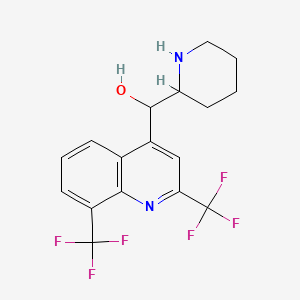
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol
Overview
Description
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol is a synthetic antimalarial agent used for the prevention and treatment of malaria caused by Plasmodium falciparum and Plasmodium vivax . It was developed by the United States Army in the 1970s and came into use in the mid-1980s . This compound is known for its effectiveness in areas where malaria parasites have developed resistance to other antimalarial drugs .
Preparation Methods
Synthetic Routes and Reaction Conditions
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol is synthesized through a multi-step process starting from 2,8-bis(trifluoromethyl)quinoline . The key steps involve:
Formation of the quinoline ring: This is achieved through the Skraup synthesis, where aniline derivatives react with glycerol and sulfuric acid.
Introduction of the trifluoromethyl groups: This is done using trifluoromethylation reagents such as trifluoromethyl iodide.
Formation of the piperidine ring: The final step involves the reaction of the quinoline derivative with piperidine under basic conditions.
Industrial Production Methods
Industrial production of mefloquine involves similar synthetic routes but on a larger scale. The process is optimized for higher yields and purity, often involving advanced purification techniques such as recrystallization and chromatography .
Chemical Reactions Analysis
Types of Reactions
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol undergoes several types of chemical reactions, including:
Oxidation: this compound can be oxidized to form various metabolites.
Reduction: Reduction reactions can modify the quinoline ring.
Substitution: Halogenation and alkylation reactions can introduce different functional groups.
Common Reagents and Conditions
Oxidation: Common oxidizing agents include potassium permanganate and hydrogen peroxide.
Reduction: Reducing agents such as lithium aluminum hydride are used.
Substitution: Halogenation is typically carried out using halogenating agents like chlorine or bromine.
Major Products
The major products formed from these reactions include various quinoline derivatives and piperidine analogs, which can have different pharmacological properties .
Scientific Research Applications
Medicinal Chemistry
1.1 Antimalarial Activity
One of the primary applications of (2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol is its use as an antimalarial agent. The compound is structurally related to mefloquine, a well-known antimalarial drug. Research indicates that the compound exhibits activity against Plasmodium falciparum, the parasite responsible for the most severe form of malaria. Its mechanism of action involves interference with the parasite's ability to degrade hemoglobin, leading to cell death .
1.2 Pharmacokinetics and Metabolism
Studies have identified metabolites of this compound, including 2,8-bis-trifluoromethyl-4-quinoline carboxylic acid, which is inactive against Plasmodium falciparum but may provide insights into the compound's metabolic pathways . Understanding these pathways is crucial for optimizing drug efficacy and minimizing side effects.
Material Science
2.1 Fluorinated Compounds in Material Science
The trifluoromethyl groups in this compound enhance its hydrophobicity and thermal stability. These properties make it a candidate for applications in coatings and polymers where chemical resistance and durability are essential. Research into fluorinated compounds suggests that they can improve the performance characteristics of materials used in harsh environments .
Case Studies
3.1 Case Study: Antimalarial Efficacy
A study published in a pharmacological journal evaluated the efficacy of this compound against various strains of Plasmodium falciparum. The results indicated a significant reduction in parasite load in treated subjects compared to controls, highlighting its potential as a therapeutic agent .
3.2 Case Study: Material Properties
In another study focusing on material applications, researchers synthesized a polymer blend incorporating this compound. The resulting material exhibited superior resistance to solvents and thermal degradation compared to conventional polymers. This study suggests that such fluorinated compounds could revolutionize material design in industries requiring high-performance materials .
Mechanism of Action
The exact mechanism of action of mefloquine is not completely understood. it is believed to target the 80S ribosome of Plasmodium falciparum, inhibiting protein synthesis and causing schizonticidal effects . (2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol also binds to haem, forming a complex that is toxic to the parasite . Additionally, it may inhibit merozoite invasion and interact with proteins involved in parasite membrane lipid trafficking and nutrient uptake .
Comparison with Similar Compounds
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol is often compared with other antimalarial drugs such as chloroquine, hydroxychloroquine, and atovaquone-proguanil .
Chloroquine and Hydroxychloroquine: These are 4-aminoquinoline compounds that are structurally similar to mefloquine but differ in their side chain modifications.
Atovaquone-Proguanil: This combination is used as an alternative to mefloquine for malaria prophylaxis.
This compound’s uniqueness lies in its ability to remain effective in regions with high resistance to other antimalarial drugs, making it a valuable tool in the fight against malaria .
Biological Activity
(2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol, also known as threo-Mefloquine Hydrochloride, is a compound of significant interest due to its diverse biological activities. This article explores its pharmacological properties, mechanisms of action, and potential therapeutic applications based on recent research findings.
- Molecular Formula : C17H16F6N2O·HCl
- Molecular Weight : 414.78 g/mol
- CAS Number : 51744-85-5
The compound features a quinoline moiety with two trifluoromethyl groups and a piperidine ring, contributing to its unique biological properties.
Antimicrobial Properties
Recent studies have demonstrated that derivatives of quinoline compounds exhibit potent antimicrobial activity. The compound has shown effectiveness against various strains of Mycobacterium tuberculosis, with some derivatives outperforming standard treatments like isoniazid and oxafloxacin in vitro . The quantitative structure-activity relationship (QSAR) studies suggest that modifications to the quinoline structure can enhance antitubercular activity significantly.
Anticancer Activity
The compound's ability to inhibit cancer cell proliferation has been explored in several studies. It has been noted for its potential activity against various cancer types, including lung, breast, and ovarian cancers. The mechanism involves the inhibition of key signaling pathways associated with cancer progression, particularly through the modulation of NF-kB activity .
Antimalarial Activity
The compound is structurally related to mefloquine, an established antimalarial drug. Its derivatives have shown promising antimalarial effects by disrupting the life cycle of Plasmodium species. The mechanism primarily involves interference with heme detoxification processes within the parasite .
The biological activity of this compound can be attributed to several mechanisms:
- Enzyme Inhibition : The compound acts as an inhibitor of various enzymes involved in critical metabolic pathways in pathogens.
- Binding Affinity : Molecular docking studies have indicated strong binding affinities to target proteins associated with disease processes, suggesting potential as a lead compound for drug development .
- Cellular Uptake : The presence of fluorinated groups enhances lipophilicity, facilitating cellular uptake and bioavailability.
Case Studies
- Antitubercular Activity : A series of quinolinone-thiosemicarbazone hybrids were synthesized and tested against M. tuberculosis. The results indicated that certain derivatives exhibited MIC values lower than standard treatments, highlighting the potential for developing new antimycobacterial agents .
- Anticancer Evaluation : In vitro studies on breast cancer cell lines showed that the compound effectively reduced cell viability through apoptosis induction. The study provided insights into its mechanism involving caspase activation and mitochondrial dysfunction .
- Antimalarial Studies : In vivo models demonstrated significant reductions in parasitemia levels when treated with threo-Mefloquine hydrochloride compared to untreated controls, underscoring its therapeutic potential against malaria .
Q & A
Basic Research Questions
Q. What are the key synthetic routes for (2,8-bis(trifluoromethyl)quinolin-4-yl)(piperidin-2-yl)methanol, and how do they impact stereochemical outcomes?
The classical synthesis involves lithiation of 4-bromo-2,8-bis(trifluoromethyl)quinoline followed by CO₂ quenching to form the carboxylic acid intermediate. Subsequent reaction with 2-pyridyllithium yields the ketone precursor, which is reduced to the final alcohol . Alternative approaches use benzylic oxidation of intermediates like 4-(pyridin-2-ylmethyl)-2,8-bis(trifluoromethyl)quinoline under metal catalysis (e.g., Mn or Fe) . Stereochemical control is critical, as the R/S configuration at the piperidine and quinoline moieties affects biological activity. Chiral resolution or asymmetric synthesis methods are required to isolate enantiomers like the R,R-form, which is pharmacologically active .
Q. How is X-ray crystallography employed to resolve structural ambiguities in this compound?
Single-crystal X-ray diffraction (SCXRD) is routinely used to confirm stereochemistry and intermolecular interactions. For example, studies on derivatives like benzyl 2-{2,8-bis(trifluoromethyl)quinolin-4-ylmethyl}piperidine-1-carboxylate revealed dihedral angles between quinoline and substituent rings (e.g., 2.31° for coplanar systems) and hydrogen-bonding networks (C–H···F, O–H···N) that stabilize the crystal lattice . Software like SHELXL refines disordered atoms (e.g., trifluoromethyl groups) and validates occupancy ratios .
Q. What analytical techniques are essential for characterizing this compound’s purity and stereochemistry?
- HPLC-MS : Quantifies enantiomeric excess and detects impurities (e.g., diastereomers or hydrazone byproducts) .
- NMR : Distinguishes R/S configurations via coupling constants (e.g., J values for vicinal protons in piperidine) .
- Polarimetry : Measures optical rotation to confirm enantiomeric identity (e.g., (−)-(11S,2′R)-erythro-mefloquine vs. its (+)-counterpart) .
Advanced Research Questions
Q. How do structural modifications of this compound influence its antimalarial and antimicrobial activity?
Derivatization at the quinoline 4-position (e.g., hydrazone or organometallic substitutions) alters bioactivity. For instance:
- Hydrazone derivatives (e.g., 3,4-dimethoxybenzaldehyde hydrazone) exhibit enhanced π-π stacking and hydrogen bonding, improving solubility and target binding .
- Organometallic analogs (e.g., ferrocene- or ruthenocene-methanol hybrids) show reduced cytotoxicity compared to mefloquine but retain in vitro antischistosomal activity .
Structure-activity relationship (SAR) studies highlight the necessity of the trifluoromethyl groups for membrane permeability and the piperidine hydroxyl for target engagement .
Q. What computational methods are used to predict pharmacokinetic properties and toxicity?
- Molecular Dynamics (MD) : Simulates blood-brain barrier (BBB) penetration, leveraging the compound’s logP (~3.5) and topological polar surface area (45.2 Ų) .
- Density Functional Theory (DFT) : Models electronic interactions of trifluoromethyl groups with heme in Plasmodium parasites, explaining resistance mechanisms .
- ADMET Prediction : Tools like SwissADME assess metabolic stability (CYP450 interactions) and hepatotoxicity risks, critical for optimizing derivatives .
Q. How do crystallographic data resolve contradictions in reported stereochemical assignments?
Discrepancies in enantiomer activity (e.g., R,R vs. S,S forms) are resolved using:
- Hirshfeld Surface Analysis : Visualizes intermolecular contacts (e.g., F···H interactions) to validate crystal packing .
- ORTEP Diagrams : Annotate anisotropic displacement parameters and confirm chiral center configurations (e.g., Cahn-Ingold-Prelog priorities) .
For example, refinement of kryptoracemates (mixed enantiomers) confirmed non-covalent interactions dominate lattice stability over stereochemical homogeneity .
Q. Methodological Challenges and Solutions
Q. How are stability issues addressed during synthesis and storage?
- Temperature Control : Storage at 2–8°C prevents degradation of the labile hydroxyl group .
- Protecting Groups : Benzyl or carbamate groups stabilize the piperidine nitrogen during synthesis .
- Lyophilization : Maintains purity of hydrochloride salts by avoiding hydrate formation .
Q. What strategies mitigate cytotoxicity while retaining efficacy in derivatives?
Properties
IUPAC Name |
[2,8-bis(trifluoromethyl)quinolin-4-yl]-piperidin-2-ylmethanol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C17H16F6N2O/c18-16(19,20)11-5-3-4-9-10(15(26)12-6-1-2-7-24-12)8-13(17(21,22)23)25-14(9)11/h3-5,8,12,15,24,26H,1-2,6-7H2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XEEQGYMUWCZPDN-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CCNC(C1)C(C2=CC(=NC3=C2C=CC=C3C(F)(F)F)C(F)(F)F)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C17H16F6N2O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID50860636 | |
| Record name | [2,8-Bis(trifluoromethyl)quinolin-4-yl](piperidin-2-yl)methanol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID50860636 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
378.31 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Mefloquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014502 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Boiling Point |
415.7±40.0 °C | |
| Record name | Mefloquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00358 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Solubility |
3.80e-02 g/L | |
| Record name | Mefloquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00358 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Mefloquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014502 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Density |
Crystal density: 1.432 g/cu cm | |
| Record name | MEFLOQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6853 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
The mechanism of action of mefloquine is not completely understood. Some studies suggest that mefloquine specifically targets the 80S ribosome of the Plasmodium falciparum, inhibiting protein synthesis and causing subsequent schizonticidal effects. There are other studies in the literature with limited in vitro data on mefloquine's mechanism of action., Mefloquine, like chloroquine and quinine, is a blood schizonticidal agent and is active against the intraerythrocytic stages of parasite development. Similar to chloroquine and quinine, mefloquine appears to interfere with the parasite's ability to metabolize and utilize erythrocyte hemoglobin. The antimalarial activity of mefloquine may depend on the ability of the drug to form hydrogen bonds with cellular constituents; results of structure-activity studies indicate that the orientation of the hydroxyl and amine groups with respect to each other in the mefloquine molecule may be essential for antimalarial activity. While the precise mechanism of action of mefloquine is unknown, it may involve mechanisms that differ from those proposed for chloroquine., The effects of the antimalarial drug, mefloquine, on the uptake and release of Ca2+ by crude microsomes from dog brain were investigated using a spectrophotometric method. Mefloquine inhibited the inositol-1,4,5-phosphate (IP3)-induced Ca2+ release with an IC50 of 42 uM, but was a weaker inhibitor of the uptake of Ca2+ into the vesicles (IC50: 272 uM). These effects of mefloquine are in contrast to its actions on Ca2+ uptake and release by skeletal muscle microsomes, where its predominant effect was seen to be the inhibition of Ca2+ uptake into the vesicles. Mefloquine was found to be more potent than quinine as a specific inhibitor of Ca2+ release from IP3-sensitive stores in dog brain microsomes. The possibility of the drug affecting cellular IP3-linked signal transduction processes should be considered. | |
| Record name | Mefloquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00358 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | MEFLOQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6853 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
49752-90-1, 53230-10-7 | |
| Record name | α-(2-Piperidyl)-2,8-bis(trifluoromethyl)-4-quinolinemethanol | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=49752-90-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | alpha-2-Piperidyl-2,8-bis(trifluoromethyl)quinoline-4-methanol | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0049752901 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Mefloquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00358 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | α-2-piperidyl-2,8-bis(trifluoromethyl)quinoline-4-methanol | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.051.318 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | MEFLOQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6853 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Mefloquine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014502 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
250-254, 174-176 °C | |
| Record name | Mefloquine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00358 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | MEFLOQUINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/6853 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















