Tafenoquine
Overview
Description
Tafenoquine is an antimalarial medication used for both the prevention and treatment of malaria. It is particularly effective against Plasmodium vivax, a parasite responsible for relapsing malaria. This compound belongs to the 8-aminoquinoline class of compounds and was approved for medical use in Australia and the United States in 2018 . It is known for its long half-life, which allows for single-dose treatment, making it a convenient option for patients .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of tafenoquine involves several steps, starting with the preparation of key intermediates. One of the critical intermediates is 4-methyl-5-(3-trifluoromethylphenoxy)-6-methoxy-8-aminoquinoline. The synthesis typically involves the following steps:
Formation of the Quinoline Core: The quinoline core is synthesized through a series of reactions, including nitration, reduction, and cyclization.
Introduction of Functional Groups: Various functional groups, such as methoxy and trifluoromethylphenoxy, are introduced through substitution reactions.
Final Assembly: The final step involves coupling the intermediate with a suitable amine to form this compound.
Industrial Production Methods: Industrial production of this compound follows similar synthetic routes but is optimized for large-scale manufacturing. This includes the use of efficient catalysts, high-yield reactions, and purification techniques to ensure the production of high-purity this compound .
Chemical Reactions Analysis
Types of Reactions: Tafenoquine undergoes several types of chemical reactions, including:
Oxidation: this compound can be oxidized to form various metabolites.
Reduction: Reduction reactions can modify the quinoline core, affecting its antimalarial activity.
Substitution: Substitution reactions are used to introduce different functional groups during synthesis.
Common Reagents and Conditions:
Oxidizing Agents: Hydrogen peroxide, potassium permanganate.
Reducing Agents: Sodium borohydride, lithium aluminum hydride.
Substitution Reagents: Halogenated compounds, alkylating agents.
Major Products: The major products formed from these reactions include various this compound analogs and metabolites, which are studied for their antimalarial properties .
Scientific Research Applications
Radical Cure of Malaria
Overview
Tafenoquine has been approved for the radical cure of Plasmodium vivax malaria. Its mechanism involves targeting the hypnozoite stage of the parasite, which is responsible for relapses. Unlike the traditional treatment with primaquine that requires a 14-day regimen, this compound can be administered as a single dose of 300 mg, significantly improving patient compliance and treatment outcomes .
Clinical Evidence
Two pivotal Phase III trials, DETECTIVE and GATHER, demonstrated this compound's efficacy in preventing relapse. In these studies involving over 500 patients from various malaria-endemic countries, this compound showed superior results compared to primaquine in terms of relapse prevention . The drug's safety profile was also favorable, with no significant black-box warnings reported .
Prophylaxis Against Malaria
Overview
this compound is marketed under the name Arakoda for prophylaxis against malaria. It offers a convenient once-weekly dosing schedule following a three-day loading dose. This regimen is especially beneficial for travelers and military personnel who may be exposed to malaria in endemic regions .
Clinical Studies
Research indicates that this compound is effective against both liver and blood stages of malaria caused by P. vivax and P. falciparum. Its prophylactic use has been associated with a low incidence of drug resistance and comparable adverse event rates to placebo over extended periods .
Potential Applications Beyond Malaria
Antifungal Activity
Emerging research suggests that this compound may possess antifungal properties. In animal models, it has shown efficacy against certain fungal strains and has been proposed for treating Pneumocystis pneumonia in immunocompromised patients. However, clinical trials are necessary to establish its safety and efficacy for these indications .
Treatment of Babesiosis
this compound has demonstrated effectiveness in cases of babesiosis, particularly in patients who did not respond to conventional antibiotic therapies. In recent clinical case studies, this compound administration led to successful treatment outcomes in immunosuppressed patients with babesiosis .
Pharmacokinetics and Safety Considerations
Pharmacokinetic Profile
The pharmacokinetics of this compound have been extensively studied, revealing important insights into its absorption, distribution, metabolism, and excretion (ADME). It exhibits linear pharmacokinetics with predictable dosing responses, making it easier to manage in clinical settings .
Safety Profile
Despite its benefits, this compound is contraindicated in patients with G6PD deficiency due to potential hemolytic toxicity. It is crucial for clinicians to perform G6PD testing prior to prescribing this compound to mitigate risks associated with hemolysis .
Summary Table: this compound Applications
Mechanism of Action
The exact mechanism of action of tafenoquine is not fully understood. it is believed to involve the following:
Molecular Targets: this compound targets both the liver and blood stages of Plasmodium parasites.
Pathways Involved: It is thought to interfere with the parasite’s mitochondrial function and redox cycling, leading to the production of reactive oxygen species that damage the parasite
Comparison with Similar Compounds
Primaquine: Another 8-aminoquinoline used for the treatment of malaria. Unlike tafenoquine, primaquine requires daily dosing for 14 days.
Chloroquine: A 4-aminoquinoline used for the treatment of malaria but less effective against relapsing malaria caused by Plasmodium vivax.
Uniqueness of this compound:
Long Half-Life: this compound’s long half-life allows for single-dose treatment, making it more convenient for patients.
Broad Spectrum: Effective against both liver and blood stages of Plasmodium parasites, providing a comprehensive treatment option
This compound represents a significant advancement in the treatment and prevention of malaria, offering a convenient and effective option for patients worldwide.
Biological Activity
Tafenoquine is a novel antimalarial agent primarily indicated for the radical cure of Plasmodium vivax malaria. Its mechanism of action, pharmacokinetics, and therapeutic efficacy have been subjects of extensive research. This article synthesizes current findings on this compound's biological activity, focusing on its mechanisms, clinical efficacy, and potential applications beyond malaria treatment.
This compound belongs to the 8-aminoquinoline class of compounds. It acts by targeting the liver stages of P. vivax, specifically the hypnozoites, which are responsible for relapses in malaria. The drug is thought to exert its effects through oxidative metabolites that induce oxidative stress within the parasite, leading to cell death. Studies have shown that this compound's terminal elimination half-life is significantly longer than that of primaquine (approximately 15 days compared to 5 hours), which complicates the measurement of its active metabolites in vivo .
Pharmacokinetics
This compound exhibits a complex pharmacokinetic profile characterized by:
- Absorption : Slow absorption with a time to maximum concentration (t_max) of approximately 12 hours.
- Elimination Half-Life : Approximately 13-15 days, allowing for sustained drug levels in the bloodstream.
- Dose-Response Relationship : Higher doses correlate with increased efficacy and reduced recurrence rates of P. vivax malaria .
The pharmacokinetic parameters are summarized in Table 1.
| Parameter | Value |
|---|---|
| Time to Maximum Concentration (t_max) | ~12 hours |
| Elimination Half-Life (t_1/2) | ~13-15 days |
| Maximum Plasma Concentration (C_max) | Dose-dependent |
Radical Cure of P. vivax
Clinical trials have demonstrated this compound's efficacy in achieving radical cure from P. vivax. A pivotal study indicated that a single dose of 300 mg resulted in a relapse-free efficacy rate of approximately 11%, which was significantly lower than the rates observed with primaquine . The efficacy increases with higher doses; for instance, doses around 450 mg can achieve up to 90% reduction in relapse risk .
Case Studies
A notable case series explored this compound's application in treating relapsing babesiosis caused by Babesia microti. In this context, this compound was administered as an adjunct therapy. The treatment regimen included an initial loading dose of 600 mg followed by maintenance doses ranging from 200 mg to 300 mg weekly. The results indicated successful clearance of the infection in most cases, showcasing this compound's potential beyond its primary indication for malaria .
Comparative Studies
A comparative analysis between this compound and other antimalarial treatments revealed significant differences in efficacy:
- This compound vs. Primaquine : this compound showed lower relapse rates compared to primaquine when co-administered with dihydroartemisinin-piperaquine.
- Efficacy Metrics : Kaplan-Meier estimates indicated a relapse-free efficacy rate of 52% for primaquine versus only 11% for this compound when used alone .
Safety Profile
The safety profile of this compound has been evaluated across multiple studies. Adverse events were reported in about 54% of patients during initial trials, with most being mild to moderate in severity. Monitoring for hemolysis is essential due to the risk associated with G6PD deficiency, which can exacerbate hemolytic reactions when treated with this compound .
Q & A
Q. Basic: What validated HPLC methodologies are available for quantifying tafenoquine in pharmaceutical formulations?
Answer:
A validated isocratic HPLC method using methanol-water (70:30 to 80:20 ratio) achieves baseline separation of this compound from degradation products. Key validation parameters include:
- Linearity : R² > 0.999 across 10–50 µg/mL.
- Precision : %RSD < 1% for retention time (RT) and peak area.
- Forced degradation : Acidic/alkaline hydrolysis, oxidation (H₂O₂), photolysis, and thermal stress are used to assess stability. Symmetrical peaks confirm method specificity under degradation conditions .
Q. Basic: What are the foundational pharmacokinetic (PK) parameters of this compound in preclinical and clinical studies?
Answer:
Key PK parameters include:
- Terminal half-life : ~14 days in humans, enabling long-acting prophylaxis.
- Clearance (CL/F) : 0.056 L/h/kg (population PK model).
- Volume of distribution (V/F) : 23.7 L/kg, indicating extensive tissue penetration.
- Metabolism : Primarily hepatic, with unstable intermediates complicating metabolite identification .
Q. Advanced: How are population pharmacokinetic (PopPK) models developed for this compound dose optimization?
Answer:
PopPK models integrate data from phase 1–3 trials using NONMEM software. Key steps:
- Covariate analysis : Weight linearly correlates with CL/F and V/F but lacks clinical significance for dose adjustment.
- Validation : Bootstrap analysis (200 samples) and visual predictive checks ensure model robustness.
- Applications : Bridging PK across formulations (e.g., pediatric studies) avoids redundant bioequivalence trials .
Q. Advanced: How can contradictions in this compound metabolism data be resolved in translational studies?
Answer:
Contradictions arise from unstable metabolites and interspecies variability. Methodological solutions include:
- Bayesian modeling : Links parent drug exposure (AUC, Cmax) and oxidative metabolites (via day 7 methaemoglobin levels) to efficacy.
- Dose-response adjustments : Higher doses (7.5 mg/kg vs. 5 mg/kg) improve radical cure rates by enhancing metabolite-driven hypnozoiticidal activity .
Q. Advanced: What mathematical modeling approaches evaluate this compound’s impact on Plasmodium vivax transmission?
Answer:
Compartmental models calibrated to regional epidemiological data (e.g., Brazil) incorporate:
- Parameters : Relapse rates, G6PD deficiency prevalence, and adherence to primaquine/tafenoquine regimens.
- Outcome : this compound reduces transmission by 60–80% in high-relapse settings due to improved compliance vs. 14-day primaquine .
Q. Basic: What methodologies compare this compound and primaquine efficacy in relapse prevention?
Answer:
Randomized controlled trials (RCTs) use:
- Primary endpoint : Parasite recurrence within 4–6 months.
- Dose standardization : this compound 300–600 mg (single dose) vs. primaquine 15 mg/day ×14 days.
- Meta-analysis : this compound shows superior efficacy (RR 0.29 vs. primaquine) but requires G6PD screening .
Q. Advanced: How are PK/PD models applied to optimize this compound dosing regimens?
Answer:
Emax models correlate mg/kg doses with radical cure efficacy:
- Current dose : 5 mg/kg achieves ~70% of maximal hypnozoiticidal effect.
- Projected dose : 7.5 mg/kg increases efficacy to 90% but requires safety trials for haemolytic risk .
Q. Basic: How is this compound stability assessed in analytical method development?
Answer:
Forced degradation under ICH guidelines includes:
- Solvent systems : Methanol-water ratios tested for peak symmetry.
- Stress conditions : Acid (0.1N HCl), base (0.1N NaOH), H₂O₂ (3%), UV light (254 nm), and heat (60°C).
- Validation : % recovery (98–102%) and precision (%RSD < 2%) confirm method reliability .
Q. Advanced: How are G6PD deficiency and haemolytic risk integrated into this compound trial design?
Answer:
- Screening : Enzyme activity thresholds (>70% population median) via quantitative assays.
- Safety endpoints : Post-treatment haemoglobin declines (0.02 g/dL per mg/kg dose increase).
- Ethical protocols : Exclude G6PD-deficient participants and monitor methaemoglobin levels .
Q. Advanced: What statistical methods reconcile conflicting efficacy data in this compound meta-analyses?
Answer:
Individual patient data (IPD) meta-analyses apply:
Properties
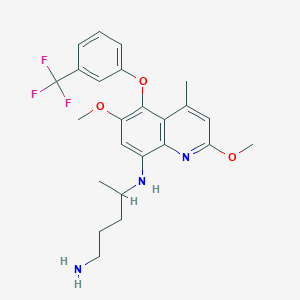
IUPAC Name |
4-N-[2,6-dimethoxy-4-methyl-5-[3-(trifluoromethyl)phenoxy]quinolin-8-yl]pentane-1,4-diamine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C24H28F3N3O3/c1-14-11-20(32-4)30-22-18(29-15(2)7-6-10-28)13-19(31-3)23(21(14)22)33-17-9-5-8-16(12-17)24(25,26)27/h5,8-9,11-13,15,29H,6-7,10,28H2,1-4H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
LBHLFPGPEGDCJG-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=CC(=NC2=C1C(=C(C=C2NC(C)CCCN)OC)OC3=CC=CC(=C3)C(F)(F)F)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C24H28F3N3O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
106635-81-8 (maleate) | |
| Record name | Tafenoquine [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0106635807 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID90869466 | |
| Record name | Tafenoquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90869466 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
463.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Insoluble | |
| Record name | Tafenoquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06608 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
The mechanism of action of tafenoquine is not well established but studies have reported a longer and more effective action when compared to primaquine. The active moiety of tafenoquine, 5,6 ortho quinone tafenoquine, seems to be redox cycled by _P. falciparum_ which are upregulated in gametocytes and liver stages. Once inside, the oxidated metabolite produces hydrogen peroxide and hydroxyl radicals. It is thought that these radicals produce leads to the parasite death. On the other hand, tafenoquine inhibits heme polymerase in blood stage of parasites which explains the activity against blood stages of parasites. | |
| Record name | Tafenoquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06608 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
106635-80-7 | |
| Record name | Tafenoquine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=106635-80-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Tafenoquine [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0106635807 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Tafenoquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06608 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Tafenoquine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90869466 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | TAFENOQUINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/262P8GS9L9 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















