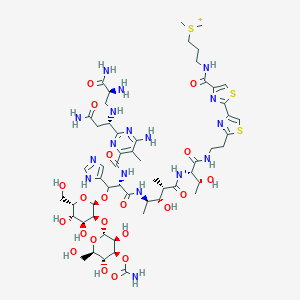
Bleomycin
Overview
Description
Bleomycin is a glycopeptide antibiotic complex produced by Streptomyces verticillus, primarily used as an antineoplastic agent. Its mechanism involves binding to DNA, inducing single- and double-strand breaks via oxidative damage, which inhibits cancer cell proliferation . Clinically, it is effective against squamous cell carcinomas, lymphomas, and germ cell tumors . A notable limitation is dose-dependent pulmonary toxicity, which restricts long-term use . This compound exists as a mixture of structurally related compounds (e.g., this compound A2, B2), with variations in terminal amine side chains influencing potency and toxicity .
Preparation Methods
Synthetic Routes and Reaction Conditions: Bleomycin is produced through the fermentation of Streptomyces verticillus. The fermentation process involves the cultivation of the bacterium in a nutrient-rich medium, followed by the extraction and purification of the compound . The production of this compound involves several steps, including the isolation of the active glycopeptide, its chemical modification, and the formation of the final product .
Industrial Production Methods: Industrial production of this compound typically involves large-scale fermentation processes. The bacterium Streptomyces verticillus is grown in bioreactors under controlled conditions to maximize the yield of this compound . The fermentation broth is then subjected to various purification steps, including filtration, chromatography, and crystallization, to obtain the pure compound .
Chemical Reactions Analysis
DNA Cleavage via Metal-Oxygen Complex Formation
Bleomycin forms an activated low-spin ferric-hydroperoxide complex (Fe<sup>III</sup>-OOH, ABLM) in the presence of Fe<sup>II</sup> and O<sub>2</sub>. This intermediate abstracts hydrogen atoms from the C4′ position of deoxyribose in DNA, leading to single- and double-strand breaks . Key steps include:
-
Activation : Fe<sup>II</sup>-BLM + O<sub>2</sub> → Fe<sup>III</sup>-OOH (ABLM) .
-
H Abstraction : ABLM extracts H from DNA’s C4′, generating a radical intermediate and Fe<sup>IV</sup>=O .
-
Strand Scission : The radical reacts with O<sub>2</sub>, forming a peroxyl radical that fragments DNA .
Kinetic and Thermodynamic Parameters of ABLM Reactions
Direct monitoring of ABLM decay and DNA interaction via circular dichroism revealed distinct kinetic profiles:
| Reaction Parameter | ABLM + DNA | ABLM Decay (No DNA) |
|---|---|---|
| Rate (s<sup>−1</sup>) | 0.044 ± 0.02 | 0.018 ± 0.003 |
| k<sub>H</sub>/k<sub>D</sub> | 1.7 ± 0.2 | 3.6 ± 0.9 |
| Activation Energy (kcal/mol) | 4.7 ± 0.9 | 9.3 ± 0.9 |
Data from .
The lower activation energy for DNA-linked reactions (4.7 vs. 9.3 kcal/mol) indicates preferential C–H bond cleavage over N–H abstraction in BLM’s degradation .
Comparative Reactivity with Enzymatic Systems
This compound’s catalytic profile aligns more closely with chloroperoxidase than cytochrome P-450 :
| Reaction Type | This compound | Chloroperoxidase | Cytochrome P-450 |
|---|---|---|---|
| Peroxidation (o-dianisidine) | Yes | Yes | No |
| N-Demethylation | Yes* | Yes | Yes |
| Aliphatic Hydroxylation | No | No | Yes |
| O<sub>2</sub> Evolution | Yes | Yes | No |
*Requires peroxides/iodosobenzene .
this compound catalyzes O<sub>2</sub> evolution from peroxyacids and chlorination via H<sub>2</sub>O<sub>2</sub>, mirroring chloroperoxidase .
Double-Strand DNA Damage Mechanism
This compound induces double-strand breaks (DSBs) at specific sites with high efficiency:
-
Target Sites : 5'-GC-3' and 5'-GT-3' sequences, with C4′ H atoms oriented toward the minor groove .
-
Efficiency : 43% of lesions at cytidine31 in hairpin DNA result in DSBs .
-
Structural Basis : Fe<sup>II</sup>-BLM binds DNA such that the metal center accesses opposing strand H atoms simultaneously .
Oxidative Reactions Beyond DNA Cleavage
This compound participates in diverse redox reactions:
-
N-Demethylation : Oxidizes N,N-dimethylaniline using H<sub>2</sub>O<sub>2</sub> or peroxyacids .
-
Mitochondrial Damage : Disrupts phosphatidylethanolamine synthesis, increasing ROS in fungi .
Role of Metal Ions in Reactivity
-
Iron Dependency : Fe<sup>II</sup> is essential for O<sub>2</sub> activation and ABLM formation .
-
Copper Binding : BLM-Cu complexes are inert but may stabilize the drug prior to Fe substitution .
This compound’s chemical reactivity hinges on its ability to harness Fe and O<sub>2</sub> for targeted DNA damage, with kinetic and structural studies elucidating its preference for H-atom abstraction over alternative mechanisms. Its dual capacity for single- and double-strand cleavage, coupled with oxidative versatility, underpins both therapeutic efficacy and toxicity .
Scientific Research Applications
Clinical Applications
Bleomycin has been widely utilized in the treatment of several malignancies. Its primary indications include:
- Hodgkin's Lymphoma: Often used in combination chemotherapy regimens such as ABVD (Adriamycin, this compound, Vinblastine, Dacarbazine).
- Testicular Cancer: Effective as part of combination therapy for germ cell tumors.
- Squamous Cell Carcinoma: Approved for use in head and neck cancers.
- Malignant Lymphoma: Utilized in treating both Hodgkin's and non-Hodgkin's lymphoma.
- Pleurodesis: Acts as a sclerosing agent to manage malignant pleural effusions by inducing adhesion of the lung to the chest wall .
Research Applications
In addition to its clinical uses, this compound serves as a critical tool in research, particularly in modeling pulmonary fibrosis:
Pulmonary Fibrosis Model
The administration of this compound to rodents is a standard model for studying idiopathic pulmonary fibrosis (IPF). This model helps researchers understand the disease's pathophysiology and evaluate potential antifibrotic therapies. Key findings from recent studies include:
- Interventional Timing: Studies indicate that timing interventions relative to this compound administration significantly affects outcomes. Early interventions (within 7 days) tend to focus on preventing fibrosis, while later interventions assess therapeutic efficacy .
- Study Characteristics: Between 2008 and 2019, approximately 74.4% of studies using this model investigated interventions aimed at fibrogenesis. A shift towards more therapeutic studies has been observed, reflecting a growing understanding of IPF and its treatment strategies .
Case Studies
Several notable case studies highlight the effectiveness and challenges associated with this compound:
- Case Study: Hodgkin's Lymphoma
- Case Study: Testicular Cancer
- Research Study: Pulmonary Fibrosis
Summary Table of this compound Applications
| Application Area | Specific Uses | Notes |
|---|---|---|
| Clinical Oncology | Hodgkin's lymphoma, testicular cancer | Often combined with other agents for effectiveness |
| Sclerotherapy | Malignant pleural effusions | Induces lung adhesion to prevent fluid accumulation |
| Research Models | Pulmonary fibrosis studies | Helps evaluate antifibrotic therapies |
| Mechanistic Studies | DNA damage response investigations | Provides insights into cancer cell biology |
Mechanism of Action
The primary mechanism of action of bleomycin involves its ability to bind to DNA and induce strand breaks . This compound forms a complex with metal ions, such as iron, creating a metallothis compound complex . This complex generates ROS, which cause oxidative damage to DNA, leading to single- and double-strand breaks . The DNA damage triggers cell cycle arrest and apoptosis, effectively killing cancer cells .
Comparison with Similar Compounds
Comparison with Structurally Similar Compounds
Pingyangmycin (Bleomycin A5)
- Structural Differences : Pingyangmycin, a component of the this compound complex, shares the core bleomycinic acid structure but differs in the terminal amine moiety .
- Efficacy: Demonstrates high activity against human colon, stomach, and nasopharyngeal cancer xenografts in nude mice .
- Toxicity : Lower pulmonary toxicity in murine models compared to standard this compound, enhancing its therapeutic index .
- Clinical Use : Widely used in China since 1979 for head and neck cancers .
Boanmycin (this compound A6, BAM)
- Structural Differences : Modified terminal amine group compared to this compound .
- Efficacy : Comparable antitumor activity to this compound, with specificity for epithelial-derived malignancies .
Table 1: Structural and Functional Comparison of this compound Derivatives
Comparison with Functionally Similar Compounds
Rapamycin
- Structural Differences: A macrolide immunosuppressant (vs. glycopeptide for this compound) .
- Clinical Use: Primarily used in immunosuppression and cancer therapy, unlike this compound’s direct DNA cleavage .
Latrunculin and Papuamide B
- Functional Similarity : Both marine sponge-derived compounds mimic this compound’s yeast deletion profile, suggesting overlapping cellular targets .
- Structural Divergence : Latrunculin (actin inhibitor) and Papuamide B (antiviral) differ chemically from this compound .
Table 2: Functional Comparison with Non-Structural Analogs
Key Research Findings
- Formulation Variability : Commercial this compound formulations exhibit compositional differences (e.g., A2/B2 ratios), impacting biological activity and regulatory compliance . For instance, one formulation exceeded pharmacopeial limits for this compound B2 (29.0% vs. 25% max), affecting efficacy .
- DNA Repair Assessment: this compound’s radiomimetic properties are utilized to measure DNA repair capacity in cancer patients. Tumor patients showed 79.4% repair efficiency vs. 92.4% in controls, highlighting its diagnostic utility .
- Clinical Efficacy in Vascular Tumors : Intralesional this compound achieved 85% cure rates in pediatric facial hemangiomas <2 cm², with minimal adverse effects .
Biological Activity
Bleomycin is a glycopeptide antibiotic derived from Streptomyces verticillus, primarily known for its use in cancer chemotherapy. Its biological activity encompasses a range of mechanisms that affect cellular processes, particularly in cancerous cells, but also extends to other biological systems, including antifungal activity and effects on telomerase activity.
This compound exerts its biological effects through several mechanisms:
- DNA Damage : this compound induces DNA strand breaks through oxidative stress, leading to the formation of free radicals. This action is particularly potent during the G2 phase of the cell cycle, inhibiting cell division and contributing to its antitumor effects .
- Telomerase Activity Modulation : Research indicates that this compound can modify telomerase activity in lung epithelial cells. Initially, this compound treatment results in an increase in telomerase activity, which may protect against apoptosis. However, prolonged exposure leads to a significant reduction in telomerase activity, correlating with increased apoptosis and potential lung fibrosis .
- Antifungal Properties : Recent studies have identified this compound as having significant antifungal activity. It disrupts mitochondrial function in fungal cells, leading to increased reactive oxygen species (ROS) generation and impaired phospholipid biosynthesis, essential for cell viability .
Clinical Applications
This compound is utilized not only in oncology but also in treating various benign conditions:
- Cancer Treatment : It is commonly used in combination chemotherapy regimens for testicular cancer and Hodgkin's lymphoma. The BEP regimen (this compound, Etoposide, and Cisplatin) is particularly effective for germ cell tumors .
- Treatment of Lymphangiomas : A study demonstrated that this compound injections resulted in an excellent response in 55% of pediatric patients with lymphangiomas, showcasing its effectiveness beyond malignancies .
- Wart Treatment : Intralesional this compound has been evaluated for treating common warts, showing a complete cure rate of approximately 84% after treatment sessions .
Case Study 1: this compound-Induced Lung Toxicity
A patient diagnosed with extragonadal non-seminomatous germ cell tumor underwent four cycles of BEP chemotherapy. Post-treatment imaging revealed pulmonary involvement attributed to this compound toxicity, highlighting the need for careful monitoring of pulmonary function during therapy .
Case Study 2: Efficacy in Lymphangiomas
In a cohort of 20 children treated with this compound for lymphangiomas, 55% exhibited an excellent response while 95% showed good response rates. This study underscores the efficacy of this compound in non-cancerous conditions and supports its continued use in clinical practice .
Telomerase Activity
A detailed investigation into the effects of this compound on telomerase activity revealed:
- In Vitro Studies : A significant elevation in mTERT mRNA and a transient increase in telomerase activity were observed within 24 hours post-treatment. However, by 72 hours, telomerase activity decreased significantly below baseline levels .
| Time Point | Telomerase Activity Change (%) |
|---|---|
| 24 hours | +41% |
| 48 hours | +12% |
| 72 hours | -26% |
Antifungal Mechanism
The antifungal mechanism of this compound was elucidated through studies showing its impact on mitochondrial integrity and phospholipid synthesis in yeast models. The compound's ability to induce oxidative stress was linked to its antifungal efficacy .
Q & A
Basic Research Questions
Q. What are the established molecular mechanisms of bleomycin-induced DNA cleavage, and how can researchers validate these mechanisms in vitro?
- Methodological Answer : this compound cleaves DNA via metal-dependent oxidative damage, primarily targeting guanine residues. To validate this, researchers can:
- Use plasmid relaxation assays with supercoiled DNA (e.g., pBR322) to visualize single- and double-strand breaks via gel electrophoresis .
- Employ electron paramagnetic resonance (EPR) spectroscopy to detect oxygen radical formation in the presence of Fe²⁺/bleomycin complexes .
- Compare cleavage patterns in oxygen-rich vs. hypoxic conditions to confirm oxygen dependency .
Q. What experimental models are commonly used to study this compound-induced pulmonary fibrosis, and what endpoints are critical for reproducibility?
- Methodological Answer : The C57BL/6 mouse model is standard for pulmonary fibrosis studies. Key endpoints include:
- Histopathology : H&E and Masson’s trichrome staining to quantify collagen deposition .
- Hydroxyproline assays : Quantify total collagen content in lung tissue lysates (e.g., using a colorimetric kit) .
- Molecular markers : qRT-PCR for fibrotic genes (e.g., α-SMA, Col1a1, Col3a1) with GAPDH normalization .
- Note: Maintain consistent this compound dosing (e.g., 1–2 U/kg intratracheally) and monitor strain-specific susceptibility .
Q. How should researchers address variability in this compound cytotoxicity assays across cell lines?
- Methodological Answer : Variability arises from differences in cell membrane permeability to this compound and endogenous antioxidant levels. Mitigate this by:
- Using this compound-hypersensitive cell lines (e.g., GM00847 fibroblasts) for consistent DNA damage readouts .
- Pre-treating cells with catalase inhibitors to amplify oxidative stress signals .
- Normalizing cytotoxicity data to cell viability controls (e.g., MTT assays) run in parallel .
Advanced Research Questions
Q. How can researchers resolve contradictions in this compound’s dose-response relationship between in vitro and in vivo studies?
- Methodological Answer : Discrepancies often stem from pharmacokinetic differences (e.g., rapid plasma clearance in vivo vs. sustained exposure in vitro). Strategies include:
- Pharmacokinetic modeling : Use LC-MS/MS to measure this compound-A2 and -B2 distribution in murine plasma and tissues .
- Tissue-specific delivery : Employ intratracheal or osmotic pump administration to mimic sustained in vitro exposure .
- Data normalization : Express in vivo results as tissue-specific AUC (area under the curve) rather than total dose .
Q. What experimental designs are optimal for studying this compound’s synergism with other anticancer agents (e.g., cisplatin) while minimizing pulmonary toxicity?
- Methodological Answer : Use factorial design to isolate interaction effects:
- Dose matrix : Test this compound (0.1–10 μM) with cisplatin (1–100 μM) in A549 lung cancer cells .
- Endpoint selection : Measure synergy via Chou-Talalay combination index (CI) and parallel RNA-seq to identify overlapping toxicity pathways (e.g., oxidative stress genes) .
- In vivo validation : Administer combinations to C57BL/6 mice with serial CT imaging to monitor fibrosis progression .
Q. How can researchers optimize this compound dosing in preclinical studies to balance antitumor efficacy and fibrosis risk?
- Methodological Answer :
- Toxicodynamic modeling : Fit dose-response curves to Hill equations, separating efficacy (tumor regression) and toxicity (hydroxyproline levels) parameters .
- Biomarker monitoring : Track plasma TGF-β1 and KL-6 levels as early fibrosis indicators .
- Alternative schedules : Compare bolus vs. fractionated dosing in xenograft models to identify regimens with lower cumulative lung damage .
Q. Methodological Challenges and Solutions
Q. What techniques validate this compound’s purity and stability in experimental formulations?
- Methodological Answer :
- HPLC-UV : Use a C18 column with 0.1% TFA/acetonitrile gradient to separate this compound-A2 (retention time ~12 min) from degradation products .
- Mass spectrometry : Confirm molecular ions (m/z 1,415 for this compound-A2) and monitor deamidation (common degradation pathway) .
- Bioactivity assays : Compare DNA cleavage activity of stored vs. fresh samples to detect functional loss .
Q. How should conflicting data on this compound’s role in senescence-associated secretory phenotype (SASP) be analyzed?
- Methodological Answer : Contradictions may arise from cell type-specific SASP responses. Resolve by:
- Single-cell RNA-seq : Cluster senescent cells by SASP gene expression (e.g., IL-6, CXCL1) in this compound-treated primary fibroblasts .
- Conditioned media transfers : Test paracrine effects of SASP factors on non-senescent cells using transwell assays .
- Pharmacologic inhibition : Use JAK/STAT or NF-κB inhibitors to dissect signaling pathways driving SASP heterogeneity .
Q. Emerging Research Directions
Q. What novel biomarkers can predict interindividual variability in this compound-induced lung toxicity?
- Methodological Answer :
- GWAS studies : Screen for SNPs in this compound hydrolase (BLMH) and xeroderma pigmentosum genes (e.g., XPF) linked to toxicity .
- Proteomics : Identify predictive serum proteins (e.g., MMP-7, periostin) via Olink or SOMAmer platforms .
- Imaging biomarkers : Quantify early fibrosis via hyperpolarized ¹²⁹Xe MRI gas-exchange metrics .
Q. How can CRISPR/Cas9 models elucidate genetic modifiers of this compound resistance in tumors?
- Methodological Answer :
- Genome-wide screens : Perform pooled CRISPR knockout screens in this compound-treated cancer cells; prioritize hits with MAGeCK or BAGEL algorithms .
- Validation : Knock out candidate genes (e.g., RAD51, BRCA1) and measure IC₅₀ shifts and γ-H2AX foci (DNA damage markers) .
- Clinical correlation : Cross-reference hits with TCGA data to assess prognostic significance .
Properties
IUPAC Name |
3-[[2-[2-[2-[[(2S,3R)-2-[[(2S,3S,4R)-4-[[(2S,3R)-2-[[6-amino-2-[(1S)-3-amino-1-[[(2S)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[3-[4-carbamoyloxy-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-3-(1H-imidazol-5-yl)propanoyl]amino]-3-hydroxy-2-methylpentanoyl]amino]-3-hydroxybutanoyl]amino]ethyl]-1,3-thiazol-4-yl]-1,3-thiazole-4-carbonyl]amino]propyl-dimethylsulfanium | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C55H83N17O21S3/c1-20-33(69-46(72-44(20)58)25(12-31(57)76)64-13-24(56)45(59)82)50(86)71-35(41(26-14-61-19-65-26)91-54-43(39(80)37(78)29(15-73)90-54)92-53-40(81)42(93-55(60)88)38(79)30(16-74)89-53)51(87)66-22(3)36(77)21(2)47(83)70-34(23(4)75)49(85)63-10-8-32-67-28(18-94-32)52-68-27(17-95-52)48(84)62-9-7-11-96(5)6/h14,17-19,21-25,29-30,34-43,53-54,64,73-75,77-81H,7-13,15-16,56H2,1-6H3,(H13-,57,58,59,60,61,62,63,65,66,69,70,71,72,76,82,83,84,85,86,87,88)/p+1/t21-,22+,23+,24-,25-,29?,30?,34-,35-,36-,37?,38?,39?,40?,41-,42?,43?,53?,54?/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
OYVAGSVQBOHSSS-WXFSZRTFSA-O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C(N=C(N=C1N)C(CC(=O)N)NCC(C(=O)N)N)C(=O)NC(C(C2=CN=CN2)OC3C(C(C(C(O3)CO)O)O)OC4C(C(C(C(O4)CO)O)OC(=O)N)O)C(=O)NC(C)C(C(C)C(=O)NC(C(C)O)C(=O)NCCC5=NC(=CS5)C6=NC(=CS6)C(=O)NCCC[S+](C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC1=C(N=C(N=C1N)[C@H](CC(=O)N)NC[C@@H](C(=O)N)N)C(=O)N[C@@H]([C@H](C2=CN=CN2)OC3C(C(C(C(O3)CO)O)O)OC4C(C(C(C(O4)CO)O)OC(=O)N)O)C(=O)N[C@H](C)[C@H]([C@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCCC5=NC(=CS5)C6=NC(=CS6)C(=O)NCCC[S+](C)C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C55H84N17O21S3+ | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
9041-93-4 (sulfate (salt)) | |
| Record name | Bleomycin [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0011056067 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
Molecular Weight |
1415.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Bleomycin appears as colorless or yellowish powder. Possible bluish color depending on copper content. (NTP, 1992), Solid | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Soluble (NTP, 1992), Soluble, Colorless or yellowish powder which becomes bluish depending on copper content. Very sol in water, methanol; practically insol in acetone, ethyl acetate, butyl acetate, ether; slightly sol in ethanol. /Bleomycins/, HIGHLY SOL IN WATER & METHANOL; SPARINGLY SOL IN ALC; INSOL IN ACETONE & ETHYL ACETATE; CREAM-COLORED POWDER OR SOFT, FLUFFY LUMPS. /SULFATE SALT/, Freely soluble in water., Sol in water and methanol but insol in acetone and ether., 2.82e-02 g/L | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Although the exact mechanism of action of bleomycin is unknown, available evidence would seem to indicate that the main mode of action is the inhibition of DNA synthesis with some evidence of lesser inhibition of RNA and protein synthesis. As evident in _in vitro_ studies, the DNA-cleaving actions of bleomycin is dependent on oxygen and metal ions. It is believed that bleomycin chelates metal ions (primarily iron) producing a pseudoenzyme that reacts with oxygen to produce superoxide and hydroxide free radicals that cleave DNA., Cytotoxic action of bleomycins results from their ability to cause fragmentation of DNA. Studies in vitro indicate that bleomycin causes accumulation of cells in the G2 phase of the cell cycle, and many of these cells display chromosomal aberrations, incl chromatid breaks, gaps, and fragments, as well as translocations. Bleomycin appears to cause scission of DNA by interacting with oxygen and iron(2+). In the presence of oxygen and a reducing agent, such as dithiothreitol, the metallobleomycin complex becomes activated and functions mechanistically as a ferrous oxidase, transferring electrons from iron(2) to molecular oxygen to produce activated species of oxygen. It has also been shown that metallobleomycin complexes can be activated by reaction with the flavin enzyme, NADPH-cytochrome p450 reductase. Bleomycin binds to DNA through its amino terminal peptide, and the activated complex generates free radicals that are responsible for scission of the DNA chain., Bleomycin is an antineoplastic antibiotic. The drug is active against gram-positive and gram-negative bacteria and fungi, but its cytotoxicity precludes its use as an anti-infective agent. The precise mechanism(s) of action of bleomycin is not fully known. Several studies in Escherichia coli and HeLa cells suggest that the drug inhibits the incorporation of thymidine into DNA. In these in vitro studies, DNA synthesis was inhibited to a greater extent than was RNA or protein synthesis. Bleomycin also appears to labilize the DNA structure, resulting in scission of both single- and double-stranded DNA. The drug has no immunosuppressive activity in mice., Bleomycin is classed as an antibiotic but is not used as an antimicrobial agent. Although bleomycin is effective against both cycling and non-cycling cells, it seems to be most effective in the G2 phase of cell division. Its exact mechanism of antineoplastic action is unknown but may involve binding to DNA, inducing lability of the DNA structure, and reduced synthesis of DNA, and to a lesser extent RNA and ptoteins., When administered into the pleural cavity in the treatment of malignant pleural effusion, /bleomycin/ acts as a sclerosing agent. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Colorless to yellow powder | |
CAS No. |
11056-06-7 | |
| Record name | BLEOMYCIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/19884 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Bleomycin [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0011056067 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BLEOMYCIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
71 °C | |
| Record name | Bleomycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00290 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Bleomycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014435 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















