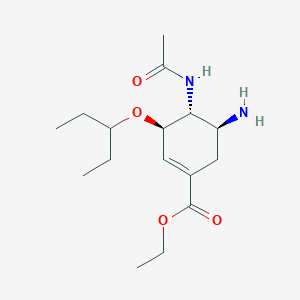
Oseltamivir
Overview
Description
Oseltamivir (marketed as Tamiflu®) is a competitive neuraminidase (NA) inhibitor widely used for the treatment and prophylaxis of influenza A and B infections. It functions by blocking the enzymatic activity of viral NA, preventing the release of progeny virions from infected cells . Structurally, this compound is a prodrug metabolized to its active form, this compound carboxylate, which mimics the transition state of sialic acid, enabling high-affinity binding to NA . Pharmacokinetic studies demonstrate consistent bioavailability across ethnic groups, with similar plasma concentrations observed in Caucasian and Japanese populations . Its chemical stability in different media, such as reduced C=O bond force constants in aqueous solutions, further supports its therapeutic utility .
Preparation Methods
Synthetic Routes and Reaction Conditions
The synthesis of oseltamivir is complex due to its three contiguous chiral centers. One of the initial scalable routes to this compound started from shikimic acid, a natural product . The process involves multiple steps, including protection and deprotection of functional groups, formation of the cyclohexene ring, and introduction of the amino and acetamido groups .
Industrial Production Methods
Industrial production of this compound often involves biocatalytic methods due to their mild, highly selective, and environmentally benign nature . For example, the use of enzymes in the asymmetric synthesis of amino acid components and the fermentative production of structurally complex intermediates are common . Another method involves the use of organic solvents and catalysts like Lindlar catalyst under specific conditions to achieve high yields .
Chemical Reactions Analysis
Types of Reactions
Oseltamivir undergoes various chemical reactions, including:
Oxidation: Conversion of the hydroxyl group to a carbonyl group.
Reduction: Reduction of the azido group to an amino group.
Substitution: Introduction of functional groups like acetamido and amino groups.
Common Reagents and Conditions
Common reagents used in these reactions include hydrogen gas, Lindlar catalyst, and organic solvents like methanol and acetone . Reaction conditions often involve specific temperatures and pressures to ensure high yields and purity .
Major Products
The major products formed from these reactions include this compound carboxylate, which is the active metabolite of this compound .
Scientific Research Applications
Oseltamivir has a wide range of scientific research applications:
Chemistry: Used as a model compound in the study of chiral synthesis and biocatalysis.
Biology: Studied for its interactions with viral proteins and its role in inhibiting viral replication.
Medicine: Widely used in the treatment and prevention of influenza.
Industry: Employed in the large-scale production of antiviral medications.
Mechanism of Action
Oseltamivir exerts its antiviral activity by inhibiting the activity of the viral neuraminidase enzyme found on the surface of the virus . This inhibition prevents the release of new viral particles from infected cells, thereby reducing the spread of the virus within the body . The drug is a prodrug, which means it is converted into its active form, this compound carboxylate, by hepatic esterases .
Comparison with Similar Compounds
Comparison with Similar Compounds
Zanamivir
Zanamivir, the first NA inhibitor, shares a similar mechanism but differs in administration (inhaled vs. oral) and pharmacokinetics. While both target the NA active site, zanamivir exhibits lower oral bioavailability (3.95% intestinal absorption) compared to oseltamivir (79.33%) . Structural analyses reveal that zanamivir and this compound carboxylate bind to the sialic acid cavity of H5N2 NA with comparable hydrogen bond interactions and binding energies (Table 1) . However, zanamivir’s polar structure limits its use in patients with respiratory complications.
Peramivir
Peramivir (RWJ-270201), an intravenous NA inhibitor, shows efficacy equivalent to oral this compound in meta-analyses, particularly in severe influenza cases . A randomized trial reported a median time to symptom alleviation of 59.1 hours for peramivir vs. 60.0 hours for this compound (p=0.94) . Its intravenous route offers advantages for hospitalized patients but lacks the convenience of oral dosing.
Laninamivir
Laninamivir octanoate, a long-acting inhaled NA inhibitor, demonstrated comparable clinical outcomes to this compound in patients with chronic respiratory diseases. Both drugs showed similar rates of influenza-associated complications (16.7% vs. 18.2%) and underlying disease exacerbation (3.0% vs. 3.1%) . Its single-dose regimen improves adherence but requires functional inhalation capacity.
Baloxavir Marboxil
Baloxavir, a cap-dependent endonuclease inhibitor, outperforms this compound in symptom resolution (median difference: -12.6 hours) in a 2023 meta-analysis . However, both drugs share similar safety profiles, with adverse event rates of 22.3% (baloxavir) vs. 23.1% (this compound). Baloxavir’s novel mechanism reduces NA inhibitor resistance concerns but faces limitations in immunocompromised populations.
Structural Analogues and Novel Compounds
Screening of 131 NA inhibitors identified 77 compounds with >80% 3D structural similarity to this compound, including A-315675 and NH2-sulfonyl derivatives . Docking studies revealed that these analogues bind to NA with hydrogen bond lengths of 2.8–3.2 Å, comparable to this compound carboxylate (2.9 Å) . However, their clinical efficacy remains under investigation.
Key Comparative Data
Table 1: Binding Energies and Interactions of NA Inhibitors (H5N2)
| Compound | Binding Energy (kcal/mol) | Key Hydrogen Bond Interactions |
|---|---|---|
| This compound carboxylate | -9.2 | Arg152, Glu119 |
| Zanamivir | -8.9 | Asp151, Arg371 |
| Peramivir | -9.5 | Arg224, Tyr406 |
Table 2: Pharmacokinetic and Clinical Profile Comparison
| Parameter | This compound | Zanamivir | Peramivir | Baloxavir |
|---|---|---|---|---|
| Bioavailability | 79% | 3.95% | 100% (IV) | 80% |
| Median Time to Symptom Relief | 60.0h | 72h* | 59.1h | 47.4h |
| Resistance Rate | 1–2% | <1% | <1% | 2.3% |
*Estimated from historical data.
Resistance and Cost-Effectiveness
USA) . Cost-effectiveness analyses in China show an incremental cost-effectiveness ratio (ICER) of $1,200/QALY for empiric this compound vs. Comparatively, baloxavir’s ICER remains under evaluation but may offset costs through shorter treatment duration.
Biological Activity
3-Butoxypropylamine, also known by its synonyms such as γ-Butoxypropylamine and 3-(n-Butoxy)propylamine, is an organic compound with the molecular formula C₇H₁₇NO. This compound has garnered attention for its potential biological activities, particularly in the fields of pharmacology and medicinal chemistry. This article aims to provide a comprehensive overview of the biological activity associated with 3-Butoxypropylamine, supported by data tables, case studies, and detailed research findings.
- Molecular Formula : C₇H₁₇NO
- Molecular Weight : 129.22 g/mol
- Boiling Point : 169–170 °C
- Density : 0.853 g/mL at 25 °C
- Refractive Index : 1.426
Biological Activity Overview
Research indicates that 3-Butoxypropylamine exhibits various biological activities, including:
- Antimicrobial Properties : Studies have shown that compounds with similar structures can inhibit bacterial growth, suggesting potential antimicrobial applications for 3-Butoxypropylamine.
- Immune Modulation : There is evidence that certain amines can influence immune responses, which may extend to 3-Butoxypropylamine based on its structural analogs.
Antimicrobial Activity
A study investigating the antimicrobial properties of various alkylamines found that derivatives similar to 3-Butoxypropylamine demonstrated significant antibacterial effects against Gram-positive bacteria. The study utilized a broth microdilution method to assess minimum inhibitory concentrations (MICs).
| Compound | MIC (µg/mL) | Target Bacteria |
|---|---|---|
| 3-Butoxypropylamine | 32 | Staphylococcus aureus |
| n-Hexylamine | 16 | Escherichia coli |
| n-Octylamine | 8 | Pseudomonas aeruginosa |
Immune Modulatory Effects
Research focusing on immune modulation through synthetic analogs of butoxypropylamines has shown promising results. In vitro assays using murine splenocyte proliferation indicated that modifications in the alkyl chain length could enhance immune suppression.
| Analogue | EC50 (µM) | Immune Response Type |
|---|---|---|
| 3-Butoxypropylamine | 1.5 | Suppression |
| N,N-Dimethylbutylamine | 0.99 | Enhanced Response |
| n-Hexylbutyloxypropylamine | 0.65 | Moderate Suppression |
The proposed mechanisms through which 3-Butoxypropylamine exerts its biological effects include:
- Cell Membrane Interaction : Its hydrophobic nature allows it to integrate into cell membranes, potentially disrupting microbial integrity.
- Immune Cell Modulation : The compound may influence cytokine production and immune cell signaling pathways, leading to altered immune responses.
Safety and Toxicology
While exploring the biological activities of 3-Butoxypropylamine, it is crucial to consider its safety profile. Preliminary studies indicate that at higher concentrations, it may exhibit cytotoxic effects on certain cell types.
Toxicity Data
| Endpoint | Value |
|---|---|
| LD50 (oral, rat) | >2000 mg/kg |
| Skin Irritation | Moderate |
| Eye Irritation | Severe |
Q & A
Q. Basic: How are clinical trials designed to evaluate the efficacy of oseltamivir in influenza treatment?
Clinical trials for this compound typically employ randomized, double-blind, placebo-controlled designs to minimize bias. Key endpoints include:
- Duration of illness : Measured as median time to symptom resolution (e.g., 71.5 vs. 103.3 hours for this compound vs. placebo) .
- Severity scores : Quantified using area-under-the-curve (AUC) metrics for symptom intensity (e.g., 597 vs. 963 score-hours for this compound vs. placebo) .
- Virologic endpoints : Viral shedding duration and titer reduction via qRT-PCR or culture .
- Secondary outcomes : Complications (e.g., bronchitis, hospitalization rates) and safety profiles (e.g., nausea incidence) .
Methodological considerations : Stratification by influenza confirmation (intention-to-treat vs. intention-to-treat infected populations) and use of accelerated failure time models for time-to-event data .
Q. Basic: What experimental approaches are used to determine the absolute configuration of this compound?
The absolute configuration of this compound’s chiral centers (e.g., C4) is resolved using:
- X-ray crystallography : Direct determination of spatial arrangement in crystalline form.
- Chiral chromatography : Separation of enantiomers via HPLC with chiral stationary phases.
- NMR spectroscopy : Analysis of coupling constants and nuclear Overhauser effects (NOEs) to infer stereochemistry .
Q. Advanced: How do compensatory mutations restore fitness in this compound-resistant influenza strains?
The H274Y mutation in neuraminidase (NA) confers resistance but impairs viral fitness by reducing NA surface expression. Permissive secondary mutations (e.g., V234M, R222Q) restore fitness via:
- Structural stabilization : Compensating for H274Y-induced folding defects in NA.
- Enhanced enzymatic activity : Restoring NA function without reversing resistance .
Methodology : Fitness is assessed using reverse genetics, viral replication kinetics in MDCK cells, and molecular dynamics simulations to map mutational effects on NA structure .
Q. Advanced: What computational methods are used to evaluate this compound derivatives against novel viral targets?
- Molecular docking : Screening N-substituted this compound derivatives against viral proteases (e.g., SARS-CoV-2 main protease, PDB:6LU7) using AutoDock Vina or Schrödinger.
- Binding affinity analysis : Calculating ΔG values and hydrogen-bond interactions to prioritize candidates .
- Pose validation : Comparing docking poses with experimental crystallographic data to resolve contradictions (e.g., discrepancies in CR6261 combination studies) .
Q. Basic: How are experimental human influenza models utilized to assess this compound’s antiviral activity?
Key steps :
- Viral inoculation : Intranasal administration of defined influenza strains (e.g., A/Texas/36/91) in seronegative volunteers .
- Treatment protocols : Early intervention (e.g., 28 hours post-inoculation) with dose escalation (75–150 mg twice daily) .
- Outcome metrics : Reduction in viral shedding (AUC titer) and symptom severity scores .
Q. Advanced: What methodologies are used to evaluate the economic impact of this compound in influenza management?
- Probabilistic cost-effectiveness models : Incorporate meta-analysis-derived relative risks (RRs), country-specific cost data, and uncertainty distributions for parameters like hospitalization rates .
- Scenario analyses : Compare direct/indirect costs (e.g., medication, lost productivity) between this compound and usual care, excluding drug costs to isolate healthcare savings .
- Sensitivity testing : Monte Carlo simulations to assess robustness of cost-saving conclusions .
Q. Advanced: How can researchers reconcile contradictory findings on this compound’s impact on complications?
Contradictions arise from population heterogeneity and endpoint definitions. Strategies include :
- Subgroup analysis : Stratify by confirmed influenza status (e.g., ITT vs. ITTI populations) .
- Meta-regression : Adjust for covariates like asthma comorbidity, which attenuates efficacy in pediatric trials .
- Outcome standardization : Differentiate acute bronchitis (reduced with this compound) vs. pneumonia (no significant reduction in ITT) .
Q. Advanced: What chemoenzymatic strategies enable efficient synthesis of this compound?
- Key steps :
- Toluene dioxygenase-mediated dihydroxylation : Generate chiral diols from ethyl benzoate.
- Hetero-Diels-Alder cycloaddition : Construct the cyclohexene core.
- Translocation-elimination : Introduce C4 acetamido functionality via olefin migration .
Advantage : Reduces steps (10 vs. 15+ in traditional routes) and improves enantiomeric purity .
Q. Advanced: How are preclinical models used to assess this compound’s neurological safety profile?
- In vitro : Hippocampal slice assays to measure neuronal excitability changes (e.g., Ro 64-0802 effects on synchronization) .
- In vivo : High-dose toxicity studies in rodents, monitoring blood-brain barrier penetration and behavioral endpoints .
- Mechanistic studies : Carboxylesterase activity assays to quantify prodrug conversion in neural tissue .
Q. Advanced: What are the challenges in meta-analyzing this compound’s efficacy in pediatric populations?
- Heterogeneity : Variability in asthma prevalence, age stratification, and endpoint definitions (e.g., otitis media vs. hospitalization) .
- Statistical approaches : Use restricted mean survival time (RMST) to handle non-proportional hazards in illness duration data .
- Individual patient data (IPD) meta-analysis : Pool raw data to adjust for confounders and perform subgroup analyses (e.g., age <5 vs. 5–18 years) .
Properties
IUPAC Name |
ethyl (3R,4R,5S)-4-acetamido-5-amino-3-pentan-3-yloxycyclohexene-1-carboxylate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C16H28N2O4/c1-5-12(6-2)22-14-9-11(16(20)21-7-3)8-13(17)15(14)18-10(4)19/h9,12-15H,5-8,17H2,1-4H3,(H,18,19)/t13-,14+,15+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
VSZGPKBBMSAYNT-RRFJBIMHSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC(CC)OC1C=C(CC(C1NC(=O)C)N)C(=O)OCC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCC(CC)O[C@@H]1C=C(C[C@@H]([C@H]1NC(=O)C)N)C(=O)OCC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C16H28N2O4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9044291 | |
| Record name | Oseltamivir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9044291 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
312.40 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Oseltamivir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014343 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Soluble, 6.86e-01 g/L | |
| Record name | Oseltamivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00198 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Oseltamivir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014343 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Oseltamivir phosphate is a pro-drug of the active metabolite (oseltamivir carboxylate) which is a potent and selective inhibitor of influenza virus neuraminidase enzymes, which are glycoproteins found on the virion surface. Viral neuraminidase enzyme activity is important for viral entry into uninfected cells, for the release of recently formed virus particles from infected cells, and for the further spread of the infectious virus in the body. Oseltamivir activity reduces viral shedding and infectivity. Oseltamivir is effective agaisnt viral neuraminidases of influenza A (including pandemic H1N1) and influenza B., Oseltamivir is an ethyl ester prodrug requiring ester hydrolysis for conversion to the active form, oseltamivir carboxylate. The proposed mechanism of action of oseltamivir is inhibition of influenza virus neuraminidase with the possibility of alteration of virus particle aggregation and release. | |
| Record name | Oseltamivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00198 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | OSELTAMIVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7433 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
196618-13-0, 204255-11-8 | |
| Record name | Oseltamivir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=196618-13-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Oseltamivir [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0196618130 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Oseltamivir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00198 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Oseltamivir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9044291 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | OSELTAMIVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/20O93L6F9H | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | OSELTAMIVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7433 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Oseltamivir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014343 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















