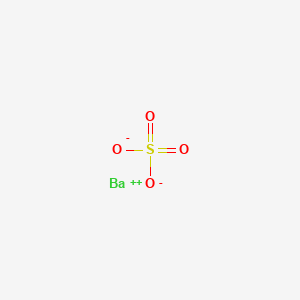
Barium sulfate
Overview
Description
Barium sulfate is an inorganic compound with the chemical formula BaSO₄. It is a white crystalline solid that is odorless and insoluble in water. This compound occurs naturally as the mineral barite, which is the primary commercial source of barium and its derivatives . This compound is known for its high density and opacity, making it useful in various industrial and medical applications .
Synthetic Routes and Reaction Conditions:
-
Using Barium Chloride and Sulfuric Acid:
-
Using Barium Sulfide and Sodium Sulfate:
Industrial Production Methods:
Direct Precipitation: This method involves the interaction of barium ions and sulfate ions in a liquid environment.
Types of Reactions:
Precipitation Reaction: this compound is formed as a precipitate when barium ions react with sulfate ions in an aqueous solution.
Common Reagents and Conditions:
Sulfuric Acid and Barium Chloride: Used in the precipitation reaction to form this compound.
Sodium Sulfate and Barium Sulfide: Alternative reagents for the precipitation of this compound.
Major Products:
Scientific Research Applications
Medical Applications
1.1. Contrast Agent in Radiology
Barium sulfate is primarily used as a contrast agent in medical imaging, particularly in gastrointestinal (GI) tract examinations. It enhances the visibility of the GI tract during X-ray imaging and fluoroscopy. The compound is ingested or administered rectally, allowing for detailed imaging of conditions such as obstructions, tumors, and ulcers.
- Case Study: A significant case involved a 47-year-old woman who experienced this compound aspiration during a barium swallow study. The aspiration led to massive deposits in her lungs, highlighting the potential risks associated with its use in diagnostic procedures .
1.2. Treatment of Intestinal Obstruction
this compound can also cause intestinal obstruction if not properly cleared from the body. A review of cases indicated that patients developed symptoms ranging from nausea to severe abdominal pain after undergoing barium examinations . In some instances, surgical intervention was required to remove residual barium.
Industrial Applications
2.1. Filler in Plastics and Paints
this compound is widely used as a filler in plastics and paints to enhance density and improve physical properties such as vibrational damping. Its high density makes it suitable for applications requiring mass damping.
- Data Table: Physical Properties of this compound Fillers
| Property | Value |
|---|---|
| Density | 4.5 g/cm³ |
| Particle Size | 0.5 - 10 μm |
| Reflectivity | High (90%+) |
2.2. Pigment in Coatings
In coatings, this compound serves as a pigment due to its high opacity and brightness. It is used in various applications including automotive paints and industrial coatings.
Environmental Applications
3.1. Soil Testing
this compound is employed in soil testing to clarify samples by binding with clay particles, thereby improving the accuracy of pH and nutrient assessments .
3.2. Wastewater Treatment
As an adsorbent, this compound can be utilized in wastewater treatment processes to remove heavy metals and other contaminants from water sources.
Emerging Research Applications
Recent studies have explored innovative uses for this compound nanoparticles in biomedical applications, such as drug delivery systems and as carriers for therapeutic agents due to their biocompatibility and stability .
Mechanism of Action
Barium sulfate acts as a radiographic contrast agent due to its high atomic number, which increases the absorption of X-rays. This property allows for the clear visualization of the gastrointestinal tract during diagnostic procedures . The compound is ingested orally or administered rectally, and it enhances the contrast of the images by delineating the structures of the GI tract .
Comparison with Similar Compounds
Calcium Sulfate (CaSO₄): Like barium sulfate, calcium sulfate is also used in medical imaging but has different sol
Biological Activity
Barium sulfate (BaSO₄) is a white crystalline solid that is widely used in various industrial applications, particularly in the medical field as a radiocontrast agent for imaging studies. Despite its common use, the biological activity of this compound, particularly its effects on human health and its interactions within biological systems, warrants detailed examination.
This compound is considered to be biologically inert , meaning it does not react chemically within the body under normal circumstances. Its primary use in medical imaging involves its ability to absorb X-rays, making it effective for visualizing the gastrointestinal tract. The compound's insolubility in water contributes to its low toxicity, as it does not readily dissolve and release barium ions into the bloodstream.
Inhalation Studies
Recent studies have evaluated the fate of this compound nanoparticles (BaSO₄ NPs) when inhaled. One study demonstrated that barium from BaSO₄ NPs was cleared from the lungs faster than other poorly soluble nanoparticles. The translocation of barium primarily occurred to bone tissues, with significant concentrations found in the lungs, lymph nodes, and bones after prolonged exposure .
Table 1: Barium Concentrations in Various Tissues Post-Exposure
| Tissue Type | Barium Concentration (µg/g) |
|---|---|
| Lungs | 285 (BaSO₄ NPs) |
| Lymph Nodes | Not specified |
| Hard Bone | Not specified |
| Bone Marrow | Not specified |
| Liver | Not specified |
Aspiration Cases
This compound aspiration can lead to severe respiratory complications. A comprehensive review of aspiration cases indicated that 75% of patients exhibited symptoms such as dyspnea and acute respiratory distress syndrome (ARDS). The mortality rate from this compound aspiration was reported to be around 36%, highlighting the potential risks associated with its use in diagnostic procedures .
Case Study: Aspiration Incident
- Patient Profile : A 47-year-old female underwent a barium swallow examination.
- Outcome : Post-examination, she experienced coughing and was found to have massive barium deposits in her lungs, leading to respiratory complications.
In Vitro Studies on Cellular Response
Research involving alveolar macrophages (NR8383 cells) exposed to various sizes of BaSO₄ particles indicated that these particles are taken up by cells but do not induce inflammatory or cytotoxic responses at concentrations up to 200 µg/mL. This suggests that while BaSO₄ can be internalized by cells, it does not trigger adverse biological responses under controlled conditions .
Table 2: Cellular Response to this compound Particle Sizes
| Particle Size (μm) | Uptake Efficiency | Inflammatory Response |
|---|---|---|
| 0.04 | High | None |
| 0.27 | High | None |
| 1.3 | Moderate | Necrosis at high concentration |
Clinical Implications
The clinical implications of this compound are significant, especially concerning its use as a contrast agent. While it is effective for imaging purposes, the potential for aspiration and subsequent respiratory complications necessitates careful consideration during administration.
Recommendations for Safe Use
- Patient Assessment : Evaluate risk factors such as dysphagia or prior gastrointestinal surgeries.
- Monitoring : Patients should be monitored closely during and after administration for signs of aspiration.
- Emergency Protocols : Establish protocols for immediate intervention in case of aspiration incidents.
Q & A
Basic Research Questions
Q. What experimental methods are most reliable for quantifying barium sulfate solubility in aqueous systems under varying conditions?
To measure solubility, use gravimetric analysis paired with inductively coupled plasma mass spectrometry (ICP-MS) to account for trace barium ions. Control temperature (e.g., 18°C vs. 25°C) and ionic strength (e.g., using NaCl to simulate saline environments). Ensure equilibrium is reached by continuous stirring for >24 hours and validate results against the solubility product constant (Ksp = 1.08×10⁻¹⁰ at 25°C) . For high-precision studies, employ synchrotron X-ray absorption spectroscopy to monitor dissolution/precipitation kinetics in real time.
Q. How can researchers optimize the synthesis of this compound nanoparticles for controlled size and morphology?
Utilize co-precipitation methods with surfactants (e.g., sodium dodecyl sulfate) to regulate nucleation. Adjust parameters:
- Barium chloride and sodium sulfate concentrations (0.1–0.5 M).
- Mixing rate (500–2000 rpm) to control shear forces.
- pH (4–10) to influence crystallinity. Characterize particles using dynamic light scattering (DLS) for size distribution and transmission electron microscopy (TEM) for morphology. Cross-validate with X-ray diffraction (XRD) to confirm crystallographic purity .
Q. What spectroscopic techniques are essential for confirming this compound purity in synthesized samples?
- XRD : Identify crystalline phases; compare peaks to JCPDS card 24-1034.
- Fourier-transform infrared spectroscopy (FTIR) : Detect organic contaminants (e.g., surfactant residues) via C-H stretching bands (2800–3000 cm⁻¹).
- Thermogravimetric analysis (TGA) : Assess thermal stability; pure BaSO₄ shows <1% mass loss up to 1000°C.
- Energy-dispersive X-ray spectroscopy (EDS) : Confirm elemental composition (Ba, S, O) .
Advanced Research Questions
Q. How can discrepancies in reported thermodynamic data for this compound (e.g., ΔG°f, solubility) be resolved in meta-analyses?
Conduct a systematic review using criteria:
- Source validation : Prioritize studies with detailed methodology (e.g., temperature control ±0.1°C, inert atmosphere to prevent oxidation).
- Data harmonization : Normalize values to standard conditions (25°C, 1 atm) using the Van’t Hoff equation.
- Outlier analysis : Apply Grubbs’ test to exclude statistically anomalous datasets. Publish findings with uncertainty intervals and recommend protocols for future studies (e.g., ISO 5725 for precision testing) .
Q. What strategies mitigate interference from this compound’s low solubility when studying its reactivity in geochemical or biological systems?
- Tracer techniques : Use ¹³³Ba-labeled BaSO₄ to track dissolution at sub-ppm levels via gamma spectroscopy.
- Surface modification : Functionalize BaSO₄ with carboxylate groups to enhance dispersibility in aqueous media.
- In situ microscopy : Employ atomic force microscopy (AFM) to observe surface reactions (e.g., phosphate adsorption) in simulated physiological fluids .
Q. How do polymorphic impurities (e.g., baryte vs. synthetic BaSO₄) affect catalytic performance in sulfate-radical advanced oxidation processes (SR-AOPs)?
Design experiments comparing natural baryte and lab-synthesized BaSO₄:
- Activity testing : Measure persulfate activation efficiency via radical quenching (e.g., using methanol for •OH and tert-butanol for SO₄•⁻).
- Surface analysis : Perform X-ray photoelectron spectroscopy (XPS) to quantify defect sites (e.g., oxygen vacancies).
- Kinetic modeling : Fit data to Langmuir-Hinshelwood mechanisms to isolate polymorph-specific rate constants .
Q. Methodological Guidance for Data Interpretation
Q. What statistical approaches are appropriate for analyzing heterogeneous dissolution rates of this compound in multiphase systems?
Apply mixed-effects models to account for batch-to-batch variability. Use Kolmogorov-Smirnov tests to compare particle size distributions. For time-series data, fit to Avrami-Erofeev equations to discern nucleation-controlled vs. diffusion-controlled dissolution .
Q. How should researchers address conflicting reports on this compound’s biocompatibility in medical imaging vs. toxicity in environmental contexts?
- Dose-response studies : Compare acute exposure (e.g., >500 mg/kg in rodents) vs. chronic low-dose leaching (e.g., <1 ppm in aquatic systems).
- Speciation analysis : Use geochemical modeling (PHREEQC) to predict bioavailability in soil/water matrices.
- In vitro assays : Test cytotoxicity (MTT assay) and inflammatory responses (IL-6/IL-8 ELISA) across cell lines .
Q. Tables for Key Data
Table 1. Comparison of Characterization Techniques for this compound
| Technique | Detection Limit | Key Output | Limitations |
|---|---|---|---|
| XRD | 5 wt% | Crystallite size, phase purity | Amorphous content not detected |
| BET Surface Area | 0.1 m²/g | Specific surface area | Pore size >2 nm required |
| ICP-MS | 0.1 ppb | Trace Ba²⁺ quantification | Requires acid digestion |
Properties
Key on ui mechanism of action |
Barium sulfate is a heavy metal with a high atomic number (Z=56) and a K shell binding energy (K-edge of 37.4 keV) very close to that of most diagnostic x-ray beams. Due to these characteristics, barium is an ideal medium for the absorption of x-rays. Barium sulfate is essentially not absorbed from the GI tract nor metabolized in the body. Barium sulfate is used to fill the gastrointestinal tract lumen or to coat the mucosal surface and is administered orally, rectally, or instilled into an enterostomy tube or catheter,. Barium sulfate enhances delineation of the GI tract. The barium suspension covers the mucosal surface of the GI tract, allowing its shape, distensibility, motion, integrity, continuity, location within the torso, relationship to other organs to be closely examined. Various abnormalities, such as benign or malignant tumors, ulcers, strictures, diverticula, inflammation or infection, altered motility, displacement and other pathology can thereby be identified,. At lower concentrations (higher dilution), barium enhances the conspicuity of the GI tract to differentiate the GI tract from various abdominal organs in computed tomography examinations (CT scans) of the abdomen. Improved delineation of the gastrointestinal tract lumen and mucosa may be reached by contrast provided by gas (by the addition of bicarbonate or gas-filled balloons) in addition to the barium. This is known as a _double-contrast procedure_. Osmotically active agents (for example, sorbitol) are also used to induce fluid accumulation and distension of the GI system to enhance visualization. |
|---|---|
CAS No. |
7727-43-7 |
Molecular Formula |
BaH2O4S |
Molecular Weight |
235.41 g/mol |
IUPAC Name |
barium(2+);sulfate |
InChI |
InChI=1S/Ba.H2O4S/c;1-5(2,3)4/h;(H2,1,2,3,4) |
InChI Key |
XUBKPYAWPSXPDZ-UHFFFAOYSA-N |
impurities |
Natural impurities are ferric oxide, silicon dioxide, and strontium sulfate. |
SMILES |
[O-]S(=O)(=O)[O-].[Ba+2] |
Canonical SMILES |
OS(=O)(=O)O.[Ba] |
boiling_point |
2912 °F at 760 mmHg (Decomposes) (NIOSH, 2024) decomposes 2912 °F (decomposes) 2912 °F (Decomposes) |
Color/Form |
Fine, heavy powder or polymorphous crystals White, orthorhombic crystals White or yellowish powder Opaque powde |
density |
4.25 to 4.5 (NIOSH, 2024) 4.49 g/cu cm 4.5 g/cm³ 4.25-4.5 |
melting_point |
2876 °F (NIOSH, 2024) 1580 °C 2876 °F |
Key on ui other cas no. |
7727-43-7 13462-86-7 |
physical_description |
Barium sulfate appears as white or yellowish odorless powder or small crystals. Mp: 1580 °C (with decomposition). Density: 4.25 -4.5 g cm-3. Insoluble in water, dilute acids, alcohol. Soluble in hot concentrated sulfuric acid. Used as a weighting mud in oil-drilling, in paints, paper coatings, linoleum, textiles, rubber. Administered internally ("barium cocktail") as a radio-opaque diagnostic aid. Pellets or Large Crystals; Dry Powder; NKRA; Water or Solvent Wet Solid; Other Solid; Dry Powder, Liquid; Liquid, Other Solid; Liquid White or yellowish, odorless powder; [NIOSH] Insoluble in water; [HSDB] ODOURLESS TASTELESS WHITE OR YELLOWISH CRYSTALS OR POWDER. White or yellowish, odorless powder. |
Pictograms |
Health Hazard |
solubility |
0.0002 % at 64 °F (NIOSH, 2024) Very slightly soluble in cold water SOLUBILITY IN WATER INCREASES CONSIDERABLY IN PRESENCE OF CHLORIDE & OTHER ANIONS 0.00031 g/100 g water at 20 °C; insol in ethanol Soluble in hot concentrated sulfuric acid; practically insoluble in dilute acids and alcohol Practically insoluble in organic solvents; very slightly soluble in alkalis and in solution of many salts Solubility in water: none (64 °F): 0.0002% |
Synonyms |
Barite Baritop Barium Sulfate Barium Sulfate (2:1) E Z CAT E-Z-CAT EZCAT Micropaque Oral Sulfate, Barium |
vapor_pressure |
0 mmHg (approx) (NIOSH, 2024) 0 mmHg (approx) |
Origin of Product |
United States |
Synthesis routes and methods I
Procedure details









Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details








Synthesis routes and methods IV
Procedure details










Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













