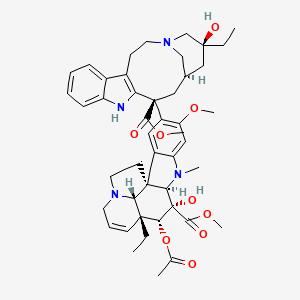
Vinblastine
Overview
Description
Vinblastine is a vinca alkaloid derived from the Madagascar periwinkle plant, Catharanthus roseus . It is primarily used as a chemotherapy medication to treat various types of cancer, including Hodgkin’s lymphoma, non-small-cell lung cancer, bladder cancer, brain cancer, melanoma, and testicular cancer . This compound works by inhibiting cell division, making it an effective antineoplastic agent .
Preparation Methods
Synthetic Routes and Reaction Conditions: Vinblastine can be synthesized through a complex biosynthetic pathway involving the coupling of vindoline and catharanthine . This process has been successfully replicated in engineered yeast, which can produce the necessary precursors . The synthetic route involves multiple steps, including the formation of strictosidine, followed by the production of catharanthine and vindoline, and their subsequent coupling to form this compound .
Industrial Production Methods: Industrial production of this compound typically involves the extraction of vindoline and catharanthine from Catharanthus roseus, followed by their chemical coupling . Advances in synthetic biology have enabled the production of these alkaloids in engineered yeast, providing a more sustainable and scalable method .
Chemical Reactions Analysis
Types of Reactions: Vinblastine undergoes various chemical reactions, including oxidation, reduction, and substitution .
Common Reagents and Conditions:
Reduction: Reduction reactions can be carried out using hydrogen gas in the presence of a palladium catalyst.
Substitution: Substitution reactions often involve nucleophiles such as hydroxide ions or amines.
Major Products: The major products formed from these reactions depend on the specific conditions and reagents used. For example, oxidation can lead to the formation of various oxidized derivatives, while reduction can yield reduced forms of this compound .
Scientific Research Applications
Vinblastine has a wide range of scientific research applications:
Chemistry: It is used as a model compound to study the synthesis and modification of complex alkaloids.
Biology: Researchers use this compound to investigate cell division and microtubule dynamics.
Medicine: this compound is a critical component of chemotherapy regimens for treating various cancers.
Mechanism of Action
Vinblastine exerts its effects by binding to tubulin, a protein that is essential for the formation of microtubules . By inhibiting microtubule formation, this compound disrupts the mitotic spindle, leading to cell cycle arrest at metaphase and ultimately causing cell death . This mechanism makes this compound an effective antineoplastic agent, particularly in rapidly dividing cancer cells .
Comparison with Similar Compounds
These compounds share a similar mechanism of action but differ in their chemical structures and clinical applications . For example:
Vincristine: Used primarily to treat leukemia and lymphoma.
Vindesine: Used in the treatment of various cancers, including melanoma and lung cancer.
Vinflunine: Primarily used for bladder cancer.
Vinblastine is unique in its specific combination of vindoline and catharanthine, which contributes to its distinct pharmacological profile and therapeutic applications .
Q & A
Basic Research Questions
Q. How do researchers optimize in vitro models to study vinblastine’s effects on cancer cell proliferation?
Methodological Answer: In vitro models require standardization of cell lines (e.g., MCF-7, HepG2) and culture conditions. For cytotoxicity assays, use the MTT cell proliferation assay with this compound concentrations ranging from 0.00625 µM to 320 µM, depending on cell type and treatment duration . Ensure synchronization of cell cycles (e.g., lag, exponential, stationary phases) to account for phase-dependent drug accumulation, as this compound uptake varies significantly (e.g., 1.74 µg/mg in lag phase vs. 2.61 µg/mg in stationary phase) .
Q. What statistical methods are recommended for analyzing contradictory data in this compound pharmacokinetics?
Methodological Answer: Use ANOVA with post-hoc tests (e.g., Tukey’s HSD) to compare means across experimental groups. For dose-response studies, apply nonlinear regression models (e.g., log-logistic curves) to calculate IC50 values. Address outliers by combining parametric tests with nonparametric alternatives (e.g., Mann-Whitney U test) . When discrepancies arise in cytotoxicity data, systematically review variables like cell density, S9 metabolic activation, or solvent controls (e.g., DMSO interference) .
Advanced Research Questions
Q. How can researchers resolve contradictions in this compound’s mechanism of resistance across different cancer types?
Methodological Answer: Design comparative studies using RNA sequencing or CRISPR-Cas9 screens to identify resistance-associated genes (e.g., ABC transporters). Validate findings with functional assays, such as efflux inhibition via verapamil co-treatment. For clinical relevance, correlate in vitro resistance data with patient-derived xenograft (PDX) models and clinical trial datasets . Cross-validate results using orthogonal methods like flow cytometry for P-glycoprotein expression analysis .
Q. What experimental designs are effective for studying synergistic interactions between this compound and novel agents (e.g., nanoliposomal ceramides)?
Methodological Answer: Use the Chou-Talalay method to calculate combination indices (CI) via CalcuSyn software. Test fixed-ratio combinations (e.g., 1:1, 1:2) across multiple concentrations (e.g., 0.008–1 µM this compound + 12–24 µM ceramide). Measure synergy through caspase 3/7 activity assays and validate with Annexin V/PI staining . Ensure statistical rigor by performing ANOVA with Dunnett’s T-test to compare monotherapy vs. combination effects .
Q. How should researchers address variability in this compound accumulation data across cell cycle phases?
Methodological Answer: Synchronize cells using serum starvation or chemical inhibitors (e.g., thymidine block) before treatment. Quantify intracellular this compound via HPLC-MS/MS, normalizing to cell dry weight. Report phase-specific accumulation with error bars representing SEM (e.g., lag phase: 1.74 ± 0.2 µg/mg; stationary phase: 2.61 ± 0.3 µg/mg) . Use time-lapse microscopy to correlate accumulation dynamics with mitotic arrest.
Q. Methodological Frameworks
Q. What protocols ensure reproducibility in this compound’s cytotoxicity assays?
- Cell Line Validation : Authenticate cell lines via STR profiling.
- Dose Standardization : Include positive controls (e.g., cyclophosphamide) and solvent controls .
- Data Reporting : Adhere to MIAME guidelines for omics data or ARRIVE guidelines for preclinical studies .
Q. How to design experiments investigating this compound’s impact on microtubule dynamics?
- Use fluorescently labeled tubulin (e.g., GFP-α-tubulin) and live-cell imaging to track polymerization rates.
- Compare this compound with other vinca alkaloids (e.g., vincristine) using EC50 values derived from dose-response curves .
Q. Data Contradiction Analysis
Q. Why do studies report conflicting EC50 values for this compound in similar cell lines?
Potential Variables:
- Metabolic Activation : Inclusion/exclusion of S9 liver fractions alters drug metabolism .
- Assay Duration : Shorter exposures (24h) may underestimate cytotoxicity vs. prolonged treatments (72h).
- Normalization Methods : Data normalized to cell count vs. protein content yield divergent results .
Q. Tables for Reference
Table 1 : this compound Accumulation in Catharanthus roseus Cell Cultures Across Growth Phases
| Growth Phase | Days Post-Culture | This compound (µg/mg dry weight) | Vincristine (µg/mg dry weight) |
|---|---|---|---|
| Lag | 3 | 1.74 ± 0.2 | 1.83 ± 0.3 |
| Exponential | 7 | 2.11 ± 0.3 | 2.27 ± 0.4 |
| Stationary | 12 | 2.61 ± 0.3 | 2.52 ± 0.2 |
Table 2 : Synergy Testing of this compound + C6-Ceramide in HepG2 Cells
| This compound (µM) | C6-Ceramide (µM) | Caspase 3/7 Activity (% Control) | p-value vs. Monotherapy |
|---|---|---|---|
| 0.008 | 12 | 145 ± 12 | <0.05 |
| 0.025 | 24 | 218 ± 18 | <0.01 |
Properties
CAS No. |
865-21-4 |
|---|---|
Molecular Formula |
C46H58N4O9 |
Molecular Weight |
811.0 g/mol |
IUPAC Name |
methyl (9R,10S,11R,12R,19R)-11-acetyloxy-12-ethyl-4-[(13S,15R,17S)-17-ethyl-17-hydroxy-13-methoxycarbonyl-1,11-diazatetracyclo[13.3.1.04,12.05,10]nonadeca-4(12),5,7,9-tetraen-13-yl]-10-hydroxy-5-methoxy-8-methyl-8,16-diazapentacyclo[10.6.1.01,9.02,7.016,19]nonadeca-2,4,6,13-tetraene-10-carboxylate |
InChI |
InChI=1S/C46H58N4O9/c1-8-42(54)23-28-24-45(40(52)57-6,36-30(15-19-49(25-28)26-42)29-13-10-11-14-33(29)47-36)32-21-31-34(22-35(32)56-5)48(4)38-44(31)17-20-50-18-12-16-43(9-2,37(44)50)39(59-27(3)51)46(38,55)41(53)58-7/h10-14,16,21-22,28,37-39,47,54-55H,8-9,15,17-20,23-26H2,1-7H3/t28-,37-,38+,39+,42-,43+,44?,45-,46-/m0/s1 |
InChI Key |
JXLYSJRDGCGARV-JQQWJEIDSA-N |
SMILES |
CCC1(CC2CC(C3=C(CCN(C2)C1)C4=CC=CC=C4N3)(C5=C(C=C6C(=C5)C78CCN9C7C(C=CC9)(C(C(C8N6C)(C(=O)OC)O)OC(=O)C)CC)OC)C(=O)OC)O |
Isomeric SMILES |
CC[C@@]1(C[C@H]2C[C@@](C3=C(CCN(C2)C1)C4=CC=CC=C4N3)(C5=C(C=C6C(=C5)C78CCN9[C@H]7[C@@](C=CC9)([C@H]([C@@]([C@@H]8N6C)(C(=O)OC)O)OC(=O)C)CC)OC)C(=O)OC)O |
Canonical SMILES |
CCC1(CC2CC(C3=C(CCN(C2)C1)C4=CC=CC=C4N3)(C5=C(C=C6C(=C5)C78CCN9C7C(C=CC9)(C(C(C8N6C)(C(=O)OC)O)OC(=O)C)CC)OC)C(=O)OC)O |
Color/Form |
Solvated needles from methanol |
melting_point |
211-216 °C |
Key on ui other cas no. |
865-21-4 |
physical_description |
Solid |
shelf_life |
SOLN MAY BE STORED IN REFRIGERATOR FOR PERIODS OF 30 DAYS WITHOUT SIGNIFICANT LOSS OF POTENCY /VINBLASTINE SULFATE/ |
solubility |
Negligible ODORLESS & HYGROSCOPIC; WHITE TO SLIGHTLY YELLOW, AMORPHOUS OR CRYSTALLINE POWDER; FREELY SOL IN WATER /VINBLASTINE SULFATE/ Practically insoluble in water, petroleum ether; soluble in alcohols, acetone, ethyl acetate, chloroform |
Synonyms |
cellblastin Lemblastine Sulfate, Vinblastine Velban Velbe Vinblastin Hexal Vinblastina Lilly Vinblastine Vinblastine Sulfate Vinblastinsulfat-Gry Vincaleukoblastine |
vapor_pressure |
1.03X10-27 mm Hg at 25 °C (est) |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















