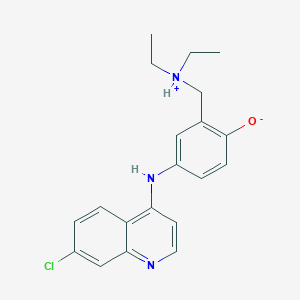
Amodiaquine
Overview
Description
Amodiaquine is a synthetic compound belonging to the class of 4-aminoquinolines. It is primarily used as an antimalarial drug, effective against Plasmodium falciparum malaria, especially in regions where resistance to other antimalarial drugs like chloroquine has developed . This compound is often used in combination with artesunate to enhance its efficacy and reduce the risk of resistance .
Preparation Methods
Synthetic Routes and Reaction Conditions
Amodiaquine is synthesized through a multi-step process involving the reaction of 4,7-dichloroquinoline with 4-aminophenol in the presence of a base. The reaction proceeds through nucleophilic aromatic substitution, resulting in the formation of the intermediate compound, which is then further reacted with diethylamine to yield this compound .
Industrial Production Methods
Industrial production of this compound involves large-scale synthesis using similar reaction conditions as in the laboratory synthesis. The process is optimized for high yield and purity, with stringent quality control measures to ensure the final product meets pharmaceutical standards .
Chemical Reactions Analysis
Types of Reactions
Amodiaquine undergoes various chemical reactions, including:
Oxidation: This compound can be oxidized to form its primary metabolite, N-desethylthis compound.
Reduction: Reduction reactions are less common but can occur under specific conditions.
Substitution: Nucleophilic substitution reactions are involved in its synthesis and modification.
Common Reagents and Conditions
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents like sodium borohydride can be used.
Substitution: Bases such as sodium hydroxide are used in nucleophilic substitution reactions.
Major Products Formed
The major product formed from the oxidation of this compound is N-desethylthis compound, which retains antimalarial activity .
Scientific Research Applications
Antimalarial Activity
Primary Use in Malaria Treatment
Amodiaquine is widely used as an antimalarial drug, particularly in combination therapies. The World Health Organization (WHO) recommends artesunate-amodiaquine (ASAQ) as a first-line treatment for uncomplicated Plasmodium falciparum malaria in many endemic regions. It is also used in seasonal malaria chemoprevention (SMC) for children aged 3 to 59 months in areas with high malaria transmission rates .
Efficacy Studies
Recent studies have demonstrated that ASAQ maintains high efficacy across various demographics, including vulnerable populations such as infants and underweight children. A pharmacokinetic study indicated that this compound exposure was not significantly reduced in these groups, suggesting its safe use in treating malaria .
Cardiovascular Effects
Safety Profile and Adverse Reactions
While generally well-tolerated, this compound has been associated with cardiovascular effects such as QT interval prolongation and sinus bradycardia. Research indicates that these effects are less pronounced compared to other antimalarials like chloroquine and lumefantrine . Understanding these cardiovascular implications is crucial for optimizing its therapeutic use.
Potential Beyond Antimalarial Use
Cholinesterase Inhibition
Emerging research suggests that this compound derivatives may exhibit significant cholinesterase inhibitory activity, indicating potential applications in treating neurodegenerative diseases such as Alzheimer's disease (AD). These derivatives could serve as multitarget drugs that not only inhibit cholinesterase but also mitigate oxidative stress associated with AD .
Antioxidant Properties
Redox Chemistry
this compound's redox properties have been investigated for their antioxidant potential. Studies show that it can donate electrons effectively, which may contribute to its protective effects against oxidative damage. This property opens avenues for further research into its use as a therapeutic agent in conditions characterized by oxidative stress .
Case Studies and Clinical Trials
Clinical Efficacy of ASAQ
A clinical trial comparing ASAQ to artemether-lumefantrine (AL) demonstrated superior efficacy of ASAQ in certain populations, with a high cure rate observed across multiple sites . Such findings reinforce the importance of this compound in current malaria treatment protocols.
Summary Table of Applications
Mechanism of Action
Amodiaquine exerts its antimalarial effects by inhibiting heme polymerase activity in the malaria parasite. This inhibition leads to the accumulation of free heme, which is toxic to the parasite. The drug binds to free heme, preventing the parasite from converting it to a less toxic form, thereby disrupting membrane function and leading to the death of the parasite . The primary molecular target is Fe(II)-protoporphyrin IX .
Comparison with Similar Compounds
Amodiaquine is similar to other 4-aminoquinoline compounds such as chloroquine, mefloquine, and piperaquine . it has unique properties that make it effective against chloroquine-resistant strains of Plasmodium falciparum . Unlike chloroquine, this compound is often used in combination therapies to enhance its efficacy and reduce resistance .
List of Similar Compounds
- Chloroquine
- Mefloquine
- Piperaquine
- Lumefantrine
- Primaquine
- Tafenoquine
Biological Activity
Amodiaquine (AQ) is a 4-aminoquinoline derivative primarily used as an antimalarial drug. Its biological activity extends beyond malaria treatment, showing potential in various other therapeutic areas. This article reviews the biological mechanisms, efficacy, safety, and additional applications of this compound, supported by diverse research findings and case studies.
This compound operates through several mechanisms that contribute to its antimalarial activity:
- Accumulation in Plasmodium falciparum : AQ accumulates in malaria parasites at levels 2-3 times greater than chloroquine. This accumulation is facilitated by a transmembrane proton gradient maintained by vacuolar ATPase, highlighting the energy-dependent nature of its uptake . The binding affinity of AQ within the parasite may also explain its superior efficacy compared to chloroquine .
- Host-Targeting Mechanism : Recent studies have identified AQ as a host-oriented inhibitor of anthrax toxin endocytosis. It reduces bacterial burden in Bacillus anthracis-infected models, suggesting that its antibacterial activity may stem from modulating host immune responses rather than direct pathogen inhibition .
- Antiviral Activity : this compound has shown antiviral properties against the Ebola virus (EBOV). In vitro studies indicate that both AQ and its active metabolite, desethylthis compound (DEAQ), inhibit EBOV replication with IC50 values ranging from 2.8 to 3.2 µM in human cell lines . However, its efficacy in vivo remains limited.
Efficacy in Malaria Treatment
A systematic review encompassing 56 studies indicated that this compound is significantly more effective than chloroquine for clearing malaria parasites. Key findings include:
- Parasite Clearance Rates : On day 7, the Peto odds ratio for AQ versus CQ was 4.42 (95% CI: 3.65–5.35), and on day 14, it was 6.44 (95% CI: 5.09–8.15) .
- Comparison with Sulfadoxine-Pyrimethamine : While AQ was generally more effective than CQ, comparisons with sulfadoxine-pyrimethamine yielded mixed results; the latter demonstrated superior effectiveness on day 28 .
Table 1: Summary of Efficacy Studies
| Study Type | Comparison | Day | Peto Odds Ratio | Confidence Interval |
|---|---|---|---|---|
| Systematic Review | This compound vs Chloroquine | 7 | 4.42 | (3.65 - 5.35) |
| Systematic Review | This compound vs Chloroquine | 14 | 6.44 | (5.09 - 8.15) |
| Mixed Studies | This compound vs Sulfadoxine-Pyrimethamine | 28 | Not specified | Not specified |
Safety Profile
This compound is generally well tolerated, with adverse effects primarily being minor or moderate:
- Common Side Effects : Gastrointestinal disorders and pruritus were reported in about 2.5% of patients during clinical trials . Serious adverse events are rare, with no life-threatening incidents documented in major studies.
- Case Reports : A notable case of this compound-induced agranulocytosis was reported in a patient four months post-treatment, indicating potential hematological side effects that warrant monitoring .
Case Studies and Clinical Trials
- Artesunate + this compound Combination Therapy : A randomized controlled trial conducted in Madagascar evaluated artesunate combined with this compound against artemether-lumefantrine for treating uncomplicated malaria. The study reported a crude adequate clinical and parasitological response rate of 100% for the ASAQ group after a follow-up period .
- Comparative Study on Dosage Regimens : A study involving 316 patients demonstrated non-inferiority between one daily intake versus two daily intakes of artesunate/amodiaquine, confirming high efficacy rates above 99% for both regimens .
Table 2: Clinical Trial Outcomes
| Treatment Regimen | Day 14 Response Rate (%) |
|---|---|
| Artesunate + this compound (One Intake) | 99.4 |
| Artesunate + this compound (Two Intakes) | 99.3 |
| Total Patients Evaluated | 316 |
Q & A
Basic Research Questions
Q. How is the anti-cancer activity of amodiaquine assessed in vitro, and what methodological considerations ensure reproducibility?
- Answer: Anti-cancer activity is evaluated using cell viability assays (e.g., MTT or resazurin) to determine IC50 values across breast cancer cell lines (e.g., MDA-MB-231). Flow cytometry is employed to differentiate apoptosis and necrosis. Key steps include:
- Validating assay conditions (e.g., λmax alignment with reference standards for drug quantification) .
- Using two-way ANOVA for statistical analysis to compare treated vs. control groups, with significance thresholds (e.g., p < 0.001) .
- Reporting data as mean ± SD to account for variability .
Q. What pharmacokinetic (PK) parameters are critical for evaluating this compound in clinical trials, and how are they derived?
- Answer: Key parameters include:
- AUC (Area Under the Curve): Measures total drug exposure, calculated via non-compartmental analysis .
- Cmax (Maximum Concentration): Determined from plasma concentration-time curves .
- Metabolite analysis: Desethylthis compound (active metabolite) is prioritized due to rapid conversion from this compound .
- Compartment modeling: A two-compartment model for this compound and one-compartment for desethylthis compound, with first-order absorption kinetics .
Q. How are crossover study designs implemented to assess drug-drug interactions, such as with herbal decoctions?
- Answer: In a three-period crossover trial:
- Subjects receive this compound alone, herbal decoction (e.g., MAMADecoction) alone, and both combined .
- Blood samples are collected at predefined intervals to compare PK parameters (e.g., volume of distribution, clearance) .
- Statistical models (e.g., mixed-effect regression) adjust for period and sequence effects .
Advanced Research Questions
Q. How do researchers resolve contradictions in cardiovascular safety data for this compound across demographic groups?
- Answer: Individual patient data meta-analyses (IPD-MA) are conducted to:
- Pool data from randomized controlled trials (RCTs) (e.g., 2,681 patients across four RCTs) .
- Stratify analyses by age (e.g., ≥12 years vs. <12 years) to identify differential risks (e.g., sinus bradycardia in adolescents/adults) .
- Use study-specific heart rate corrections (QTcS) to compare QT prolongation against other antimalarials (e.g., piperaquine, lumefantrine) .
Q. What advanced modeling techniques are used to predict this compound distribution in organ-on-chip systems?
- Answer: Microfluidic lung chips simulate human tissue interfaces:
- Drug adsorption to chip walls is quantified via mass spectrometry of effluent samples .
- Log P (partition coefficient) is estimated experimentally (log P = 1.3–1.8) rather than relying on theoretical values (log P = 3.7), accounting for extracellular matrix effects .
- Simulations iteratively fit experimental data to refine permeability coefficients (e.g., P = 20–60) .
Q. How are herb-drug interactions (e.g., MAMADecoction) mechanistically explored in this compound pharmacokinetics?
- Answer: Mechanistic PK models identify:
- Reduced apparent volume of distribution (VAQ/F) by 41% when co-administered with herbal decoctions, suggesting altered tissue partitioning .
- Phytochemical-induced diuresis may enhance renal clearance, redistributing this compound from tissues to blood .
- Sensitivity analyses validate model robustness, censoring data below LLOQ (lower limit of quantification) .
Q. What methodologies address conflicting preclinical data on this compound’s potential for COVID-19 repurposing?
- Answer: Preclinical validation includes:
- Head-to-head comparisons in animal models (e.g., hamsters) with hydroxychloroquine as a control .
- Subcutaneous delivery with solubilizers (e.g., sulfobutylether-β-cyclodextrin) to enhance bioavailability .
- Caution in extrapolating surrogate virus results (e.g., human-lung-cell assays) due to model limitations .
Q. Methodological Best Practices
Q. How should researchers document experimental protocols to ensure reproducibility in this compound studies?
- Guidelines:
- Report randomization procedures, blinding status, and outcome definitions explicitly (e.g., WHO clinical trial critiques) .
- Provide raw data tables in appendices and processed data in the main text, adhering to journal standards .
- Reference established protocols for new methods (e.g., USP monographs for λmax validation) .
Q. What statistical approaches mitigate bias in analyzing this compound’s hematological effects?
- Approach:
- Use log-transformed ANOVA for cross-over designs to account for within-patient variability .
- Apply mixed-effect models for longitudinal data (e.g., platelet counts), adjusting for period-treatment interactions .
Properties
IUPAC Name |
4-[(7-chloroquinolin-4-yl)amino]-2-(diethylaminomethyl)phenol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C20H22ClN3O/c1-3-24(4-2)13-14-11-16(6-8-20(14)25)23-18-9-10-22-19-12-15(21)5-7-17(18)19/h5-12,25H,3-4,13H2,1-2H3,(H,22,23) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OVCDSSHSILBFBN-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCN(CC)CC1=C(C=CC(=C1)NC2=C3C=CC(=CC3=NC=C2)Cl)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C20H22ClN3O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID2022597 | |
| Record name | Amodiaquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022597 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
355.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Amodiaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014751 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
24.9 [ug/mL] (The mean of the results at pH 7.4), 8.80e-03 g/L | |
| Record name | SID50085969 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Amodiaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014751 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The mechanism of plasmodicidal action of amodiaquine is not completely certain. Like other quinoline derivatives, it is thought to inhibit heme polymerase activity. This results in accumulation of free heme, which is toxic to the parasites. The drug binds the free heme preventing the parasite from converting it to a form less toxic. This drug-heme complex is toxic and disrupts membrane function., Amodiaquine is a Mannich base 4-aminoquinoline with a mode of action similar to that of chloroquine. It is effective against some chloroquine-resistant strains of P. falciparum, although there is cross-resistance., The 4-aminoquinoline derivatives appear to bind to nucleoproteins and interfere with protein synthesis in susceptible organisms; the drugs intercalate readily into double-stranded DNA and inhibit both DNA and RNA polymerase. In addition, the drugs apparently concentrate in parasite digestive vacuoles, increase the pH of the vacuoles, and interfere with the parasite's ability to metabolize and utilize erythrocyte hemoglobin. Plasmodial forms that do not have digestive vacuoles and do not utilize hemoglobin, such as exoerythrocytic forms, are not affected by /these medications/., The 4-aminoquinoline derivatives ... have anti-inflammatory activity; however, the mechanism(s) of action of the drugs in the treatment of rheumatoid arthritis and lupus erythematosus has not been determined. /4-aminoquinoline derivatives/ reportedly antagonizes histamine in vitro, has antiserotonin effects, and inhibits prostaglandin effects in mammalian cells presumably by inhibiting conversion of arachidonic acid to prostaglandin F2., The mode of action of amodiaquine has not yet been determined. 4-Aminoquinolines depress cardiac muscle, impair cardiac conductivity, and produce vasodilatation with resultant hypotension; they depress respiration and cause diplopia, dizziness and nausea. | |
| Record name | Amodiaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00613 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | AMODIAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7457 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from absolute ethanol | |
CAS No. |
86-42-0 | |
| Record name | Amodiaquine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=86-42-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Amodiaquine [USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000086420 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Amodiaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00613 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | amodiaquine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=13453 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Amodiaquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022597 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Amodiaquine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.001.518 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | AMODIAQUINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/220236ED28 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | AMODIAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7457 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Amodiaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014751 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
206-208, 208 °C (decomposes), Yellow crystals from methanol. Melting point 243 °C. Slightly soluble in water and alcohol /Amodiaquine dihydrochloride hemihydrate/, 208 °C | |
| Record name | Amodiaquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00613 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | AMODIAQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7457 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Amodiaquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014751 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















