Saquinavir
Overview
Description
Saquinavir is an antiretroviral medication used in combination with other medications to treat or prevent HIV/AIDS. It belongs to the protease inhibitor class and works by blocking the HIV protease enzyme, which is essential for the virus to mature and replicate . This compound was the first protease inhibitor approved by the FDA in 1995 and is marketed under brand names such as Invirase and Fortovase .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Saquinavir involves multiple steps, starting with the preparation of key intermediates. One method involves the methyl sulfonylation of (1S, 2S)-(1-benzyl-3-chloro-2-hydroxypropyl) tert-butyl carbamate using methylsulfonyl chloride in the presence of triethylamine and methylbenzene under a nitrogen atmosphere. This intermediate is then reacted with metallic acetate in the presence of 18-crown ether-6 and methylbenzene to obtain (1S, 2R)-(1-benzyl-3-chloro-2-acetic acid propyl) tert-butyl carbamate. Further reactions with KOH, THF, and ethanol yield (2R, 3S)-1,2-epoxy-3-tert-butyloxycarboryl amino-4-phenyl butane .
Industrial Production Methods: Industrial production of this compound typically involves large-scale synthesis using optimized reaction conditions to ensure high yield and purity. The process includes the use of advanced techniques such as differential scanning calorimetry (DSC), powder X-ray diffraction (PXRD), and Fourier transform infrared spectroscopy (FT-IR) for characterization and quality control .
Chemical Reactions Analysis
Types of Reactions: Saquinavir undergoes various chemical reactions, including oxidation, reduction, and substitution. These reactions are crucial for its metabolism and elimination from the body.
Common Reagents and Conditions:
Oxidation: this compound can be oxidized using reagents such as hydrogen peroxide or potassium permanganate.
Reduction: Reduction reactions may involve agents like sodium borohydride or lithium aluminum hydride.
Substitution: Substitution reactions often use halogenating agents like thionyl chloride or phosphorus tribromide.
Major Products: The major products formed from these reactions include various metabolites that are excreted primarily through feces and urine .
Scientific Research Applications
HIV Treatment
Saquinavir has revolutionized the treatment of HIV/AIDS since its approval in 1995. It is primarily used in combination with other antiretroviral agents as part of Highly Active Antiretroviral Therapy (HAART). Its mechanism involves inhibiting the HIV protease enzyme, which is crucial for viral replication.
Clinical Efficacy:
- This compound has shown significant viral load reduction in patients when used alongside nucleoside reverse transcriptase inhibitors (NRTIs) and non-nucleoside reverse transcriptase inhibitors (NNRTIs) .
Cancer Treatment
Recent studies have indicated that this compound may serve as an effective treatment for various cancers, particularly anal cancer and cervical cancer.
Anal Cancer
A study highlighted this compound's ability to reduce tumor spheroid growth in a transgenic mouse model of anal cancer associated with HPV. The results showed a statistically significant reduction in tumor growth compared to control groups, suggesting its potential as an adjuvant therapy alongside existing protocols .
Cervical Cancer
This compound has also been investigated for its effects on cervical cancer cells. It demonstrated inhibitory effects on cell proliferation and invasion, indicating potential pathways for therapeutic intervention .
COVID-19 Treatment
This compound's antiviral properties have prompted research into its effectiveness against SARS-CoV-2. Molecular docking studies revealed that this compound can bind to the 3CLpro enzyme of the virus, inhibiting its activity. This suggests a possible role in treating COVID-19, especially given its low IC50 value of approximately 9.92 μM .
Tuberculosis Therapy
Emerging evidence supports this compound's use as a host-directed therapy for tuberculosis (TB). Research indicates that this compound enhances the intracellular killing of Mycobacterium tuberculosis (Mtb) in infected macrophages while improving antigen presentation and T-cell responses . This dual action positions it as a promising adjunct treatment for TB.
Microbicide Development
This compound has been explored as a candidate microbicide to prevent HIV transmission. Studies demonstrated its ability to inhibit early events associated with HIV infection in various cell types, including CD4 T cells and macrophages. This positions this compound as a potential preventive measure against HIV in at-risk populations .
Summary of Applications
Mechanism of Action
Saquinavir exerts its antiviral activity by inhibiting the HIV-1 protease enzyme. This enzyme is crucial for the cleavage of polyproteins into functional viral proteins. By blocking this enzyme, this compound prevents the maturation of the virus, leading to the formation of non-infectious viral particles . The primary molecular target is the HIV-1 protease, and the pathway involves the disruption of viral replication and assembly .
Comparison with Similar Compounds
Ritonavir: Another protease inhibitor used in combination with other antiretrovirals. It is known for its ability to boost the effectiveness of other protease inhibitors by inhibiting their metabolism.
Indinavir: A protease inhibitor that also targets the HIV-1 protease but has different pharmacokinetic properties.
Lopinavir: Often used in combination with Ritonavir, it has a similar mechanism of action but different resistance profiles.
Uniqueness of Saquinavir: this compound was the first protease inhibitor to be approved and has a relatively benign adverse effect profile compared to other antiretroviral therapies . Its unique structure and binding affinity to the HIV-1 protease make it a valuable component of combination therapies for HIV/AIDS .
Biological Activity
Saquinavir is a protease inhibitor that has been pivotal in the treatment of HIV/AIDS since its introduction. Its biological activity extends beyond antiviral effects, showing potential applications in cancer therapy and other diseases. This article provides a comprehensive overview of the biological activity of this compound, highlighting its mechanisms, efficacy, and case studies.
This compound functions primarily by inhibiting the HIV-1 protease enzyme, which is crucial for the maturation of viral proteins. By preventing this cleavage, this compound effectively reduces viral replication. The compound's limited bioavailability (approximately 4%) necessitates co-administration with ritonavir to enhance its serum concentration and therapeutic efficacy .
Antiviral Activity
- Inhibition of HIV Replication : this compound demonstrates dose-dependent inhibition of HIV-1 replication across various cell types, including CD4 T cells and macrophages . It significantly reduces viral load and improves immune response markers such as HLA class II expression and IFN-γ secretion in infected macrophages .
- Resistance Patterns : Clinical studies indicate that patients with prior exposure to this compound may develop resistance, complicating subsequent treatment with other protease inhibitors like nelfinavir .
Anticancer Potential
Recent studies have explored this compound's role in cancer treatment, particularly in cervical cancer. This compound has been shown to inhibit cell proliferation, invasion, and clonogenicity in HeLa cells at concentrations lower than those required to affect proteasomal activity . This suggests that this compound may exert its effects through alternative oncogenic pathways.
Lipid Metabolism Effects
This compound also impacts lipid metabolism significantly. Research indicates that it inhibits lipoprotein lipase (LPL) activity and lipid synthesis in adipocytes, which may contribute to metabolic complications observed in patients undergoing protease inhibitor therapy . Notably, after removal of this compound, LPL activity partially recovers, indicating a reversible effect on lipid metabolism.
Table 1: Summary of Key Research Findings on this compound
Clinical Implications
A randomized study evaluated the safety and efficacy of a regimen including this compound, ritonavir, and atazanavir over 48 weeks. The results indicated a modest increase in CD4 lymphocyte counts but highlighted adverse effects such as scleral icterus in some patients . These findings underscore the need for careful monitoring during treatment.
Q & A
Q. Basic Research: What are the primary metabolic pathways of saquinavir, and how do they influence experimental design in pharmacokinetic studies?
This compound is predominantly metabolized by hepatic CYP3A4, with >90% biotransformation occurring via this enzyme . Its first-pass metabolism results in low oral bioavailability (~4%), which is significantly enhanced by co-administration with ritonavir (a CYP3A4 inhibitor) or high-fat meals .
Methodological Guidance :
- Use in vitro liver microsome assays to quantify CYP3A4-mediated metabolism.
- Conduct pharmacokinetic studies in animal/human models with controlled diets (e.g., high-fat vs. fasting) to assess food effects .
- Include CYP3A4 inhibitors/inducers (e.g., ketoconazole, rifampicin) to validate metabolic pathways.
Q. Basic Research: How does this compound interact with P-glycoprotein (P-gp), and what are the implications for cellular uptake studies?
This compound is a substrate of P-gp, an efflux transporter localized on apical surfaces of hepatocytes, renal proximal tubules, and intestinal epithelia . This interaction limits its intracellular accumulation and contributes to variable tissue penetration.
Methodological Guidance :
- Use polarized cell lines (e.g., Caco-2 monolayers) to measure bidirectional transport and P-gp inhibition/induction.
- Quantify intracellular this compound levels via LC-MS/MS in the presence/absence of P-gp inhibitors (e.g., verapamil) .
Q. Advanced Research: How can researchers resolve contradictory data on this compound’s off-target inhibition of cathepsin L versus its lack of effect on proteasome activity in certain models?
This compound inhibits cathepsin L (IC50: 13.4 µM) but shows negligible proteasome inhibition at clinically relevant concentrations (<30 µM) . Discrepancies arise from dose-dependent effects and assay conditions (e.g., exposure time, cell type).
Methodological Guidance :
- Standardize assays using recombinant enzymes (e.g., cathepsin L) and fluorescent substrates to measure inhibition kinetics .
- For proteasome activity, employ cell-based assays with prolonged exposure (≥24 hrs) and higher doses (≥60 µM) to detect modulation .
- Apply statistical tools like two-way ANOVA with Bonferroni correction to compare dose-response curves across cell lines .
Q. Advanced Research: What experimental strategies are recommended to investigate this compound’s potential repurposing for SARS-CoV-2 or cancer therapy?
This compound inhibits SARS-CoV-2 spike protein-mediated cell fusion via cathepsin L blockade and reduces cervical cancer cell proliferation/invasion independently of proteasome modulation .
Methodological Guidance :
- Use pseudovirus entry assays with TMPRSS2/ACE2-expressing cells to quantify SARS-CoV-2 inhibition .
- For cancer studies, perform clonogenic assays and 3D invasion models (e.g., Matrigel) with primary cervical cancer cell lines .
- Validate findings in vivo using xenograft models, monitoring this compound tissue penetration via HPLC .
Q. Advanced Research: How can structural biology elucidate this compound’s resistance profile in HIV protease mutants?
This compound loses efficacy against HIV protease mutants (e.g., G48V) due to disrupted hydrogen bonding, whereas darunavir retains activity . X-ray crystallography reveals differential binding interactions.
Methodological Guidance :
- Solve co-crystal structures of this compound with mutant proteases (e.g., G48V, PRI50V) to map residue-specific interactions .
- Combine molecular dynamics simulations with enzymatic inhibition assays (e.g., fluorescence-based protease activity) to correlate structural changes with resistance .
Q. Advanced Research: What factorial design approaches optimize this compound delivery systems (e.g., transdermal nanoemulsions)?
Self-nanoemulsifying drug delivery systems (SNEDDS) improve this compound bioavailability by enhancing solubility and bypassing first-pass metabolism .
Methodological Guidance :
- Apply Box-Behnken or central composite designs to optimize SNEDDS variables (e.g., oil phase ratio, surfactant concentration) .
- Characterize ex vivo skin permeation using Franz diffusion cells and confocal microscopy for tissue distribution analysis .
Q. Basic Research: How do drug-drug interactions (DDIs) between this compound and CYP3A4 modulators affect experimental outcomes?
This compound’s AUC increases 177% when co-administered with clarithromycin (a CYP3A4 inhibitor), while CYP3A4 inducers (e.g., rifampicin) reduce exposure .
Methodological Guidance :
Properties
IUPAC Name |
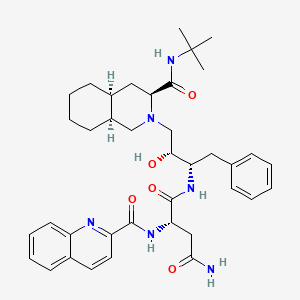
(2S)-N-[(2S,3R)-4-[(3S,4aS,8aS)-3-(tert-butylcarbamoyl)-3,4,4a,5,6,7,8,8a-octahydro-1H-isoquinolin-2-yl]-3-hydroxy-1-phenylbutan-2-yl]-2-(quinoline-2-carbonylamino)butanediamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C38H50N6O5/c1-38(2,3)43-37(49)32-20-26-14-7-8-15-27(26)22-44(32)23-33(45)30(19-24-11-5-4-6-12-24)41-36(48)31(21-34(39)46)42-35(47)29-18-17-25-13-9-10-16-28(25)40-29/h4-6,9-13,16-18,26-27,30-33,45H,7-8,14-15,19-23H2,1-3H3,(H2,39,46)(H,41,48)(H,42,47)(H,43,49)/t26-,27+,30-,31-,32-,33+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
QWAXKHKRTORLEM-UGJKXSETSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)(C)NC(=O)C1CC2CCCCC2CN1CC(C(CC3=CC=CC=C3)NC(=O)C(CC(=O)N)NC(=O)C4=NC5=CC=CC=C5C=C4)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)(C)NC(=O)[C@@H]1C[C@@H]2CCCC[C@@H]2CN1C[C@H]([C@H](CC3=CC=CC=C3)NC(=O)[C@H](CC(=O)N)NC(=O)C4=NC5=CC=CC=C5C=C4)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C38H50N6O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
149845-06-7 (monomethanesulfonate (salt)) | |
| Record name | Saquinavir [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0127779208 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID6044012 | |
| Record name | Saquinavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6044012 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
670.8 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Insoluble, In water, 0.22 g/100 mL @ 25 °C | |
| Record name | Saquinavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01232 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | SAQUINAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7161 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Vapor Pressure |
2X10-31 mm Hg @ 25 °C /Estimated/ | |
| Record name | SAQUINAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7161 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
The HIV lifecycle is comprised of 3 distinct stages: assembly, involving creation and packaging of essential viral components; budding, wherein the viral particle crosses the host cell plasma membrane and forms a lipid envelope; and maturation, wherein the viral particle alters its structure and becomes infectious. At the center of this lifecycle is the Gag polyprotein which, along with the products of its proteolysis, coordinate these stages and function as the major structural proteins of the virus. The HIV-1 protease enzyme, a dimeric aspartic protease, is the enzyme responsible for cleaving the Gag polyprotein and thus plays a critical role in many aspects of the HIV viral lifecycle. Saquinavir is an inhibitor of the HIV-1 protease enzyme. Its design is based on the "peptidomimetic" principle, wherein the molecule contains a hydroxyethylene scaffold that mimics the normal peptide linkage (cleaved by HIV protease) but which itself cannot be cleaved. By preventing HIV-1 protease activity, and thus the proteolysis of the Gag polyprotein, saquinavir results in the production of immature, non-infectious viral particles., While the complete mechanisms of antiviral activity of saquinavir have not been fully elucidated, saquinavir apparently inhibits replication of retroviruses, including human immunodeficiency virus type 1 (HIV-1) and type 2 (HIV-2), by interfering with HIV protease. The drug, therefore, exerts a virustatic effect against retroviruses by acting as an HIV protease inhibitor., Saquinavir is a selective, competitive, reversible inhibitor of HIV protease. HIV protease, an aspartic endopeptidase that functions as a homodimer, plays an essential role in the replication cycle of HIV and the formation of infectious virus. During HIV replication, HIV protease cleaves viral polypeptide products of the gag and gag-pol genes (i.e., p55 and p160) to form structural proteins of the virion core (i.e., p17, p24, p9, and p7) and essential viral enzymes (i.e., reverse transcriptase, integrase, and protease). Because saquinavir is a structural analog of the HIV Phe-Pro protease cleavage site, the drug inhibits the function of the enzyme. By interfering with the formation of these essential proteins and enzymes, saquinavir blocks maturation of the virus and causes the formation of nonfunctional, immature, noninfectious virions. Saquinavir is active in both acutely and chronically infected cells since it targets the HIV replication cycle after translation and before assembly. Thus, the drug is active in chronically infected cells (e.g., monocytes and macrophages) that generally are not affected by nucleoside reverse transcriptase inhibitors (e.g., didanosine, lamivudine, stavudine, zalcitabine, zidovudine). Saquinavir does not affect early stages of the HIV replication cycle; however, the drug interferes with the production of infectious HIV and limits further infectious spread of the virus., Unlike nucleoside antiretroviral agents, the antiviral activity of saquinavir does not depend on intracellular conversion to an active metabolite. Saquinavir and other HIV protease inhibitors (e.g., amprenavir, indinavir, lopinavir, nelfinavir, ritonavir) act at a different stage of the HIV replication cycle than nucleoside and nonnucleoside reverse transcriptase inhibitors, and results of in vitro studies indicate that the antiretroviral effects of some nucleoside reverse transcriptase inhibitors and HIV protease inhibitors may be additive or synergistic. | |
| Record name | Saquinavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01232 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | SAQUINAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7161 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White crystalline solid, Off-white to white very fine powder | |
CAS No. |
127779-20-8 | |
| Record name | Saquinavir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=127779-20-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Saquinavir [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0127779208 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Saquinavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01232 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Saquinavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6044012 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | SAQUINAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/L3JE09KZ2F | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | SAQUINAVIR | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7161 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















