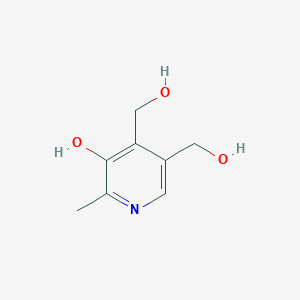
Pyridoxine
Overview
Description
Pyridoxine, a water-soluble vitamin B6 vitamer, is essential for amino acid metabolism, neurotransmitter synthesis, and hemoglobin formation. It exists alongside pyridoxal and pyridoxamine, collectively termed "vitamin B6," and is converted in vivo to its active coenzyme form, pyridoxal 5'-phosphate (PLP) . Dietary sources include meats, grains, and vegetables. This compound is also used therapeutically, such as in managing isoniazid-induced seizures and chemotherapy-related side effects like hand-foot syndrome (HFS) .
Preparation Methods
Synthetic Routes and Reaction Conditions: The industrial synthesis of pyridoxine typically involves the condensation of cyanoacetamide with 1,3-dicarbonyl compounds. This method, developed in the mid-20th century, has been refined over the years to increase yield and reduce costs. The process involves the formation of 1,3-oxazole derivatives, which undergo diene condensation to form pyridine bases. These intermediates are then converted into this compound through catalytic hydrogenation .
Industrial Production Methods: this compound hydrochloride is the main commercial form of vitamin B6. It is produced by several pharmaceutical companies worldwide, including those in the United States, Switzerland, Japan, and Germany. The industrial production process involves the use of symmetric ethylene derivatives as dienophiles, which react with substituted oxazoles to form this compound intermediates. These intermediates are then subjected to catalytic hydrogenation to yield this compound .
Chemical Reactions Analysis
Types of Reactions: Pyridoxine undergoes various chemical reactions, including oxidation, reduction, and substitution. It can react with metal ions to form complexes, which have been studied for their potential biological activities .
Common Reagents and Conditions:
Oxidation: this compound can be oxidized to pyridoxal using oxidizing agents such as potassium permanganate or hydrogen peroxide.
Reduction: this compound can be reduced to pyridoxamine using reducing agents like sodium borohydride.
Substitution: this compound can undergo nucleophilic substitution reactions, where the hydroxyl group is replaced by other functional groups.
Major Products:
Oxidation: Pyridoxal
Reduction: Pyridoxamine
Substitution: Various substituted this compound derivatives, depending on the nucleophile used.
Scientific Research Applications
Biochemical Functions of Pyridoxine
This compound is essential for over 100 enzymatic reactions in the human body. It primarily exists in three natural forms: this compound (PN), pyridoxal (PL), and pyridoxamine (PM), which are converted into the active coenzyme form, pyridoxal 5'-phosphate (PLP). PLP is vital for:
- Amino Acid Metabolism : Involved in transamination and decarboxylation reactions.
- Neurotransmitter Synthesis : Facilitates the production of neurotransmitters such as serotonin, dopamine, norepinephrine, and gamma-aminobutyric acid (GABA).
- Glycogenolysis and Gluconeogenesis : Plays a role in glucose metabolism.
- Hemoglobin Synthesis : Contributes to the formation of hemoglobin and other important biomolecules like sphingolipids .
Treatment of Vitamin B6 Deficiency
This compound is primarily used to treat vitamin B6 deficiency, which can lead to symptoms such as seborrheic dermatitis, microcytic anemia, and peripheral neuropathy. Deficiency may arise from various factors including poor dietary intake, certain medications (e.g., isoniazid), and metabolic disorders .
Neurological Disorders
This compound has been shown to alleviate neurological symptoms associated with vitamin B12 deficiency when used in conjunction with cobalamin therapy. Its role in neurotransmitter synthesis makes it a candidate for managing conditions like depression and seizures .
Pregnancy-Related Nausea
This compound is approved for use in combination with doxylamine to treat nausea and vomiting during pregnancy. This combination is marketed under the brand name Diclectin .
Cardiovascular Health
Research indicates that this compound may lower blood pressure and improve lipid profiles by reducing total cholesterol levels while increasing HDL cholesterol levels. This suggests potential benefits for cardiovascular health .
Emerging Research Findings
Recent studies have explored the broader implications of this compound supplementation:
- Anti-inflammatory Properties : High-dose vitamin B6 has demonstrated strong anti-inflammatory effects, potentially benefiting individuals with inflammatory-related conditions .
- Immune Function : this compound plays a role in antibody production and overall immune response, indicating its importance in maintaining immune health .
- Cancer Research : Some studies suggest that increased intake of vitamin B6 may be linked to a decreased risk of colorectal cancer, highlighting its potential protective effects against certain malignancies .
Case Studies and Clinical Trials
Mechanism of Action
Pyridoxine is part of the vitamin B6 group, which includes pyridoxal and pyridoxamine. These compounds share similar structures and functions but differ in their specific roles and activities:
Pyridoxal: Involved in the synthesis of neurotransmitters and the metabolism of amino acids.
Pyridoxamine: Plays a role in the metabolism of amino acids and the synthesis of nucleic acids.
Uniqueness of this compound: this compound is unique in its ability to be converted into pyridoxal 5’-phosphate, the active coenzyme form that participates in a wide range of biochemical reactions. Its stability and availability in various dietary sources make it an essential nutrient for human health .
Comparison with Similar Compounds
Structural and Functional Comparison with Related Compounds
Key Vitamin B6 Vitamers
- Pyridoxine : Contains a hydroxymethyl group at the 4-position.
- Pyridoxal : Features an aldehyde group at the 4-position.
- Pyridoxamine: Substitutes the hydroxymethyl group with an aminomethyl group.
Structural Implications :
- The aldehyde group in pyridoxal enhances its enzymatic activity, particularly in α-glucosidase inhibition (IC₅₀: 4.15 mg/mL vs. This compound’s 5.02 mg/mL) .
- Cationic properties of this compound and pyridoxamine facilitate recognition by transporters SLC19A2/SLC19A3, unlike anionic derivatives like PLP or 4-pyridoxic acid .
Transport Mechanisms and Substrate Specificity
Role of SLC19 Transporters
- SLC19A2 and SLC19A3 : These proton-coupled transporters mediate this compound uptake in intestinal and pancreatic cells. They exhibit multispecificity, also transporting thiamine and cationic compounds .
- Competitive Inhibition : Pyridoxamine and pyridoxal compete with this compound for binding, suggesting shared structural recognition motifs .
- Species Differences : Human SLC19A3 requires specific transmembrane domains (TMDs 3, 4, 6) for this compound transport, which are absent in murine homologs .
Enzymatic and Metabolic Roles
α-Glucosidase Inhibition
Pyridoxal demonstrates superior inhibition (79.83% at 7 mg/mL) compared to this compound and pyridoxamine, attributed to its aldehyde group enhancing interaction with the enzyme’s active site .
Coenzyme Activity
- PLP Dependency : Enzymes like homoserine/cystathionine lyase require PLP for activation. This compound, pyridoxamine, and their phosphate derivatives fail to activate these enzymes, underscoring PLP’s irreplaceable role .
- Metabolic Interconversion : this compound is phosphorylated to this compound 5'-phosphate (PNP) and oxidized to PLP via pyridoxamine phosphate oxidase (PNPO) .
Chemotherapy-Induced Toxicity Mitigation
- This compound prophylaxis reduces HFS incidence in multiple myeloma patients treated with pegylated liposomal doxorubicin (PLD).
Neuroprotective and Cardiovascular Effects
- Alzheimer’s Disease: this compound-derived dual inhibitors of acetylcholinesterase (AChE) and monoamine oxidase-B (MAO-B) show promise in preclinical models .
- Diabetic Cardiomyopathy : this compound supplementation ameliorates oxidative stress and metabolic dysregulation in diabetic rats, though mechanisms remain under investigation .
Comparative Clinical Efficacy
- Hyperkinesis Management : this compound elevates serotonin levels in hyperkinetic children, matching methylphenidate’s symptom suppression but with sustained post-treatment effects .
- Antiemetic Use: Doxylamine/pyridoxine combinations outperform placebos in reducing pregnancy-related nausea, though somnolence is a noted side effect .
Degradation Pathways and Toxicity
Degradation Pathways
- Bacterial Degradation : Arthrobacter spp. utilize this compound 4-oxidase and pyridoxal 4-dehydrogenase to convert this compound into 4-pyridoxolactone, a key anabolic intermediate .
Cytotoxicity Profile
| Compound | IC₅₀ (Normal Cells) | IC₅₀ (Tumor Cells) |
|---|---|---|
| This compound | >10 mM | ~8 mM |
| B6NO* | >10 mM | ~6 mM |
*B6NO: A this compound-derived nitric oxide donor. Tumor cells (HepG2) show greater sensitivity, suggesting selective toxicity .
Biological Activity
Pyridoxine, commonly known as Vitamin B6, is a water-soluble vitamin that plays a crucial role in numerous biological processes within the human body. This article explores the biological activity of this compound, its mechanisms of action, clinical applications, and relevant research findings.
Overview of this compound
This compound is one of the three main forms of Vitamin B6, along with pyridoxal and pyridoxamine. It is primarily involved in amino acid metabolism, neurotransmitter synthesis, and the regulation of homocysteine levels. The active form of this compound is pyridoxal 5'-phosphate (PLP), which serves as a coenzyme in various enzymatic reactions.
This compound exerts its biological effects through several mechanisms:
- Amino Acid Metabolism : PLP is essential for transamination reactions, where amino groups are transferred between amino acids and keto acids. This process is vital for amino acid synthesis and degradation.
- Neurotransmitter Synthesis : this compound is crucial for synthesizing neurotransmitters such as serotonin, dopamine, norepinephrine, and gamma-aminobutyric acid (GABA) from their respective precursors .
- Glycogenolysis : It plays a role in glycogen metabolism by facilitating the conversion of glycogen to glucose .
1. Vitamin B6 Deficiency
Vitamin B6 deficiency can lead to various health issues, including:
- Neurological Disorders : Symptoms may include irritability, depression, and confusion due to impaired neurotransmitter synthesis.
- Dermatological Issues : Deficiency can result in seborrheic dermatitis and cheilosis .
2. Clinical Uses
This compound is used therapeutically in several conditions:
- Nausea and Vomiting in Pregnancy : It is often combined with doxylamine to alleviate symptoms associated with morning sickness .
- Isoniazid-Induced Peripheral Neuropathy : this compound supplementation is recommended to prevent neuropathy caused by isoniazid treatment for tuberculosis .
Research Findings
Recent studies have highlighted the complex role of this compound in health:
- Cellular Metabolism : Research indicates that this compound influences cellular metabolism significantly by regulating enzyme activity associated with PLP .
- Toxicity Concerns : High doses of this compound have been linked to potential toxicity, particularly in patients with specific genetic disorders such as primary hyperoxaluria type 1 (PH1), where excessive supplementation can exacerbate kidney crystal formation .
Table 1: Summary of this compound Functions
| Function | Description |
|---|---|
| Amino Acid Metabolism | Coenzyme for transamination reactions |
| Neurotransmitter Synthesis | Precursor for serotonin, dopamine, GABA |
| Glycogenolysis | Facilitates conversion of glycogen to glucose |
| Immune Function | Supports immune response through metabolic pathways |
Table 2: Clinical Applications of this compound
| Condition | Application |
|---|---|
| Vitamin B6 Deficiency | Supplementation to restore normal levels |
| Nausea and Vomiting in Pregnancy | Combined with doxylamine (Diclectin) |
| Isoniazid-Induced Peripheral Neuropathy | Preventive supplementation |
Case Study 1: this compound Supplementation in Pregnant Women
A randomized controlled trial demonstrated that supplementation with this compound significantly reduced the incidence of nausea and vomiting during pregnancy compared to placebo groups. The study highlighted the safety and efficacy of this compound when used in conjunction with doxylamine.
Case Study 2: Toxicity in PH1 Patients
In a cohort study involving patients with primary hyperoxaluria type 1, high doses of this compound were found to correlate with increased kidney stone formation. This finding underscores the importance of monitoring this compound levels in susceptible populations.
Q & A
Q. Basic: What methodological approaches are recommended for quantifying pyridoxine in complex matrices like pharmaceuticals or biological samples?
Answer: Analytical quantification of this compound in multicomponent systems (e.g., overlapping UV-Vis spectra) requires robust regression models to resolve spectral interference. Studies recommend using Theil-Sen or RANSAC regression for outlier-resistant calibration curves, validated via Wilcoxon rank tests to confirm no significant differences between experimental and predicted values . For example, in caffeine-pyridoxine HCl mixtures, RANSAC outperformed Huber regression in handling large datasets with outliers . High-performance liquid chromatography (HPLC) with fluorescence detection is also validated for dose-response studies, ensuring accuracy through triplicate analysis and comparison with calculated dietary levels .
Q. Basic: How should researchers design dose-response experiments to determine this compound requirements in animal models?
Answer: Utilize a completely randomized design with one-way ANOVA to test linear and quadratic effects of this compound supplementation. For example, White Pekin duck studies defined the supplemental requirement as 95% of the level achieving maximum growth response, using quadratic regression analysis in SAS . Ensure basal diets are vitamin B6-deficient, and validate this compound concentrations via HPLC to align calculated and analyzed values .
Q. Advanced: How can researchers resolve overlapping spectral data in this compound analysis when using UV-Vis spectrophotometry?
Answer: Apply robust regression techniques (e.g., RANSAC) to mitigate outliers in full-spectrum UV-Vis data. Comparative studies show RANSAC and Theil-Sen regressions yield higher accuracy than Huber regression for caffeine-pyridoxine mixtures, with p-values >0.05 confirming no significant deviations . Pair these methods with cross-validation using test solutions to assess model performance .
Q. Advanced: How should conflicting pharmacokinetic or efficacy data for this compound be reconciled in systematic reviews?
Answer: Conduct integrated pharmacokinetic-pharmacodynamic (PK-PD) analyses to bridge clinical and preclinical data. For instance, CHMP guidelines recommend comparing bioavailability of this compound in combination products (e.g., doxylamine/pyridoxine FDC tablets) with mono-component studies using bioequivalence trials and literature meta-analyses . In clinical reviews, assess heterogeneity via sensitivity analysis and GRADE criteria, as seen in this compound trials for chemotherapy-induced hand-foot syndrome .
Q. Basic: What thermodynamic parameters are critical for studying this compound’s acid-base interactions in aqueous solutions?
Answer: Key parameters include dissociation constants (pKa) and enthalpy changes . Calorimetric studies at 298.15 K report pKa1 = 4.85 ± 0.02 and pKa2 = 8.96 ± 0.03, with equilibrium diagrams generated using computational tools like the "KEV" program . Background electrolyte concentration must be controlled to minimize ionic strength effects during calorimetry .
Q. Advanced: What experimental frameworks are used to investigate this compound-induced neuropathy in preclinical studies?
Answer: Design dose-duration response studies modeling inverse relationships between this compound intake (0.2–5 g/day) and symptom onset. Retrospective clinical analyses highlight pure sensory axonopathy as a hallmark, with improvement post-cessation . Use electrophysiological assays (e.g., nerve conduction studies) and histopathology to validate neurotoxic thresholds in animal models.
Q. Basic: How does this compound function in amino acid metabolism, and what assays validate its enzymatic role?
Answer: this compound 5′-phosphate (PLP), the active cofactor, mediates transamination and decarboxylation. Assess PLP-dependent enzymes (e.g., cystathionine β-synthase) via kinetic assays and HPLC-based substrate depletion monitoring. Clinical studies correlate PLP levels with homocysteine reduction, validated in vitamin B6-responsive homocystinuria .
Q. Advanced: What statistical methods address variability in this compound’s growth-promoting effects across studies?
Answer: Apply mixed-effects models to account for inter-study variability. For example, quadratic regression in SAS identifies non-linear responses in poultry growth, while ANOVA partitions variance between dietary treatments and individual variability . Meta-regression can further adjust for covariates like basal diet composition.
Q. Basic: How are this compound analytical methods validated per pharmacopeial standards?
Answer: Follow ICH Q2(R1) guidelines for specificity, linearity, and accuracy. Studies using this compound HCl reference standards (e.g., Sigma-Aldrich) validate HPLC methods with fluorescence detection, achieving ≤2% RSD in repeatability tests . Cross-validate with spectrophotometric methods for ternary mixtures (e.g., meclozine-pyridoxine-caffeine) .
Q. Advanced: What kinetic insights exist for this compound 5′-phosphate oxidase (PNPO) enzymes in microbial systems?
Answer: Characterize PNPOs (e.g., Mycobacterium tuberculosis Rv2607) via steady-state kinetics and X-ray crystallography. Studies show Rv2607’s substrate specificity for this compound 5′-phosphate, with Km values determined using spectrophotometric NADH-coupled assays . Bioinformatics tools (e.g., BLAST) identify conserved catalytic residues across microbial homologs .
Properties
IUPAC Name |
4,5-bis(hydroxymethyl)-2-methylpyridin-3-ol;hydrochloride | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C8H11NO3.ClH/c1-5-8(12)7(4-11)6(3-10)2-9-5;/h2,10-12H,3-4H2,1H3;1H | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
ZUFQODAHGAHPFQ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=NC=C(C(=C1O)CO)CO.Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C8H12ClNO3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
65-23-6 (Parent) | |
| Record name | Pyridoxine hydrochloride [USP:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000058560 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID1040792 | |
| Record name | Pyridoxine hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1040792 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
205.64 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Dry Powder, Colorless or white solid; [HSDB] White powder; [Sigma-Aldrich MSDS] | |
| Record name | 3,4-Pyridinedimethanol, 5-hydroxy-6-methyl-, hydrochloride (1:1) | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Pyridoxine hydrochloride | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/17042 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
Boiling Point |
Sublimes | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1212 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Solubility |
1 g dissolves in about 4.5 ml water, 90 ml alcohol; sol in propylene glycol; sparingly sol in acetone; insol in ether, chloroform, Soluble in alc, acetone; slightly soluble in other organic solvents., In water, 2.2X10+5 mg/l, temp not specified. | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1212 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Vapor Pressure |
0.00000006 [mmHg] | |
| Record name | Pyridoxine hydrochloride | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/17042 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
Mechanism of Action |
BRAIN GABA PRODN BEGAN TO RISE 7.5 MIN AFTER IP ADMIN OF 10 MG PYRIDOXINE HYDROCHLORIDE & CONTINUED FOR 40 MIN, THEN DECLINED., NUTRITIONAL NEED FOR VITAMIN B6 IS BASED ON ITS COENZYME FUNCTION IN AN UNUSUALLY LARGE NUMBER OF METABOLIC REACTIONS. ... REQUIRED IN METABOLISM OF AMINO ACIDS, FATS & CARBOHYDRATES & FOR SYNTHESIS OF PHYSIOLOGIC REGULATORS SUCH AS NOREPINEPHRINE, SEROTONIN & HISTAMINE., PYRIDOXAL PHOSPHATE IS INVOLVED IN SEVERAL METABOLIC TRANSFORMATIONS OF AMINO ACIDS INCLUDING DECARBOXYLATION, TRANSAMINATION, & RACEMIZATION, AS WELL AS ENZYMATIC STEPS IN METABOLISM OF SULFUR-CONTAINING & HYDROXY AMINO ACIDS. /PYRIDOXINE/, PYRIDOXINE ... IS AN ESSENTIAL PART OF THE ENZYME GLYCOGEN PHOSPHORYLASE. /PYRIDOXINE/ | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1212 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Platelets or thick, birefringent rods from alcohol + acetone, PLATES FROM ALCOHOL, ACETONE, COLORLESS OR WHITE CRYSTALS OR WHITE CRYSTALLINE POWDER, White to practically white crystals or crystalline powder., Colorless-white platelets | |
CAS No. |
58-56-0, 12001-77-3 | |
| Record name | Pyridoxine hydrochloride | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=58-56-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Pyridoxine hydrochloride [USP:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000058560 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Vitamin B6, hydrochloride | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0012001773 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=36225 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 3,4-Pyridinedimethanol, 5-hydroxy-6-methyl-, hydrochloride (1:1) | |
| Source | EPA Chemicals under the TSCA | |
| URL | https://www.epa.gov/chemicals-under-tsca | |
| Description | EPA Chemicals under the Toxic Substances Control Act (TSCA) collection contains information on chemicals and their regulations under TSCA, including non-confidential content from the TSCA Chemical Substance Inventory and Chemical Data Reporting. | |
| Record name | Pyridoxine hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1040792 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Pyridoxine hydrochloride | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.351 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/68Y4CF58BV | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1212 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Pyridoxine hydrochloride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0301760 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
207 °C | |
| Record name | PYRIDOXINE HYDROCHLORIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1212 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details











Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














