Spermine
Overview
Description
Spermine is a naturally occurring polyamine involved in cellular metabolism and found in all eukaryotic cells. It is derived from the amino acid ornithine and plays a crucial role in stabilizing the helical structure of nucleic acids, particularly in viruses . This compound is also known for its role as an intracellular free radical scavenger, protecting DNA from oxidative damage .
Preparation Methods
Synthetic Routes and Reaction Conditions: Spermine is synthesized from spermidine through the action of the enzyme this compound synthase. The process involves the transfer of an aminopropyl group to spermidine . The reaction conditions typically require the presence of decarboxylated S-adenosyl methionine as a cofactor .
Industrial Production Methods: Industrial production of this compound involves the decarboxylation of ornithine to produce putrescine, which is then converted to spermidine. Finally, spermidine is transformed into this compound through N-alkylation reactions . The process is optimized for large-scale production by controlling reaction conditions such as temperature, pH, and the concentration of reactants .
Chemical Reactions Analysis
Types of Reactions: Spermine undergoes various chemical reactions, including oxidation, reduction, and substitution .
Common Reagents and Conditions:
Reduction: Reduction reactions involving this compound are less common but can occur under specific conditions with reducing agents.
Substitution: this compound can participate in substitution reactions, particularly in the presence of strong nucleophiles.
Major Products: The major products formed from these reactions include hydrogen peroxide, aminoaldehydes, and various substituted this compound derivatives .
Scientific Research Applications
Anticancer Properties
Spermine has been extensively studied for its anticancer properties. Research indicates that it interferes with tumor cell cycles, inhibiting proliferation and promoting apoptosis in cancer cells.
- Mechanism of Action : this compound induces autophagy, which is crucial for cellular homeostasis and can suppress oncogenesis. It regulates key oncogenic pathways, enhancing the immune response against tumors .
- Clinical Implications : Chemically modified derivatives of this compound are being explored for diagnostic and therapeutic applications in various malignancies. For instance, studies have shown that this compound N-1-acetyltransferase (SSAT) serves as a diagnostic marker in human cancers, indicating its potential role in cancer prognosis .
Neuroprotective Effects
This compound exhibits neuroprotective properties that may reduce the risk of neurodegenerative diseases.
- Cognitive Decline : Increased intake of this compound has been associated with a decrease in cognitive decline due to its ability to clear amyloid-beta plaques in the brain. This suggests a protective role against conditions like Alzheimer's disease .
- Mitochondrial Function : this compound supplementation enhances mitochondrial metabolism and translational activity, further supporting neuronal health .
Ocular Health
Recent studies have identified this compound's potential in treating various ocular diseases.
- Therapeutic Applications : this compound has shown promise in treating conditions such as glaucoma, age-related macular degeneration (AMD), and dry eye syndrome. Its protective effects against oxidative stress contribute to improved ocular health .
- Mechanisms : By acting as an antioxidant and modulating calcium levels within cells, this compound helps to prevent apoptosis induced by oxidative stress .
Gastrointestinal Health
This compound's role extends to gastrointestinal health, particularly concerning inflammatory conditions.
- Antidiarrheal Effects : Research indicates that this compound can mitigate secretory diarrhea induced by cholera toxin and other pathogens by regulating calcium-sensing receptor (CaSR) pathways .
Metabolic Regulation
This compound influences metabolic processes that can protect against metabolic diseases.
- Metabolic Diseases : Increased levels of this compound have been linked to reduced risks of metabolic disorders, including obesity and diabetes . Its role in modulating insulin sensitivity is an area of ongoing research.
Case Studies
Mechanism of Action
Spermine exerts its effects through several mechanisms:
Free Radical Scavenging: this compound acts as a free radical scavenger, protecting DNA from oxidative damage.
Gene Regulation: It is involved in the regulation of gene expression and the stabilization of chromatin.
Interaction with Molecular Targets: this compound binds to DNA, preventing endonuclease-mediated DNA fragmentation and stabilizing the helical structure.
Comparison with Similar Compounds
Spermine is part of a family of polyamines that includes spermidine, putrescine, and cadaverine .
Spermidine: Similar to this compound but with one less aminopropyl group.
Putrescine: A precursor to both spermidine and this compound, it plays a role in cell growth and differentiation.
Cadaverine: Structurally similar but with different biological functions, primarily involved in bacterial metabolism.
This compound is unique due to its higher number of amino groups, which enhances its ability to stabilize nucleic acids and protect against oxidative damage .
Biological Activity
Spermine, a polyamine compound, is involved in various biological processes and exhibits significant physiological activities. This article delves into the biological activity of this compound, highlighting its mechanisms, effects on cellular processes, and potential therapeutic applications.
Overview of this compound
This compound is a naturally occurring polyamine derived from spermidine through the action of spermidine synthase. It plays crucial roles in cellular functions such as growth, differentiation, and apoptosis. Its positive charge allows it to interact with nucleic acids and proteins, influencing various biochemical pathways.
This compound exerts its biological effects through several mechanisms:
- Regulation of Ion Channels : this compound modulates the activity of potassium channels and other ion channels, impacting cellular excitability and signaling pathways .
- Inhibition of Protein Synthesis : Research indicates that this compound can repress protein synthesis in mammalian cells, particularly during oocyte maturation .
- Reactive Oxygen Species (ROS) Generation : this compound has been shown to enhance ROS production, which is crucial for its antimycobacterial activity against Mycobacterium tuberculosis (M.tb) .
Biological Activities
The biological activities of this compound can be categorized as follows:
1. Cell Growth and Proliferation
This compound is essential for cell proliferation. It influences the cell cycle by promoting progression through the G1 phase and enhancing mitochondrial function. Studies have shown that this compound supplementation can increase the proportion of cells in the S phase, facilitating growth and division .
2. Antimicrobial Activity
This compound exhibits antimicrobial properties, particularly against M.tb. It enhances the efficacy of existing tuberculosis treatments and has been proposed as a less toxic alternative to conventional antibiotics due to its natural occurrence in human tissues .
3. Role in Aging and Longevity
This compound has been implicated in longevity mechanisms. It promotes autophagy, reduces inflammation, and regulates metabolic pathways associated with aging. Elevated levels of this compound have been linked to improved healthspan in various model organisms .
Case Studies
Several studies highlight the diverse roles of this compound:
- Oocyte Maturation : A study demonstrated that this compound inhibits progesterone-induced maturation in oocytes by blocking protein synthesis, indicating its regulatory role in reproductive biology .
- Tuberculosis Treatment : In a clinical study, this compound was shown to enhance the activity of standard tuberculosis drugs, suggesting its potential use as an adjunct therapy for drug-resistant strains .
- Aging Research : Research on fasting regimens indicated that this compound levels increase significantly during caloric restriction, correlating with enhanced autophagy and longevity markers in humans .
Data Table: Effects of this compound on Cellular Processes
Q & A
Basic Research Questions
Q. What are the established methods for quantifying spermine levels in eukaryotic cell cultures, and how do their accuracy compare in different experimental conditions?
- Methodological Answer : this compound quantification typically employs high-performance liquid chromatography (HPLC) coupled with fluorescence detection or mass spectrometry (LC-MS/MS). For cell cultures, sample preparation involves acid extraction to stabilize polyamines, followed by derivatization (e.g., dansyl chloride) for enhanced detection . Researchers must validate methods using internal standards (e.g., 1,6-hexanediamine) to account for matrix effects. Accuracy varies: LC-MS/MS offers higher specificity for low-concentration samples, while fluorescence-based HPLC is cost-effective for high-throughput screens. Include controls for pH-dependent degradation during extraction .
Q. What experimental controls are essential when investigating this compound's effect on ion channel activity in electrophysiological studies?
- Methodological Answer : Key controls include:
- Blank controls : Measure baseline currents in the absence of this compound.
- Pharmacological blockers : Use specific inhibitors (e.g., Ba²⁺ for inward rectifier K⁺ channels) to confirm this compound’s target specificity .
- Concentration gradients : Test dose-response relationships (0.1–10 mM) to assess saturation effects.
- Cell viability assays : Ensure observed effects are not artifacts of cytotoxicity. Document buffer composition (e.g., Mg²⁺/ATP levels) as these modulate this compound-channel interactions .
Advanced Research Questions
Q. How can researchers resolve contradictory findings regarding this compound's dual role in promoting and inhibiting apoptosis across different cancer cell models?
- Methodological Answer : Contradictions often arise from context-dependent factors:
- Cell-type specificity : Profile apoptotic markers (e.g., caspase-3, Bcl-2) in paired cell lines (e.g., HeLa vs. MCF-7) under identical this compound concentrations.
- Microenvironment modulation : Replicate physiological conditions (e.g., hypoxia, serum starvation) to test if this compound’s redox activity (pro-oxidant vs. antioxidant) dictates apoptotic outcomes .
- Omics integration : Combine metabolomics (this compound/spermidine ratios) with transcriptomics (p53, NF-κB pathways) to identify upstream regulators. Use siRNA knockdown of polyamine transporters to isolate extracellular vs. intracellular effects .
Q. What integrative approaches are recommended for correlating this compound metabolic flux with transcriptomic changes under oxidative stress conditions?
- Methodological Answer :
- Isotopic tracing : Use ¹³C-labeled putrescine to track this compound biosynthesis flux via spermidine synthase and this compound synthase. Pair with RNA-seq to link flux alterations to gene expression (e.g., SAT1, SMS) .
- Multi-omics alignment : Apply pathway enrichment analysis (e.g., KEGG, Reactome) to overlay flux data with differentially expressed genes. Validate using CRISPR-Cas9 knockout models of rate-limiting enzymes (e.g., AMD1) .
- Time-course designs : Capture dynamic interactions by sampling at intervals (0–24 hrs post-stress) to distinguish primary transcriptional responses from secondary metabolic adaptations .
Q. How should researchers optimize in vitro models to study this compound’s role in neurodegenerative protein aggregation (e.g., tau, α-synuclein)?
- Methodological Answer :
- Model selection : Use neuronal cell lines (SH-SY5Y, iPSC-derived neurons) with inducible aggregation systems. Include isogenic controls to isolate this compound-specific effects .
- Aggregation assays : Combine thioflavin-T fluorescence with super-resolution microscopy to quantify fibril formation. Pre-treat cells with DFMO (a polyamine synthesis inhibitor) to deplete endogenous this compound and establish baseline aggregation .
- Cross-validation : Compare results across 3D spheroid models and in vivo (e.g., Drosophila) to assess translational relevance. Monitor autophagic flux (LC3-II/p62) to determine if this compound modulates clearance mechanisms .
Q. Guidance for Addressing Methodological Challenges
- Data Contradictions : Perform meta-analyses of published datasets (e.g., GEO, PRIDE) to identify confounding variables (e.g., cell cycle stage, nutrient availability) .
- Reproducibility : Adhere to FAIR principles (Findable, Accessible, Interoperable, Reusable) by documenting experimental parameters (e.g., buffer pH, exact this compound salt form) in supplemental materials .
- Ethical Considerations : For animal studies, include polyamine-depletion rescue experiments to distinguish physiological roles from pharmacological effects .
Properties
IUPAC Name |
N,N'-bis(3-aminopropyl)butane-1,4-diamine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H26N4/c11-5-3-9-13-7-1-2-8-14-10-4-6-12/h13-14H,1-12H2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
PFNFFQXMRSDOHW-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
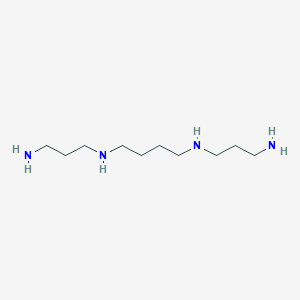
Canonical SMILES |
C(CCNCCCN)CNCCCN | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H26N4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
71052-31-8 | |
| Record name | 1,4-Butanediamine, N1,N4-bis(3-aminopropyl)-, homopolymer | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=71052-31-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
DSSTOX Substance ID |
DTXSID9058781 | |
| Record name | N1,N4-Bis(3-aminopropyl)-1,4-butanediamine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9058781 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
202.34 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid; Absorbs carbon dioxide from air; [Merck Index] Colorless solidified mass or fragments; mp = 28-30 deg C; [Sigma-Aldrich MSDS], Solid | |
| Record name | Spermine | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/21744 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
| Record name | Spermine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001256 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Boiling Point |
150-150 °C | |
| Record name | Spermine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00127 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Solubility |
> 100 mg/mL | |
| Record name | Spermine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00127 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Spermine is derived from spermidine by spermine synthase. Spermine is a polyamine, a small organic cations that is absolutely required for eukaryotic cell growth. Spermine, is normally found in millimolar concentrations in the nucleus. Spermine functions directly as a free radical scavenger, and forms a variety of adducts that prevent oxidative damage to DNA. Oxidative damage to DNA by reactive oxygen species is a continual problem that cells must guard against to survive. Hence, spermine is a major natural intracellular compound capable of protecting DNA from free radical attack. Spermine is also implicated in the regulation of gene expression, the stabilization of chromatin, and the prevention of endonuclease-mediated DNA fragmentation. | |
| Record name | Spermine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00127 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
71-44-3, 68956-56-9 | |
| Record name | Spermine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=71-44-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Spermine | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000071443 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Hydrocarbons, terpene processing by-products | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0068956569 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Spermine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00127 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | spermine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=268508 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 1,4-Butanediamine, N1,N4-bis(3-aminopropyl)- | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | N1,N4-Bis(3-aminopropyl)-1,4-butanediamine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9058781 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 4,9-diazadodecamethylenediamine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.686 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | SPERMINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/2FZ7Y3VOQX | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Spermine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001256 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
29 °C | |
| Record name | Spermine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00127 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Spermine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0001256 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














