, a Phase I/II trial combining this compound with gemcitabine, which used a 3+3 dose-escalation model to determine maximum tolerated dose (MTD) .
Q. How is this compound’s safety profile characterized in clinical settings?
Common adverse events (Grade ≥3) include mucositis (22%), thrombocytopenia (22%), and neutropenia (14%). Safety assessments in trials like PROPEL relied on CTCAE criteria, with dose adjustments (e.g., vitamin B12/folate supplementation) to mitigate toxicity. Real-world retrospective analyses corroborate these findings but note variability in tolerability based on patient comorbidities .
Advanced Research Questions
Q. How can preclinical data on this compound’s synergy with other agents inform clinical trial design?
Preclinical studies in NHL cell lines demonstrated schedule-dependent synergy between this compound and gemcitabine, attributed to sequential inhibition of nucleotide synthesis pathways. This informed PDX-009’s Phase I design, where this compound was administered 24 hours before gemcitabine to maximize synergy. However, clinical translation showed limited efficacy (ORR: 15% in Phase IIa), highlighting the need for pharmacodynamic biomarkers to optimize scheduling .
Q. What methodological challenges arise when repurposing this compound for COVID-19 treatment?
Computational screening identified this compound as a potent SARS-CoV-2 inhibitor (EC50: 0.008 µM vs. remdesivir’s EC50: 1.65 µM). In vitro validation used Vero E6 cells infected with SARS-CoV-2, with viral load quantified via RT-PCR. Challenges include reconciling its high cytotoxicity (peak plasma concentration = 10.5 µM) with therapeutic index requirements and designing adaptive clinical trials to assess antiviral vs. toxic effects .
Q. How do real-world efficacy data for this compound compare with controlled trial outcomes?
A multicenter retrospective analysis of relapsed/refractory PTCL patients (n=109) reported lower ORR (15%) vs. PROPEL (29%), attributed to heterogeneous prior therapies and poorer baseline performance status. Methodologically, such studies use propensity scoring to adjust for confounding variables but remain limited by selection bias, underscoring the need for registry-based prospective cohorts .
Q. What strategies address contradictions between preclinical and clinical data for this compound combinations?
Despite preclinical synergy with carboplatin in ovarian cancer models, Phase I/II trials (e.g., carboplatin AUC5 + this compound) achieved only modest efficacy (median PFS: 7.2 months). Discrepancies may stem from tumor microenvironment factors or folate pathway redundancy. Current strategies include integrating patient-derived xenografts (PDXs) to model resistance mechanisms and using functional imaging (e.g., FDG-PET) to assess early metabolic response .
Q. How are pharmacogenomic factors incorporated into this compound research?
RFC-1 expression and polymorphisms in DHFR and thymidylate synthase (TYMS) are explored as predictive biomarkers. For example, RNA-seq profiling in PTCL patients correlates RFC-1 mRNA levels with response duration. However, validation requires multicenter consortia to aggregate sufficient samples, given PTCL’s rarity .
Q. Methodological Recommendations
- For combination trials : Prioritize sequential dosing guided by preclinical synergy studies and incorporate biomarker-driven patient stratification.
- For repurposing studies : Use physiologically based pharmacokinetic (PBPK) modeling to balance antiviral efficacy and toxicity .
- For real-world data : Apply standardized RECIST criteria across retrospective cohorts to reduce heterogeneity in response assessment .
Properties
IUPAC Name |
(2S)-2-[[4-[1-(2,4-diaminopteridin-6-yl)pent-4-yn-2-yl]benzoyl]amino]pentanedioic acid | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C23H23N7O5/c1-2-3-14(10-15-11-26-20-18(27-15)19(24)29-23(25)30-20)12-4-6-13(7-5-12)21(33)28-16(22(34)35)8-9-17(31)32/h1,4-7,11,14,16H,3,8-10H2,(H,28,33)(H,31,32)(H,34,35)(H4,24,25,26,29,30)/t14?,16-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OGSBUKJUDHAQEA-WMCAAGNKSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C#CCC(CC1=CN=C2C(=N1)C(=NC(=N2)N)N)C3=CC=C(C=C3)C(=O)NC(CCC(=O)O)C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C#CCC(CC1=CN=C2C(=N1)C(=NC(=N2)N)N)C3=CC=C(C=C3)C(=O)N[C@@H](CCC(=O)O)C(=O)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C23H23N7O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3048578 | |
| Record name | Pralatrexate | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3048578 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
477.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Practically insoluble in shloroform and ethanol, Soluble in aqueous solutions at pH 6.5 or higher | |
| Record name | Pralatrexate | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7786 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Pralatrexate is a folate analog metabolic inhibitor that competitively inhibits dihydrofolate reductase (DHFR) selectively in cancer cells overexpressing the reduced folate carrier protein-1 (RFC-1). Folate is a water-soluble vitamin required for DNA synthesis and maintenance as well as DNA, RNA, and protein methylation. As cancer cells are rapidly replicating, they require a lot of folates to accommodate an accelerated cell division and DNA and protein modification for cellular transformation. Therefore, interruption with folate metabolism can inhibit tumor growth. Additionally, pralatrexate also undergoes polyglutamylation catalyzed by folyopolyglutamate synthase (FPGS). This reaction both increases cellular retention of pralatrexate for extended drug action and impedes the uptake of folate, also a substrate of FPGS, to further inhibit folate metabolism in cancer cells., Pralatrexate is a folate analogue metabolic inhibitor that competitively inhibits dihydrofolate reductase. It is also a competitive inhibitor for polyglutamylation by the enzyme folylpolyglutamyl synthetase. This inhibition results in the depletion of thymidine and other biological molecules the synthesis of which depends on single carbon transfer., This study evaluated mechanistic differences of pralatrexate, methotrexate, and pemetrexed. Inhibition of dihydrofolate reductase (DHFR) was quantified using recombinant human DHFR. Cellular uptake and folylpolyglutamate synthetase (FPGS) activity were determined using radiolabeled pralatrexate, methotrexate, and pemetrexed in NCI-H460 non-small cell lung cancer (NSCLC) cells. The tumor growth inhibition (TGI) was assessed using MV522 and NCI-H460 human NSCLC xenografts. Apparent K ( i ) values for DHFR inhibition were 45, 26, and >200 nM for pralatrexate, methotrexate, and pemetrexed, respectively. A significantly greater percentage of radiolabeled pralatrexate entered the cells and was polyglutamylatated relative to methotrexate or pemetrexed. In vivo, pralatrexate showed superior anti-tumor activity in both NSCLC models, with more effective dose-dependent TGI in the more rapidly growing NCI-H460 xenografts. Pralatrexate demonstrated a distinct mechanistic and anti-tumor activity profile relative to methotrexate and pemetrexed. Pralatrexate exhibited enhanced cellular uptake and increased polyglutamylation, which correlated with increased TGI in NSCLC xenograft models. | |
| Record name | Pralatrexate | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06813 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Pralatrexate | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7786 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Off-white to yellow solid | |
CAS No. |
146464-95-1 | |
| Record name | Pralatrexate | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=146464-95-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Pralatrexate [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0146464951 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Pralatrexate | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06813 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Pralatrexate | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=754230 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Pralatrexate | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3048578 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (2S)-2-[[4-[4-[(1RS)-1-[(2,4-diaminopteridin-6-yl)methyl]but-3-ynyl]benzoyl]amino]pentanedioc acid | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PRALATREXATE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/A8Q8I19Q20 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Pralatrexate | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7786 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















