Levamisole
Overview
Description
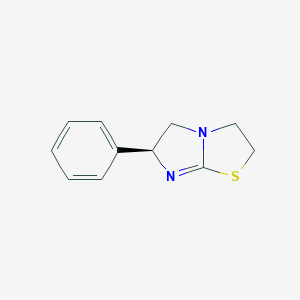
Levamisole, chemically known as (6S)-6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b]thiazole, is a chiral imidazothiazole derivative initially developed as a broad-spectrum anthelmintic agent . It is highly effective against gastrointestinal nematodes in livestock and humans, acting as a nicotinic acetylcholine receptor (nAChR) agonist that induces paralysis and subsequent expulsion of parasites . Beyond its antiparasitic use, this compound has been repurposed as an immunomodulator in conditions like rheumatoid arthritis and nephrotic syndrome due to its ability to enhance T-cell function . However, its misuse as a cutting agent in cocaine has raised significant public health concerns, as its metabolite aminorex exhibits psychostimulatory properties .
Structurally, this compound shares similarities with nicotine and other ganglionic stimulants, which explains its neurological side effects, including agitation, convulsions, and encephalopathy . Its molecular weight is 240.75 g·mol⁻¹, and it is commonly formulated as a hydrochloride or phosphate salt for improved solubility .
Preparation Methods
Synthetic Routes and Reaction Conditions
Levamisole can be synthesized through several methods. One common method involves the reaction of 2-imino-1,3-thiazolidine with 2-phenyl-2-hydroxyethylamine . This reaction is followed by cyclization to form the imidazothiazole ring structure. The reaction conditions typically involve the use of thionyl chloride and acetic anhydride .
Industrial Production Methods
Industrial production of this compound hydrochloride involves dissolving L-tetramisole in acetone and adding sodium selenite . The mixture is then decolorized and filtered using activated carbon. Dry hydrogen chloride gas is introduced while stirring until the pH value reaches 3-5. The reaction mixture is then separated and purified to obtain the final product .
Chemical Reactions Analysis
Types of Reactions
Levamisole undergoes various chemical reactions, including oxidation, reduction, and substitution .
Common Reagents and Conditions
Substitution: Substitution reactions often involve nucleophiles like halides or amines under basic conditions.
Major Products
The major products formed from these reactions depend on the specific reagents and conditions used. For example, oxidation of this compound can lead to the formation of sulfoxides or sulfones .
Scientific Research Applications
Medical Applications
1. Anthelmintic Use
Levamisole's primary application is as an anthelmintic drug, effective against intestinal roundworms (ascariasis) and hookworms (ancylostomiasis). It functions by acting as a nicotinic acetylcholine receptor agonist, leading to paralysis of the worms and subsequent expulsion from the host's body .
2. Immunomodulation
this compound has been recognized for its immunomodulatory effects, particularly in conditions characterized by immune dysfunction. It has been studied in the context of:
- Rheumatoid Arthritis : this compound was evaluated for its ability to improve immune responses in patients with rheumatoid arthritis, showing some efficacy in restoring T-cell function .
- Nephrotic Syndrome : Clinical trials have indicated that this compound may benefit children with nephrotic syndrome by enhancing immune response and reducing proteinuria .
3. Cancer Therapy
this compound has been investigated as an adjunct treatment in various cancers:
- Colon Cancer : It gained attention for its use in combination with fluorouracil (5-FU) as adjuvant therapy for stage III colon cancer. The combination has shown improved outcomes compared to 5-FU alone .
- Melanoma and Other Cancers : While trials in advanced breast cancer and lung cancer have yielded inconclusive results, some studies suggest potential benefits when used as part of a combination therapy .
Veterinary Applications
This compound is extensively used in veterinary medicine to treat parasitic infections in livestock and pets. Its applications include:
- Cattle and Sheep : It is commonly used to control gastrointestinal roundworms, lungworms, and other parasites .
- Aquaculture : this compound is employed by aquarists to treat specific parasitic infections in fish, such as Camallanus roundworm infestations .
Laboratory Applications
In research settings, this compound serves several functions:
- Enzyme Inhibition : It acts as a reversible inhibitor of alkaline phosphatase, which is useful in various biochemical assays such as Western blotting and in situ hybridization .
- Nematode Studies : this compound is utilized to immobilize nematodes like Caenorhabditis elegans for imaging or dissection purposes .
Table 1: Summary of this compound Applications
Case Studies
Case Study 1: this compound in Colon Cancer Treatment
A randomized clinical trial involving patients with Dukes' C stage C colon carcinoma demonstrated that the addition of this compound to standard chemotherapy (5-FU) improved disease-free survival rates compared to 5-FU alone. This study highlighted this compound's role as an effective adjuvant therapy despite its withdrawal from the market due to safety concerns related to impurities .
Case Study 2: Immunomodulatory Effects in Nephrotic Syndrome
In a cohort study of children with nephrotic syndrome, patients treated with this compound showed significant reductions in proteinuria and improvements in renal function markers. This study supports the hypothesis that this compound can modulate immune responses effectively in pediatric populations .
Mechanism of Action
Levamisole acts as a nicotinic acetylcholine receptor agonist, causing continuous stimulation of parasitic worm muscles, leading to paralysis and expulsion . In its immunomodulatory role, this compound stimulates the formation of antibodies, enhances T-cell responses, and potentiates monocyte and macrophage functions . It also increases neutrophil mobility, adherence, and chemotaxis .
Comparison with Similar Compounds
Comparison with Similar Compounds
Anthelmintic Agents
Nicotine and Ganglionic Stimulants
Levamisole’s mechanism of action and toxicity closely resemble nicotine. Both compounds induce vasopressor effects, muscle tremors, and hyperesthesia by stimulating nAChRs. However, this compound’s vasopressor activity is selectively blocked by α-adrenergic antagonists (e.g., phentolamine), unlike nicotine, which is inhibited by ganglionic blockers like hexamethonium .
| Property | This compound | Nicotine |
|---|---|---|
| Receptor Specificity | nAChR agonist | nAChR agonist |
| Vasopressor Blockade | α-adrenergic antagonists | Ganglionic blockers |
| Neurotoxicity | Encephalopathy, seizures | Convulsions, respiratory failure |
| Structural Features | Imidazothiazole core | Pyridine alkaloid |
Benzimidazoles (Albendazole, Mebendazole)
This compound is often combined with benzimidazoles for synergistic anthelmintic effects. In trichuriasis, albendazole + this compound achieves a 95.8% cure rate compared to 46.2% with albendazole alone, likely due to this compound’s neuromuscular action enhancing benzimidazole’s tubulin-disrupting effects .
| Combination Therapy | Cure Rate (Mild Trichuriasis) | Egg Reduction Rate |
|---|---|---|
| Albendazole alone | 46.2% | 88.3% |
| Albendazole + this compound | 95.8% | 96.7% |
| Mebendazole + this compound | 83.3% | 91.7% |
Immunomodulators and Chemotherapeutics
Steroid-Sparing Agents (Gold, D-Penicillamine)
In idiopathic nephrotic syndrome, this compound demonstrates comparable efficacy to gold and D-penicillamine but with a superior safety profile. It reduces relapse rates by 50–70% without nephrotoxicity or hepatotoxicity, unlike gold, which carries risks of proteinuria and cytopenia .
Suramin and Angiogenesis Inhibitors
N-alkylated this compound derivatives, such as N-methylthis compound, exhibit antiangiogenic activity distinct from suramin. While suramin inhibits vascular network formation via small cord morphology, N-methylthis compound induces cluster formation at lower concentrations (30 µM), suggesting a unique mechanism .
Alkaline Phosphatase Inhibitors
This compound and the experimental compound R8231 differentially inhibit alkaline phosphatases (ALPs). Mammalian bone ALP is 10–100× more sensitive to this compound than avian ALP, whereas R8231 shows potent inhibition across species. This selectivity makes R8231 a superior probe for studying ALP’s role in bone metabolism .
| Inhibitor | IC₅₀ (Mammalian ALP) | IC₅₀ (Avian ALP) |
|---|---|---|
| This compound | 10⁻⁶ M | 10⁻⁴ M |
| R8231 | 10⁻⁷ M | 10⁻⁶ M |
Structural and Functional Derivatives
Tetramisole
Tetramisole, the racemic mixture of this compound, has weaker anthelmintic activity. The (S)-enantiomer (this compound) is 10× more potent than the (R)-form, highlighting the importance of chirality in efficacy .
N-Alkylated Derivatives
N-methylthis compound and p-bromothis compound disrupt angiogenesis more effectively than the parent compound. N-methylthis compound achieves 97.7% intestinal region activation in Ascaris suum when combined with Cry5B, demonstrating synergistic anthelmintic effects .
Biological Activity
Levamisole, a synthetic derivative of imidazothiazole, has garnered attention for its diverse biological activities, primarily as an antihelminthic agent and immunomodulator. Initially developed for veterinary use, it has been repurposed in various medical contexts, including oncology and dermatology. This article delves into the biological activity of this compound, highlighting its mechanisms, clinical applications, and associated case studies.
This compound exerts its effects through multiple pathways:
- Immunomodulation : this compound enhances immune responses by stimulating T-lymphocytes and macrophages. It increases the production of interleukin-2 (IL-2), interleukin-12 (IL-12), and interferon-gamma (IFN-γ), which are crucial for T helper cell activation. It also inhibits immunosuppressive factors, thereby restoring immune function in depressed states .
- Antiparasitic Action : The drug acts as an agonist at nicotinic acetylcholine receptors in nematodes, leading to paralysis and death of the parasites. This mechanism is pivotal in its use as an antihelminthic .
- Antiangiogenic Properties : Recent studies have indicated that this compound and its derivatives can inhibit angiogenesis. For instance, N-methylthis compound demonstrated significant antiangiogenic activity in vitro, suggesting potential applications in cancer therapy .
Clinical Applications
This compound has been utilized in various clinical settings:
- Dermatology : It is employed to treat skin conditions such as cutaneous leishmaniasis and warts due to its immunomodulatory properties .
- Oncology : this compound has been used as an adjuvant therapy in colorectal cancer, enhancing the efficacy of fluorouracil by modulating immune responses .
- Cocaine Adulteration : In recent years, this compound has been identified as a common adulterant in cocaine. Its presence is associated with severe adverse effects, including vasculitis and nephrotoxicity .
Case Study 1: this compound-Induced Vasculitis
A study reported cases of purpuric skin eruptions linked to this compound exposure among illicit drug users. The patients exhibited leukocytoclastic vasculitis upon skin biopsy, highlighting this compound's potential to induce severe dermatological reactions when used illicitly .
Case Study 2: Cognitive Impairments in Cocaine Users
Research indicates that cocaine users exposed to high levels of this compound demonstrate significant cognitive impairments. MRI studies revealed reduced cortical thickness in areas associated with executive function, suggesting that this compound may exacerbate cognitive deficits linked to cocaine use .
Research Findings
Recent investigations into this compound's biological activities have yielded significant insights:
Q & A
Basic Research Questions
Q. What experimental models are most effective for studying Levamisole’s immunomodulatory mechanisms in preclinical research?
- Methodological Answer : Use in vitro assays (e.g., T-cell proliferation or cytokine profiling) combined with animal models (e.g., murine colitis or autoimmune disease models) to isolate this compound’s effects on immune cells. Validate findings via flow cytometry for lymphocyte subsets and ELISA for cytokine levels .
Q. How should researchers design dose-response studies to assess this compound’s efficacy in autoimmune or parasitic disease models?
- Methodological Answer : Employ a factorial design with varying doses (e.g., 2–10 mg/kg in rodents) and standardized outcome measures (e.g., parasite load reduction, histopathological scoring). Include positive controls (e.g., dexamethasone) and account for interspecies metabolic differences using allometric scaling .
Q. What are the key solubility parameters for this compound hydrochloride, and how do they influence formulation strategies?
- Methodological Answer : Determine solubility in polar solvents (e.g., water, DMSO) using gravimetric or UV-spectrophotometric methods at controlled temperatures (283–333 K). Apply thermodynamic models (e.g., NRTL or UNIQUAC) to predict solvent-drug interactions and optimize crystallization processes .
Advanced Research Questions
Q. How can researchers reconcile contradictory clinical data on this compound’s adjuvant efficacy in colorectal cancer?
- Methodological Answer : Perform a retrospective meta-analysis using Mantel-Haenszel methods to adjust for confounding variables (e.g., tumor staging, comorbidities). Stratify data by treatment duration (6 vs. 12 months) and analyze survival via Kaplan-Meier curves with log-rank tests .
Q. What statistical approaches are optimal for evaluating this compound’s interaction with other immunotherapies in combination trials?
- Methodological Answer : Use Cox proportional hazards models to assess time-to-event outcomes (e.g., recurrence-free survival). Incorporate interaction terms in regression models to quantify synergistic effects and adjust for multiplicity with Bonferroni corrections .
Q. How can thermodynamic properties inform this compound formulation stability under varying storage conditions?
- Methodological Answer : Conduct accelerated stability studies (40°C/75% RH) and correlate degradation kinetics with Hansen solubility parameters. Use differential scanning calorimetry (DSC) to detect polymorphic transitions and X-ray diffraction (PXRD) to confirm crystallinity .
Q. What ethical and practical considerations are critical when designing this compound trials in pediatric populations?
- Methodological Answer : Adhere to FINER criteria (Feasible, Interesting, Novel, Ethical, Relevant) for participant selection. Use age-adjusted dosing (e.g., 2.5 mg/kg) and monitor adverse events (e.g., leukopenia) via longitudinal CBC panels. Include placebo-controlled arms with DSMB oversight .
Q. Methodological Guidance
- Data Contradiction Analysis : Apply stratified subgroup analysis to isolate variables (e.g., smoking status in bronchogenic carcinoma trials) and report effect sizes with 95% confidence intervals .
- Experimental Reproducibility : Standardize immune assays using reference materials (e.g., WHO cytokine standards) and pre-register protocols on platforms like ClinicalTrials.gov .
- Thermodynamic Modeling : Validate solubility predictions with in silico tools (e.g., COSMO-RS) and cross-reference with experimental DSC/TGA data .
Properties
IUPAC Name |
(6S)-6-phenyl-2,3,5,6-tetrahydroimidazo[2,1-b][1,3]thiazole | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C11H12N2S/c1-2-4-9(5-3-1)10-8-13-6-7-14-11(13)12-10/h1-5,10H,6-8H2/t10-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HLFSDGLLUJUHTE-SNVBAGLBSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CSC2=NC(CN21)C3=CC=CC=C3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1CSC2=N[C@H](CN21)C3=CC=CC=C3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C11H12N2S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
16595-80-5 (hydrochloride) | |
| Record name | Levamisole [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0014769734 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID4023206 | |
| Record name | Levamisole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4023206 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
204.29 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Levamisole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014986 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
1.44e+00 g/L, Water 210 (mg/mL), Methanol sol. (mg/mL), Propylene glycol sol. (mg/mL), Ethanol sl. sol. (mg/mL) | |
| Record name | Levamisole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00848 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Levamisole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014986 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
| Record name | LEVAMISOLE | |
| Source | NCI Investigational Drugs | |
| URL | http://dtp.nci.nih.gov/NCI-InvestigationalDrugsCI92/177023%20(1992).txt | |
| Description | An investigational drug is one that is under study but does not have permission from the U.S. Food and Drug Administration (FDA) to be legally marketed and sold in the United States. NCI provides the investigational drug to the physicians who are participating in clinical trials or TRC protocols. For more information please visit NCI investigational drug website: https://www.cancer.gov/about-cancer/treatment/drugs/investigational-drug-access-fact-sheet | |
Mechanism of Action |
The mechanism of action of levamisole as an antiparasitic agent appears to be tied to its agnositic activity towards the L-subtype nicotinic acetylcholine receptors in nematode muscles. This agonistic action reduces the capacity of the males to control their reproductive muscles and limits their ability to copulate. The mechanism of action of Levamisole as an anticancer drug in combination with fluorouracil is unknown. The effects of levamisole on the immune system are complex. The drug appears to restore depressed immune function rather than to stimulate response to above-normal levels. Levamisole can stimulate formation of antibodies to various antigens, enhance T-cell responses by stimulating T-cell activation and proliferation, potentiate monocyte and macrophage functions including phagocytosis and chemotaxis, and increase neutrophil mobility, adherence, and chemotaxis. | |
| Record name | Levamisole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00848 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
14769-73-4 | |
| Record name | Levamisole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=14769-73-4 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Levamisole [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0014769734 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Levamisole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00848 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Levamisole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4023206 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Levamisole | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.035.290 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | LEVAMISOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/2880D3468G | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Levamisole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014986 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
227-227.5, 264 - 265 °C | |
| Record name | Levamisole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00848 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Levamisole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014986 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details





Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














