Praziquantel
Overview
Description
Praziquantel is a pyrazinoisoquinoline derivative and the first-line treatment for schistosomiasis, a parasitic disease affecting over 200 million people globally . Synthesized and administered as a racemic mixture, its active enantiomer (R-praziquantel) exhibits antischistosomal activity, while the inactive (S-enantiomer) contributes to adverse effects like dizziness and the drug’s bitter taste . This compound acts primarily by disrupting the tegument of adult schistosomes, leading to calcium influx, paralysis, and immune-mediated clearance . Despite widespread use for over four decades, emerging drug resistance and variable cure rates (CRs: 27.6–93.5% depending on parasite species and setting) underscore the need for improved alternatives .
Preparation Methods
Praziquantel can be synthesized through various methods. One approach involves using phenylethylamine as the starting material. The synthesis generally involves steps like acylation, amination, cyclization, hydrogenation, and acylation . Another method uses a flow-chemistry approach, which significantly reduces the production time from days to minutes and can be scaled up for industrial production . This method involves the use of phenylethylamine, chloroacetyl chloride, and cyclohexanecarbonyl chloride as key reagents .
Chemical Reactions Analysis
Praziquantel undergoes various chemical reactions, including:
Oxidation: This reaction can be carried out using oxidizing agents like potassium permanganate.
Reduction: Hydrogenation reactions are used to reduce intermediate compounds during synthesis.
Cyclization: Cyclization reactions are crucial in forming the tetrahydroisoquinoline scaffold.
Common reagents used in these reactions include chloroacetyl chloride, cyclohexanecarbonyl chloride, and phenylethylamine . The major products formed from these reactions are intermediates that eventually lead to the final this compound compound .
Scientific Research Applications
Treatment of Schistosomiasis
Overview
Praziquantel is the first-line treatment for schistosomiasis, a neglected tropical disease caused by trematode worms of the genus Schistosoma. It is effective against all Schistosoma species and is typically administered as a single oral dose.
Efficacy Data
A systematic review and meta-analysis conducted in Ethiopia highlighted the efficacy of this compound. The pooled cure rate for a single dose of 40 mg/kg was found to be 89.8% , with specific rates for S. mansoni at 89.2% and S. haematobium at 93.6% .
| Study Location | Cure Rate (%) | Schistosoma Species |
|---|---|---|
| Ethiopia | 89.8 | All species |
| Various locations | 73.6 - 100 | S. mansoni and S. haematobium |
Veterinary Applications
Use in Livestock
this compound is also utilized in veterinary medicine, particularly for treating parasitic infections in cattle and other livestock. Its safety profile and effectiveness make it a preferred choice in veterinary antiparasitic treatments .
Potential in Cancer Therapy
Recent studies have explored the potential of this compound as an adjunct therapy in cancer treatment. Research indicates that PZQ may enhance the immune response against tumors, possibly due to its immunomodulatory effects observed in schistosomiasis treatment .
Vaccine Adjuvant
This compound has been investigated for its role as a vaccine adjuvant, particularly in enhancing immune responses against viral infections . This application is still under research but shows promise in improving vaccine efficacy.
Side Effects and Safety Profile
While this compound is generally well-tolerated, some side effects include malaise, headache, dizziness, and nausea . Long-term studies have indicated no significant adverse effects on fertility or developmental outcomes during pregnancy .
Case Study 1: Efficacy in Ethiopia
A study conducted in Ethiopia assessed the effectiveness of this compound among infected individuals, revealing high cure rates and supporting its continued use in mass drug administration programs aimed at controlling schistosomiasis .
Case Study 2: Veterinary Use
In a controlled trial involving cattle, this compound demonstrated significant efficacy in reducing parasite load and improving overall health outcomes, underscoring its importance in veterinary medicine .
Mechanism of Action
The exact mechanism of action of praziquantel is not fully understood. it is believed to increase the permeability of the membranes of schistosome cells towards calcium ions, leading to muscle contraction and paralysis of the parasites . This results in the dislodgement and death of the parasites. This compound targets the β subunits of voltage-gated calcium channels, particularly in Schistosoma mansoni and Schistosoma haematobium .
Comparison with Similar Compounds
Comparison with Similar Compounds
Artemisinin Derivatives (e.g., Artemether, Dihydroartemisinin-Piperaquine)
- Mechanism : Artemisinin derivatives generate free radicals, damaging parasite membranes and proteins. Unlike praziquantel, they target both juvenile and adult schistosomes .
- Efficacy :
- In hamster models, artemether achieved 95–100% worm burden reduction against juvenile Schistosoma japonicum, outperforming this compound (36.6% at 100 mg/kg) .
- In human trials, combining this compound with dihydroartemisinin-piperaquine increased CRs from 54.7% (this compound alone) to 76.4% for intestinal schistosomiasis, demonstrating superior activity against immature worms .
- Safety: Adverse events (nausea, headache) were comparable between artemisinin-praziquantel combinations and this compound monotherapy .
Table 1: Comparative Efficacy of this compound and Artemisinin Derivatives
*ERR: Egg Reduction Rate
Benzothiazole Derivatives
- Novel benzothiazole-dithiocarbamate hybrids (e.g., compounds 4a-c) demonstrated 100% worm mortality at 10 μg/mL, matching this compound’s in vitro efficacy against S. mansoni . Structural optimization of these compounds may yield candidates with broader stage-specific activity.
Tetrahydroisoquinoline Analogs
- Compounds 3a and 3j, featuring a tetrahydroisoquinoline subunit akin to this compound, showed EC50 values of 1.8 μM and 1.5 μM against S. mansoni schistosomula, surpassing this compound’s EC50 (2.2 μM) . These analogs also induced tegumental damage similar to this compound but via undefined mechanisms .
Table 2: In Vitro Activity of this compound and Structural Analogs
| Compound | EC50 (μM) | Target Stage | Reference |
|---|---|---|---|
| This compound | 2.2 | Schistosomula/Adult | |
| Tetrahydroisoquinoline 3a | 1.8 | Schistosomula | |
| Tetrahydroisoquinoline 3j | 1.5 | Schistosomula |
Benzodiazepines (e.g., Meclonazepam)
- Meclonazepam, though less studied, exhibits antischistosomal activity through GABA receptor modulation, distinct from this compound’s calcium-dependent mechanism .
Natural Products (e.g., Hederacolchiside A1, Bergapten)
- Bergapten, a furanocoumarin, induced coiling and tegument damage in vitro, mimicking this compound’s effects .
Q & A
Basic Research Questions
Q. What experimental methods are used to investigate praziquantel’s mechanism of action against schistosomes?
- Methodological Answer: Studies employ calcium flux assays to measure drug-induced Ca²⁺ influx in schistosome teguments, a hallmark of this compound’s activity. RNA interference (RNAi) is used to knock down candidate targets like TRPMPZQ ion channels, followed by dose-response assays to assess changes in drug sensitivity. In vitro assays using adult worms or larval stages are paired with confocal microscopy to visualize tegumental disruption .
Q. How can researchers design clinical trials to evaluate this compound’s efficacy against Schistosoma mansoni?
- Methodological Answer: Trials should follow the PICOT framework:
- Population: School-aged children in endemic regions.
- Intervention: Single vs. repeated dosing (e.g., 40 mg/kg vs. 60 mg/kg).
- Comparison: Placebo or alternative anthelmintics (e.g., oxamniquine).
- Outcome: Parasitological cure rate (egg-negative conversion) at 1–3 months.
- Time: Longitudinal follow-up for reinfection rates.
Cluster-adjusted logistic regression models are recommended to account for within-school variability in infection intensity .
Q. What methodological approaches differentiate the pharmacological activity of this compound’s enantiomers?
- Methodological Answer: Chiral chromatography isolates (R)- and (S)-praziquantel for in vitro testing. Metabolic profiling via LC-MS identifies enantiomer-specific hepatic metabolism, while in vivo studies in rodents compare dose-response curves for efficacy (e.g., worm burden reduction) and toxicity (e.g., neurobehavioral effects). (S)-praziquantel shows negligible activity but contributes to adverse effects like bitter taste .
Advanced Research Questions
Q. How can genomic sequencing identify this compound resistance markers in Schistosoma mansoni?
- Methodological Answer: Whole-genome sequencing of field isolates identifies non-synonymous mutations in TRPMPZQ (e.g., p.Y1554C, p.Q1670K). Functional validation uses CRISPR/Cas9-edited parasites in Ca²⁺ reporter assays to quantify EC₅₀ shifts. Population genetics tools (e.g., integrated haplotype scores) detect selection pressures in endemic regions after mass drug administration (MDA) .
Q. What factors explain pharmacokinetic variability of this compound in pediatric populations?
- Methodological Answer: Population pharmacokinetic (PopPK) modeling incorporates covariates like hepatic CYP450 activity, body weight, and co-administered drugs (e.g., albendazole). Stable isotope-labeled (R)-praziquantel (e.g., deuterated D11) tracks enantiomer-specific metabolism. Pediatric formulations are optimized using dissolution testing to improve bioavailability and palatability .
Q. How do species-specific differences in schistosome biology impact this compound efficacy?
- Methodological Answer: Comparative studies using S. mansoni and S. haematobium assess tegumental protein expression (e.g., via proteomics) and drug uptake rates (radiolabeled this compound). Meta-analyses of clinical trial data reveal S. mansoni has higher cure rates with repeated dosing (89% egg reduction vs. 83% single dose), while S. haematobium shows minimal incremental benefit .
Q. What in vitro models evaluate synergistic combinations of this compound with next-generation anthelmintics?
- Methodological Answer: High-throughput screening assays pair this compound with thioredoxin-glutathione reductase inhibitors (e.g., auranofin). Isobologram analysis quantifies synergism (combination index <1). Larval motility assays and adult worm viability tests (ATP-based luminescence) validate combinations in co-culture systems .
Q. How should researchers address contradictory data on this compound’s dose-response relationships?
- Methodological Answer: Sensitivity analyses stratify data by confounding variables:
Properties
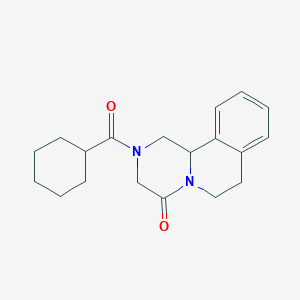
IUPAC Name |
2-(cyclohexanecarbonyl)-3,6,7,11b-tetrahydro-1H-pyrazino[2,1-a]isoquinolin-4-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C19H24N2O2/c22-18-13-20(19(23)15-7-2-1-3-8-15)12-17-16-9-5-4-6-14(16)10-11-21(17)18/h4-6,9,15,17H,1-3,7-8,10-13H2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FSVJFNAIGNNGKK-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CCC(CC1)C(=O)N2CC3C4=CC=CC=C4CCN3C(=O)C2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C19H24N2O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9021182 | |
| Record name | Praziquantel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9021182 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
312.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Praziquantel | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015191 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>46.9 [ug/mL] (The mean of the results at pH 7.4), 3.81e-01 g/L | |
| Record name | SID858012 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Praziquantel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01058 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Praziquantel | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015191 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Praziquantel works by causing severe spasms and paralysis of the worms' muscles. This paralysis is accompanied - and probably caused - by a rapid Ca 2+ influx inside the schistosome. Morphological alterations are another early effect of praziquantel. These morphological alterations are accompanied by an increased exposure of schistosome antigens at the parasite surface. The worms are then either completely destroyed in the intestine or passed in the stool. An interesting quirk of praziquantel is that it is relatively ineffective against juvenile schistosomes. While initially effective, effectiveness against schistosomes decreases until it reaches a minimum at 3-4 weeks. Effectiveness then increases again until it is once again fully effective at 6-7 weeks. Glutathione S-transferase (GST), an essential detoxification enzyme in parasitic helminths, is a major vaccine target and a drug target against schistosomiasis. Schistosome calcium ion channels are currently the only known target of praziquantel. | |
| Record name | Praziquantel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01058 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
55268-74-1 | |
| Record name | Praziquantel | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=55268-74-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Praziquantel [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0055268741 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Praziquantel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01058 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | praziquantel | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757285 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Praziquantel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9021182 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Praziquantel | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.054.126 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PRAZIQUANTEL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6490C9U457 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Praziquantel | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015191 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
136 °C | |
| Record name | Praziquantel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01058 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Praziquantel | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015191 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















