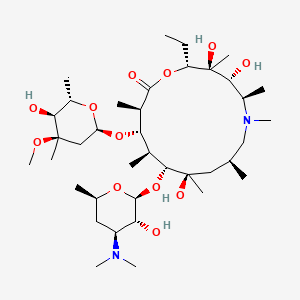
Azithromycin
Overview
Description
Azithromycin is a broad-spectrum macrolide antibiotic used to treat a variety of bacterial infections. It was discovered in the 1980s by the pharmaceutical company Pliva in Croatia and approved for medical use in 1988 . This compound is known for its long half-life and high tissue penetration, making it effective against respiratory, enteric, and genitourinary infections .
Preparation Methods
Synthetic Routes and Reaction Conditions: Azithromycin is synthesized from erythromycin A through a series of chemical reactions. The process involves the conversion of erythromycin A into its oxime, followed by Beckmann rearrangement to form the amino ether of erythromycin A. This intermediate is then reduced to 9-deoxo-9a-aza-9a-homoerythromycin, and finally, reductive N-methylation yields this compound .
Industrial Production Methods: Industrial production of this compound often involves hot-melt extrusion to prepare this compound amorphous solid dispersion. This method improves the solubility and taste-masking properties of the drug. The optimal extrusion parameters include a temperature of 150°C, a screw speed of 75 rpm, and a drug percentage of 25% .
Chemical Reactions Analysis
Types of Reactions: Azithromycin undergoes various chemical reactions, including oxidation, reduction, and substitution. For example, during oxidation, the bromine ion attaches to the lone pair electron of this compound, forming a yellow coupling product .
Common Reagents and Conditions: Common reagents used in the reactions of this compound include bromine ions for oxidation and various solvents and surfactants for nanoparticle preparation .
Major Products: The major products formed from these reactions include this compound nanoparticles and amorphous solid dispersions, which enhance the drug’s solubility and bioavailability .
Scientific Research Applications
Antimicrobial Applications
Azithromycin is primarily recognized for its effectiveness against a range of bacterial pathogens. It is particularly noted for its activity against both gram-positive and gram-negative bacteria, as well as atypical pathogens.
FDA-Approved Indications
- Community-Acquired Pneumonia : Effective against Streptococcus pneumoniae, Haemophilus influenzae, and Moraxella catarrhalis.
- Upper Respiratory Infections : Used for acute otitis media and exacerbations of chronic obstructive pulmonary disease.
- Skin Infections : Treats infections caused by Streptococcus pyogenes, Staphylococcus aureus, and others.
- Sexually Transmitted Infections : Effective for urethritis and cervicitis caused by Chlamydia trachomatis and Neisseria gonorrhoeae.
- Mycobacterial Infections : Used in treating Mycobacterium avium complex infections in advanced AIDS patients.
Off-Label Uses
- Asthma Management : A study indicated that this compound may induce remission in patients with persistent uncontrolled asthma, suggesting its potential role in respiratory therapies .
- Salmonella Typhi Infections : this compound has been used in treating enteric fever caused by this pathogen .
Immunomodulatory Effects
Recent research has highlighted this compound's immunomodulatory properties, which extend its utility beyond mere antibacterial action.
Public Health Impact
One of the most significant applications of this compound has been in public health initiatives aimed at controlling infectious diseases.
Trachoma Elimination
Research led by the London School of Hygiene & Tropical Medicine demonstrated that mass treatment with this compound can effectively eliminate trachoma, a leading cause of blindness. Over 900 million doses have been donated globally, significantly reducing the number of people at risk from 2002 to 2020 .
Case Studies and Observational Studies
Several studies have examined the efficacy of this compound in various clinical settings:
COVID-19 Treatment Trials
An observational study involving hospitalized COVID-19 patients found no significant clinical benefit from this compound when used alone, although it was part of combination therapies . Another trial assessed its effectiveness for community treatment but did not demonstrate substantial advantages over usual care .
Case Study Example
A case study highlighted a 45-year-old female patient treated with this compound for a purulent cough and fever, illustrating its application in acute respiratory infections .
Table 1: FDA-Approved Indications for this compound
| Indication | Pathogen(s) Involved |
|---|---|
| Community-Acquired Pneumonia | Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis |
| Upper Respiratory Infections | Various pathogens including Streptococcus pyogenes |
| Skin Infections | Staphylococcus aureus, Streptococcus agalactiae |
| Urethritis/Cervicitis | Chlamydia trachomatis, Neisseria gonorrhoeae |
| Mycobacterial Infections | Mycobacterium avium complex |
Table 2: Observational Study Characteristics
| Characteristic | This compound Group (n=239) | Control Group (n=179) | p-value |
|---|---|---|---|
| Mean Age (years) | 65.4 | 64.9 | 0.320 |
| Hospital Stay (days) | 10.0 | 6.7 | 0.025 |
| Mortality Rate (%) | 18.9 | N/A | N/A |
Mechanism of Action
Azithromycin is structurally related to erythromycin and clarithromycin, both of which are also macrolide antibiotics. this compound has a 15-membered ring with a methyl-substituted nitrogen, which differentiates it from erythromycin’s 14-membered lactone ring . This structural difference gives this compound a longer half-life and better tissue penetration compared to erythromycin . Other similar compounds include clarithromycin and roxithromycin, which share similar mechanisms of action but differ in their pharmacokinetic properties .
Comparison with Similar Compounds
- Erythromycin
- Clarithromycin
- Roxithromycin
Azithromycin’s unique structure and properties make it a valuable antibiotic with broad applications in medicine and research.
Q & A
Basic Research Questions
Q. What are the standard methodologies for assessing azithromycin resistance in bacterial pathogens, and how have these evolved with genomic sequencing technologies?
- Methodological Answer : Traditional methods include in vitro susceptibility testing (e.g., minimum inhibitory concentration assays) and phenotypic screening. However, whole genome sequencing (WGS) has revolutionized resistance tracking by identifying genetic markers, such as mutations in the 23S rRNA gene or efflux pump regulators. For example, CDC studies used WGS to link reduced this compound susceptibility in Neisseria gonorrhoeae to specific mutations (e.g., mtrR promoter variants) and horizontally acquired resistance genes like erm . Recent protocols recommend combining WGS with antimicrobial susceptibility data to monitor resistance trends and inform treatment guidelines .
Q. How should researchers design mass drug administration (MDA) studies to evaluate this compound’s impact on infectious disease burden in resource-limited settings?
- Methodological Answer : Systematic reviews emphasize using standardized search strategies (e.g., combining keywords like "this compound," "mass drug administration," and "MDA" across PubMed, Embase, and clinical trial registries) to identify relevant studies . Trials should specify dosing regimens (e.g., single 20 mg/kg dose vs. multi-day courses), target populations (e.g., pregnant women, children), and co-administered drugs. Harmonized data collection on adverse events, resistance emergence, and coverage rates is critical for cross-study comparisons .
Q. What are the best practices for monitoring this compound’s impact on maternal and neonatal sepsis in clinical trials?
- Methodological Answer : Trials like SANTE and A-PLUS employ factorial designs to randomize participants to antenatal/intrapartum this compound or placebo, with predefined primary endpoints (e.g., sepsis incidence, mortality). Secondary objectives should include microbiome analysis (e.g., 16S rRNA sequencing) to assess dysbiosis risks and resistance gene quantification via PCR or metagenomics . Standardized case definitions for sepsis (e.g., WHO criteria) and blinded endpoint adjudication reduce bias .
Advanced Research Questions
Q. How can researchers reconcile contradictory findings on this compound’s efficacy in reducing mortality across trials in low- and middle-income countries (LMICs)?
- Methodological Answer : Conflicting results (e.g., reduced mortality in some trials vs. null effects in others) may arise from population heterogeneity, background antibiotic use, or resistance profiles. Meta-analyses should stratify by contextual factors (e.g., malaria endemicity, baseline sepsis rates) and use individual participant data (IPD) to adjust for confounders. Prospective harmonization of endpoints, as seen in the PregnAnZI-2 and SANTE trials, improves comparability . Sensitivity analyses can explore whether mortality reductions are driven by direct antimicrobial effects or immunomodulatory properties .
Q. What genomic and bioinformatic approaches are most effective for tracking this compound resistance evolution in Campylobacter and Shigella species?
- Methodological Answer : WGS paired with phylogenetic analysis can identify clonal spread of resistant strains (e.g., Campylobacter with ermB genes) and distinguish acquired resistance from spontaneous mutations. For Shigella, plasmid sequencing (e.g., identifying mobile elements carrying mphA or mef genes) clarifies horizontal gene transfer dynamics. CDC’s GISP program uses such methods to detect resistance clusters and inform empiric treatment guidelines .
Q. How should researchers optimize this compound dosing regimens in pregnant women to balance efficacy with microbiome disruption risks?
- Methodological Answer : Pharmacokinetic/pharmacodynamic (PK/PD) modeling can determine optimal dosing by correlating maternal plasma levels with fetal tissue penetration and bacterial load reduction. Ancillary microbiome studies (e.g., longitudinal stool sampling with shotgun metagenomics) quantify taxonomic shifts (e.g., reduced Bifidobacterium) and functional gene loss. Trials should prespecify stopping rules for significant dysbiosis or resistance gene enrichment .
Q. Data Contradiction and Synthesis
Q. What statistical methods are recommended for analyzing discordant this compound trial data, particularly when mortality outcomes conflict?
- Methodological Answer : Bayesian hierarchical models can account for between-study heterogeneity, while IPD meta-analyses explore subgroup effects (e.g., greater mortality reduction in high-malaria settings). Frailty models adjust for unmeasured covariates, and trial sequential analysis (TSA) evaluates whether cumulative evidence meets futility/efficacy thresholds. Sensitivity analyses excluding outliers (e.g., trials with high attrition bias) enhance robustness .
Q. How can researchers address the trade-off between this compound’s short-term benefits in MDA programs and long-term resistance risks?
- Methodological Answer : Dynamic transmission models incorporating resistance gene fitness costs and horizontal transfer rates can project resistance trajectories under different MDA scenarios. Paired with empirical surveillance (e.g., annual WGS of pathogen isolates), these models inform optimal dosing frequency and geographic targeting. The WHO recommends staggered MDA rollouts with resistance monitoring as a precondition for scale-up .
Q. Tables for Key Data Trends
Properties
Key on ui mechanism of action |
Azithromycin usually is bacteriostatic, although the drug may be bactericidal in high concentrations against selected organisms. Bactericidal activity has been observed in vitro against Streptococcus pyogenes, S. pneumoniae, and Haemophilus influenzae. Azithromycin inhibits protein synthesis in susceptible organisms by penetrating the cell wall and binding to 50S ribosomal subunits, thereby inhibiting translocation of aminoacyl transfer-RNA and inhibiting polypeptide synthesis. The site of action of azithromycin appears to be the same as that of the macrolides (i.e., erythromycin, clarithromycin), clindamycin, lincomycin, and chloramphenicol. The antimicrobial activity of azithromycin is reduced at low pH. Azithromycin concentrates in phagocytes, including polymorphonuclear leukocytes, monocytes, macrophages, and fibroblasts. Penetration of the drug into phagocytic cells is necessary for activity against intracellular pathogens (e.g., Staphylococcus aureus, Legionella pneumophila, Chlamydia trachomatis, Salmonella typhi). |
|---|---|
CAS No. |
83905-01-5 |
Molecular Formula |
C38H72N2O12 |
Molecular Weight |
749.0 g/mol |
IUPAC Name |
(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-11-[4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-2-ethyl-3,4,10-trihydroxy-13-(5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl)oxy-3,5,6,8,10,12,14-heptamethyl-1-oxa-6-azacyclopentadecan-15-one |
InChI |
InChI=1S/C38H72N2O12/c1-15-27-38(10,46)31(42)24(6)40(13)19-20(2)17-36(8,45)33(52-35-29(41)26(39(11)12)16-21(3)48-35)22(4)30(23(5)34(44)50-27)51-28-18-37(9,47-14)32(43)25(7)49-28/h20-33,35,41-43,45-46H,15-19H2,1-14H3/t20-,21?,22+,23-,24-,25?,26?,27-,28?,29?,30+,31-,32?,33-,35?,36-,37?,38-/m1/s1 |
InChI Key |
MQTOSJVFKKJCRP-FHZDSTMTSA-N |
SMILES |
CCC1C(C(C(N(CC(CC(C(C(C(C(C(=O)O1)C)OC2CC(C(C(O2)C)O)(C)OC)C)OC3C(C(CC(O3)C)N(C)C)O)(C)O)C)C)C)O)(C)O |
Isomeric SMILES |
CC[C@@H]1[C@@]([C@@H]([C@H](N(C[C@@H](C[C@@]([C@@H]([C@H]([C@@H]([C@H](C(=O)O1)C)OC2CC(C(C(O2)C)O)(C)OC)C)OC3C(C(CC(O3)C)N(C)C)O)(C)O)C)C)C)O)(C)O |
Canonical SMILES |
CCC1C(C(C(N(CC(CC(C(C(C(C(C(=O)O1)C)OC2CC(C(C(O2)C)O)(C)OC)C)OC3C(C(CC(O3)C)N(C)C)O)(C)O)C)C)C)O)(C)O |
Appearance |
Solid powder |
Color/Form |
Amorphous solid |
melting_point |
113-115 °C White crystalline powder. mp: 126 °C. Optical rotation: -41.4 deg at 26 °C/D (c = 1 in CHCl3) /Azithromycin dihydrate/ |
Key on ui other cas no. |
83905-01-5 |
physical_description |
Solid |
Pictograms |
Irritant; Health Hazard; Environmental Hazard |
Purity |
>98% (or refer to the Certificate of Analysis) |
Related CAS |
117772-70-0 (dihydrate) 121470-24-4 (monohydrate) |
shelf_life |
>2 years if stored properly |
solubility |
soluble in ethanol and DSMO, minimally soluble in water 5.14e-01 g/L |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
Azadose Azithromycin Azithromycin Dihydrate Azithromycin Monohydrate Azitrocin Azythromycin CP 62993 CP-62993 CP62993 Dihydrate, Azithromycin Goxal Monohydrate, Azithromycin Sumamed Toraseptol Ultreon Vinzam Zentavion Zithromax Zitromax |
vapor_pressure |
2.65X10-24 mm Hg at 25 °C (est) |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















