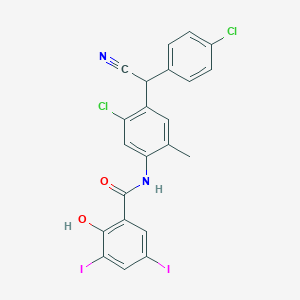
Closantel
Overview
Description
Closantel is a synthetic antiparasitic agent belonging to the chemical class of salicylanilides. It is primarily used in veterinary medicine to control internal parasites such as roundworms and liver flukes, as well as some external parasites like sheep nasal bots and cattle grubs . This compound is not used against agricultural or household pests and is not intended for human use .
Preparation Methods
Closantel can be synthesized through various methods. One method involves the preparation of this compound sodium intermediate by catalytic hydrogenation . Another method includes the preparation of this compound sodium injection, which involves mixing ethanol and propylene glycol, heating the mixture, adding sodium this compound, and adjusting the pH with sodium hydroxide solution . These methods are efficient and suitable for industrial production.
Chemical Reactions Analysis
Closantel undergoes several types of chemical reactions, including oxidation, reduction, and substitution. For example, the enantiomeric separation of this compound can be achieved using high-performance liquid chromatography (HPLC) with different chiral stationary phases and mobile phase compositions . The major products formed from these reactions are the enantiomers of this compound, which can be separated and analyzed for further research .
Scientific Research Applications
Antiparasitic Applications
Closantel is widely recognized for its effectiveness against various parasitic infections in ruminants, particularly against blood-feeding helminths such as Fasciola hepatica and Haemonchus contortus. It operates by inhibiting the metabolism of these parasites, leading to their death.
Efficacy Against Specific Parasites
- Fasciola spp. : this compound has shown a 100% efficacy rate against Fasciola spp. in clinical settings, significantly reducing fecal egg counts within a week post-treatment .
- Haemonchus spp. : In studies comparing various anthelmintics, this compound demonstrated superior effectiveness against Haemonchus spp., outperforming other treatments like levamisole and fenbendazole .
Pharmacokinetics and Bioavailability
The pharmacokinetic profile of this compound has been extensively studied to optimize its use in livestock. Key findings include:
- High Plasma Protein Binding : this compound exhibits a high degree of plasma protein binding (approximately 99%), contributing to its prolonged action in the body .
- Bioequivalence Studies : Research has confirmed that different formulations of this compound show similar pharmacokinetic parameters, ensuring consistent therapeutic outcomes across formulations .
Table 1: Pharmacokinetic Parameters of this compound
| Parameter | Value |
|---|---|
| Cmax | Varies by formulation |
| Tmax | 24-48 hours |
| Half-life | 5-7 days |
| Plasma Protein Binding | ~99% |
Clinical Studies and Milk Production
A randomized controlled trial evaluated the impact of this compound on milk production in dairy cattle. The study found that administering this compound improved milk yield and reduced antibody levels against Fasciola hepatica in milk samples .
Study Design Overview
- Population : First-calf heifers.
- Dosage : 0.2 ml/kg body weight.
- Duration : Administered at dry-off, between 80 and 42 days before calving.
Novel Applications in Oncology
Recent research indicates potential applications of this compound beyond veterinary medicine. It has been identified as a BRAFV600E enzyme inhibitor, suggesting its potential use in preparing drugs for treating certain tumors, including melanoma and colorectal cancer .
Conclusion and Future Directions
This compound remains a vital compound in veterinary medicine with proven efficacy against various parasitic infections in livestock. Its pharmacokinetic properties support its long-term use, while emerging research suggests novel applications in oncology. Future studies should focus on expanding its therapeutic uses and understanding its mechanisms further.
Mechanism of Action
Closantel exerts its antiparasitic effects by uncoupling oxidative phosphorylation in the mitochondria of parasites, which disrupts the production of adenosine triphosphate (ATP), the cellular "fuel" . This mechanism leads to the accumulation of fumarate and ultimately results in the parasite’s demise . This compound also inhibits fumarate reductase, an essential enzyme involved in fumarate synthesis .
Comparison with Similar Compounds
Closantel is unique among antiparasitic agents due to its specific mechanism of action and broad-spectrum activity against various parasites. Similar compounds include other salicylanilide derivatives such as oxyclozanide and rafoxanide . These compounds share similar chemical structures and mechanisms of action but may differ in their spectrum of activity and efficacy against specific parasites .
Biological Activity
Closantel is an anthelmintic compound primarily used in veterinary medicine for the treatment of parasitic infections, particularly those caused by trematodes and nematodes. Recent studies have expanded its application to include antibacterial properties, showcasing its potential in combating multidrug-resistant bacterial infections. This article explores the biological activity of this compound, focusing on its antimicrobial effects, toxicity, and pharmacokinetics.
Antimicrobial Activity
This compound has demonstrated significant antibacterial activity, particularly against gram-negative bacteria. A study highlighted the synergistic effects of this compound when combined with polymyxin B against Acinetobacter baumannii, a notorious multidrug-resistant pathogen. The combination therapy was effective in inhibiting the development of resistance to polymyxin B, suggesting that this compound can serve as a valuable adjuvant in treating severe bacterial infections.
Key Findings:
- Minimum Inhibitory Concentration (MIC) : this compound alone showed MIC values greater than 128 mg/L against most A. baumannii isolates but achieved lower MICs (0.5 mg/L) against certain polymyxin-resistant strains when used in combination with polymyxin B .
- Synergistic Effects : The combination of polymyxin B (2 mg/L) with this compound (4-16 mg/L) effectively inhibited bacterial growth and prevented the emergence of resistance .
Case Studies on Toxicity
Despite its therapeutic benefits, this compound has been associated with adverse effects, particularly ocular toxicity. A notable case involved a 20-year-old female patient who experienced reversible blindness after receiving an incorrect prescription of this compound instead of triclabendazole for Fasciola hepatica infection. The patient's vision impairment was linked to retinal damage caused by the drug.
Case Highlights:
- Symptoms : The patient reported bilateral blurred vision and color blindness after taking this compound for three days.
- Treatment : Plasma exchange and high-dose corticosteroids were administered, resulting in partial recovery of vision over a follow-up period .
Pharmacokinetics
This compound exhibits a long half-life and high plasma protein binding capacity, which influences its distribution and elimination from the body. Studies indicate that after oral administration, this compound's half-life ranges from 22.7 to 26.7 days, with approximately 80% of the administered dose excreted in feces within eight weeks .
Pharmacokinetic Data:
| Administration Route | Half-Life (Days) | Excretion (Feces) | Excretion (Urine) |
|---|---|---|---|
| Oral | 26.7 | ~80% | ~0.5% |
| Intramuscular | 22.7 | ~80% | ~0.5% |
Q & A
Basic Research Questions
Q. What are the primary molecular mechanisms underlying Closantel’s anthelmintic activity, and how can these be experimentally validated?
this compound acts as a proton ionophore, disrupting mitochondrial membrane potential in parasites by binding to the H+/K+-ATPase transporter . To validate this, researchers can:
- Use in vitro assays (e.g., fluorescence-based mitochondrial membrane potential assays in Haemonchus contortus L3 larvae).
- Compare dose-response curves between wild-type and ATPase-knockout parasite strains.
- Employ molecular docking simulations to confirm binding affinity to target proteins. Table 1 : Key assays for mechanistic studies
| Assay Type | Target | Outcome Metric | Reference |
|---|---|---|---|
| Fluorescence polarization | H+/K+-ATPase | Binding affinity (IC₅₀) | |
| Resazurin reduction assay | Parasite viability | EC₅₀ values |
Q. How should researchers select appropriate in vivo models to evaluate this compound’s efficacy against resistant nematode strains?
- Prioritize naturally resistant isolates (e.g., Teladorsagia circumcincta from field surveys) over lab-induced strains to reflect real-world resistance patterns.
- Use FECRT (Fecal Egg Count Reduction Test) with controlled dosing in sheep or cattle, ensuring sample sizes ≥10 animals/group to account for host variability .
- Include pharmacokinetic monitoring to correlate plasma concentrations with efficacy.
Q. What standardized protocols exist for quantifying this compound residues in animal tissues, and how can cross-lab reproducibility be ensured?
- Follow HPLC-MS/MS protocols validated by the Codex Alimentarius Commission (CAC/GL 71-2009). Key steps:
Homogenize tissue samples in acetonitrile:water (80:20).
Use deuterated this compound as an internal standard.
Validate recovery rates (≥85%) across triplicate runs .
- Share raw chromatograms and calibration curves in supplementary materials to aid reproducibility .
Advanced Research Questions
Q. How can contradictory data on this compound’s efficacy in different host species (e.g., cattle vs. sheep) be systematically analyzed?
- Conduct a meta-analysis of existing studies, stratifying by:
-
Host pharmacokinetics (e.g., plasma half-life differences: 14 days in cattle vs. 10 days in sheep).
-
Parasite burden metrics (eggs per gram vs. adult worm counts).
- Use mixed-effects models to account for variability in dosing regimens and resistance profiles .
Table 2 : Species-specific PK parameters of this compound
Species Half-life (days) Cₘₐₓ (μg/mL) Reference Cattle 14.2 32.5 Sheep 9.8 28.1 - Use mixed-effects models to account for variability in dosing regimens and resistance profiles .
Q. What experimental strategies can resolve conflicting hypotheses about this compound’s role in inducing oxidative stress in parasites?
- Combine transcriptomic profiling (RNA-seq) of this compound-exposed parasites with ROS (reactive oxygen species) detection assays (e.g., DCFH-DA fluorescence).
- If ROS levels increase but antioxidant genes (e.g., sod-1, gst) are downregulated, this supports mitochondrial dysfunction as the primary mechanism over oxidative stress .
- Validate using gene knockout strains (e.g., sod-1 mutants) to isolate contributing pathways.
Q. How can researchers design studies to investigate this compound’s potential synergism with benzimidazoles while avoiding confounding variables?
- Use fractional inhibitory concentration (FIC) indices in in vitro larval development assays.
- Control for:
- Metabolism interactions (e.g., CYP450 enzyme inhibition by this compound).
- Differential absorption rates via co-administration with pharmacokinetic enhancers (e.g., cyclodextrins) .
- Apply response surface methodology (RSM) to model dose-response surfaces and identify optimal ratios.
Q. Methodological and Ethical Considerations
Q. What statistical approaches are recommended for addressing high inter-individual variability in this compound pharmacokinetic studies?
- Use non-linear mixed-effects modeling (NONMEM) to partition variability into host (e.g., weight, age) and environmental (e.g., feed composition) factors .
- Report coefficients of variation (CV%) for key parameters (AUC, Cₘₐₓ) with 90% confidence intervals.
Q. How should researchers mitigate bias when interpreting this compound resistance data from field trials?
- Blind laboratory analysts to treatment groups during fecal egg counts.
- Include negative controls (untreated animals) and positive controls (known susceptible strains) in each assay batch .
- Disclose funding sources and conflicts of interest in compliance with ICMJE guidelines .
Q. Emerging Research Frontiers
Q. What methodologies are suited for investigating this compound’s off-target effects on host gut microbiota?
- Perform 16S rRNA sequencing on fecal samples pre- and post-treatment, using Qiime2 for differential abundance analysis.
- Correlative analysis: Link microbiota shifts (e.g., Bacteroidetes/Firmicutes ratio) with host metabolic markers (e.g., serum SCFA levels) .
Q. How can machine learning improve predictive models of this compound resistance evolution?
- Train random forest classifiers on genomic data (SNPs in Hco-acr-8 gene) and treatment histories from surveillance databases.
- Validate models using spatial cross-validation to account for regional resistance patterns .
Properties
IUPAC Name |
N-[5-chloro-4-[(4-chlorophenyl)-cyanomethyl]-2-methylphenyl]-2-hydroxy-3,5-diiodobenzamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H14Cl2I2N2O2/c1-11-6-15(17(10-27)12-2-4-13(23)5-3-12)18(24)9-20(11)28-22(30)16-7-14(25)8-19(26)21(16)29/h2-9,17,29H,1H3,(H,28,30) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
JMPFSEBWVLAJKM-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=CC(=C(C=C1NC(=O)C2=C(C(=CC(=C2)I)I)O)Cl)C(C#N)C3=CC=C(C=C3)Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H14Cl2I2N2O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID6040662 | |
| Record name | Closantel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6040662 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
663.1 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
57808-65-8 | |
| Record name | Closantel | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=57808-65-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Closantel [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0057808658 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Closantel | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759819 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Closantel | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=335306 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Closantel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6040662 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Closantel | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.055.407 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CLOSANTEL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/EUL532EI54 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















