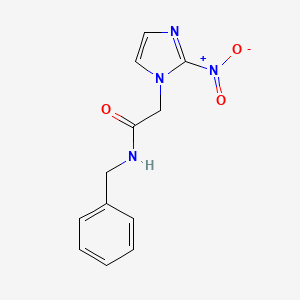
Benznidazole
Overview
Description
Benznidazole is an antiparasitic medication primarily used for the treatment of Chagas disease, which is caused by the protozoan parasite Trypanosoma cruzi . It belongs to the nitroimidazole class of compounds and works by producing free radicals that damage the DNA and cellular machinery of the parasite . This compound is considered the first-line treatment for Chagas disease due to its moderate side effects compared to other treatments .
Preparation Methods
Synthetic Routes and Reaction Conditions: Benznidazole can be synthesized through a multi-step process involving the following key steps:
Nitration: The nitration of benzylamine to form 2-nitrobenzylamine.
Cyclization: The cyclization of 2-nitrobenzylamine with glyoxal to form 2-nitroimidazole.
Reduction: The reduction of 2-nitroimidazole to 2-aminoimidazole.
Acylation: The acylation of 2-aminoimidazole with benzoyl chloride to form this compound.
Industrial Production Methods: Industrial production of this compound typically involves the same synthetic route but on a larger scale. The process is optimized for higher yields and purity, often using advanced techniques such as continuous flow reactors and automated synthesis .
Chemical Reactions Analysis
Metabolic Activation Pathways
Benznidazole undergoes enzymatic reduction primarily in the liver and Trypanosoma cruzi parasites, mediated by:
This activation generates electrophilic metabolites that alkylate DNA (forming 8-oxoguanine adducts) and deplete cellular thiols .
Reductive Metabolism in Trypanosomes
The trypanocidal mechanism involves a ping-pong enzymatic mechanism with T. cruzi nitroreductase (TcNTR):
Reaction sequence :
-
Initial reduction :
-
Secondary dehydration :
Forms 4,5-dihydro-4,5-dihydroxyimidazole (detected via LC/MS) -
Glyoxal release :
Glyoxal reacts with guanosine to form stable adducts (e.g., 1,N2-glyoxal-guanine), disrupting parasite DNA replication .
Oxidative Stress Pathways
Under aerobic conditions, this compound undergoes futile redox cycling:
| Process | Chemical Equation | Biological Impact |
|---|---|---|
| Superoxide generation | Mitochondrial damage in parasites | |
| Thiol depletion | Antioxidant defense collapse |
This dual mechanism explains its selective toxicity toward trypanosomes over mammalian cells .
Synthetic Routes and Key Reactions
Industrial synthesis employs nucleophilic substitution (SN2):
Primary method :
-
Reactants : 2-Nitroimidazole, N-benzyl-2-chloroacetamide
-
Conditions :
-
Base: K₂CO₃ (1:3.9 molar ratio)
-
Catalyst: Tetrabutylammonium bromide
-
Temperature: 70°C, 72 hr reaction time
-
-
Yield : 87% after recrystallization (acetone:methanol:water = 49.9:49.9:5.3)
Critical quality control parameters :
| Parameter | Specification | Analytical Method |
|---|---|---|
| Purity | ≥99.5% | HPLC with photodiode array |
| Residual solvents | <500 ppm | GC-MS |
| Crystal form | Monoclinic | X-ray diffraction |
Adverse Reaction Chemistry
Cutaneous toxicity arises from:
-
Hapten formation : Nitroso metabolites covalently bind to skin proteins (e.g., keratin)
-
Immune response : MHC-I presentation of drug-protein adducts triggers CD8+ T-cell activation
Dose-dependent effects:
| Metabolite Concentration | Clinical Manifestation |
|---|---|
| <50 μM | Maculopapular rash |
| >100 μM | Exfoliative dermatitis |
Antihistamines mitigate early-stage reactions by blocking histamine H1 receptors, but delayed hypersensitivity requires corticosteroid intervention .
Stability and Degradation
This compound degrades under alkaline conditions:
-
Hydrolysis :
-
Photodegradation :
UV exposure generates nitroso derivatives (λmax = 340 nm) requiring amber glass packaging .
This comprehensive profile underscores this compound's dual role as a prodrug and toxicant, guided by its intricate redox chemistry. Optimizing therapeutic outcomes requires balancing metabolic activation against off-target reactivity through dose modulation and adjunct therapies.
Scientific Research Applications
Benznidazole has a wide range of scientific research applications, including:
Chemistry: this compound is used as a model compound for studying nitroimidazole chemistry and its reactivity.
Biology: this compound is used to study the biology of Trypanosoma cruzi and the mechanisms of antiparasitic action.
Medicine: this compound is extensively used in clinical research for the treatment of Chagas disease and other parasitic infections.
Mechanism of Action
Benznidazole exerts its effects by being reduced to various electrophilic metabolites by nitroreductases present in Trypanosoma cruzi . These metabolites bind to proteins, lipids, DNA, and RNA, resulting in damage to these macromolecules. This damage leads to the death of the parasite. This compound has also been found to increase trypanosomal death through interferon-γ, which is likely present in increased amounts due to inflammation caused by macromolecule damage .
Comparison with Similar Compounds
Nifurtimox: Another nitroimidazole compound used for the treatment of Chagas disease.
Pentamidine: An antimicrobial used for the treatment of trypanosomiasis and leishmaniasis.
Uniqueness of Benznidazole: this compound is unique in its moderate side effect profile and its effectiveness in the early stages of Chagas disease. It is also the first treatment approved for Chagas disease in the United States .
Biological Activity
Benznidazole is a nitroimidazole derivative primarily used in the treatment of Chagas disease, caused by the parasite Trypanosoma cruzi. Its biological activity is characterized by its mechanism of action, efficacy in clinical settings, and associated side effects. This article synthesizes findings from various studies to provide a comprehensive overview of the biological activity of this compound.
This compound exerts its antiparasitic effects through the generation of reactive nitrogen species (RNS) upon reduction by nitroreductases present in the parasite. These RNS interact with cellular components, leading to oxidative stress and ultimately cell death in T. cruzi . The drug's effectiveness is influenced by its ability to penetrate the parasite's cellular environment and disrupt critical metabolic processes.
Efficacy in Clinical Trials
Numerous clinical trials have demonstrated the efficacy of this compound in treating Chagas disease. A systematic review indicated that this compound significantly increases the likelihood of therapeutic response compared to placebo, with an odds ratio (OR) of 18.8 (95% CI: 5.2–68.3) . In a Phase II trial, sustained parasitological clearance was observed in 89% of patients receiving a daily dose of 300 mg for eight weeks, compared to only 3% in the placebo group .
Summary of Clinical Findings
| Study Type | Treatment Regimen | Sustained Clearance (%) | Odds Ratio (vs. Placebo) |
|---|---|---|---|
| Clinical Trials | 300 mg daily for 8 weeks | 89% | 70.8 |
| Observational Studies | Various regimens | 7.8 | 7.8 |
| Randomized Control | 60 days treatment | 55.8% | - |
Case Studies and Observational Data
In a randomized trial involving schoolchildren in Brazil, this compound treatment resulted in a significant reduction of T. cruzi antibodies, indicating effective clearance of the infection . The study reported a negative seroconversion rate of 58% among treated children compared to only 5% in the placebo group.
Another study focused on adults with chronic Chagas disease found that this compound treatment led to a decrease in clinical events, with an OR of 0.29 (95% CI: 0.16–0.53) for adverse outcomes among treated patients .
Adverse Effects and Tolerability
This compound is associated with several adverse effects, including cutaneous reactions and gastrointestinal disturbances, leading to treatment discontinuation in approximately 18% of patients . A recent study highlighted that while adverse events were common, they were generally manageable and less frequent in shorter treatment regimens .
Adverse Effects Summary
| Adverse Effect | Incidence (%) | Comments |
|---|---|---|
| Cutaneous reactions | ~12-18 | Common but manageable |
| Gastrointestinal issues | ~10-15 | Less frequent in children |
| Serious events | ~3 | Rare but documented |
Innovations in Drug Delivery
Recent research has explored novel formulations to enhance this compound's efficacy and reduce toxicity. Encapsulation in nanostructured lipid carriers (NLC) has shown promise, resulting in improved bioavailability and reduced hemolytic activity compared to free this compound . This approach may facilitate better patient adherence by minimizing side effects while maintaining therapeutic effectiveness.
Q & A
Basic Research Questions
Q. What are the standardized protocols for benznidazole administration in preclinical and clinical studies?
this compound is typically administered at 5–7.5 mg/kg/day in two or three divided doses for 60 days in clinical trials . In murine models, dosages range from 50–100 mg/kg/day for acute-phase studies, adjusted based on parasite strain and disease progression . Researchers must account for interpatient variability in pharmacokinetics, particularly in pediatric populations, where lower plasma concentrations still correlate with efficacy .
Q. How should this compound be handled in laboratory settings to mitigate safety risks?
this compound is classified as a skin/eye irritant and potential respiratory hazard. Safe handling requires:
- Use of PPE (gloves, lab coats, goggles).
- Ventilation controls to avoid dust inhalation .
- Immediate decontamination of spills with water or ethanol-based solvents . Stability studies recommend storage at 2–8°C in airtight containers to prevent degradation .
Q. What factors contribute to treatment suspension in this compound clinical trials?
Up to 33% of patients discontinue treatment due to adverse drug reactions (ADRs), primarily dermatological (e.g., rash, photosensitivity) and gastrointestinal effects. Female patients and those with pre-existing skin conditions are at higher risk . Mitigation strategies include dose titration, antihistamine co-administration, and close monitoring during the first 30 days .
Advanced Research Questions
Q. How do Trypanosoma cruzi discrete typing units (DTUs) influence this compound sensitivity?
T. cruzi DTUs exhibit marked variability in drug susceptibility:
- TcI : High LC50 (137.62 μM for trypomastigotes), indicating resistance .
- TcII/TcVI : Lower LC50 (25.81–52.09 μM), suggesting higher sensitivity . Meta-analyses of in vitro data reveal significant differences in IC50/LC50 between DTUs (p<0.05), necessitating strain-specific dosing in experimental models .
Q. Why do this compound-induced PCR conversion rates fail to correlate with clinical outcomes in chronic Chagas cardiomyopathy?
In the BENEFIT trial, this compound achieved PCR negativity in 66.2% of patients post-treatment vs. 33.5% with placebo (p<0.001), yet no significant reduction in cardiac events occurred (HR 0.93, p=0.31) . This discrepancy may arise from:
- Persistent low-level parasitism undetectable by PCR.
- Irreversible myocardial damage prior to treatment .
- Regional variations in DTU distribution impacting drug efficacy .
Q. What methodological approaches optimize combination therapies involving this compound?
Co-administration with azoles (e.g., posaconazole, itraconazole) enhances efficacy:
- Murine models : this compound + posaconazole reduced parasitemia by 98% vs. 70% with monotherapy .
- Synergy mechanisms : Azoles inhibit ergosterol biosynthesis, while this compound generates nitro-reductive radicals, targeting multiple parasite pathways . Sequential dosing (this compound followed by azoles) may prevent relapse in chronic infections .
Q. How should researchers address confounding biases in observational studies of this compound efficacy?
Key strategies include:
- Propensity score matching : To balance covariates (e.g., age, comorbidities) between treated and untreated cohorts .
- Counterfactual analysis : Excluding patients with baseline cardiac abnormalities to isolate treatment effects .
- Sensitivity analyses : Testing models with/without this compound as a variable to assess robustness .
Q. Methodological Guidance
Designing dose-response studies for this compound-resistant T. cruzi strains
- Use nested PCR to confirm DTU classification pre-treatment .
- Incorporate time-to-event endpoints (e.g., parasite recrudescence) rather than binary PCR outcomes .
- Adjust dosing regimens using pharmacokinetic/pharmacodynamic (PK/PD) modeling, particularly for TcI-dominated cohorts .
Evaluating long-term this compound safety in pediatric populations
- Monitor neurodevelopmental endpoints : Murine studies associate nitroimidazoles with Purkinje cell damage .
- Track chromosomal aberrations : Mean incidence increases 2-fold post-treatment, necessitating cytogenetic analysis in longitudinal cohorts .
Addressing regional heterogeneity in this compound clinical trials
The BENEFIT trial showed geographic variability:
- Brazil/Argentina : OR 2.63–3.03 for PCR conversion (p<0.001).
- Colombia/El Salvador : No significant benefit (OR 1.33, p=0.16) .
Stratify randomization by DTU prevalence and incorporate geospatial mapping to contextualize results.
Contradictions and Recommendations
Properties
IUPAC Name |
N-benzyl-2-(2-nitroimidazol-1-yl)acetamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C12H12N4O3/c17-11(14-8-10-4-2-1-3-5-10)9-15-7-6-13-12(15)16(18)19/h1-7H,8-9H2,(H,14,17) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CULUWZNBISUWAS-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=CC=C(C=C1)CNC(=O)CN2C=CN=C2[N+](=O)[O-] | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C12H12N4O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9046570 | |
| Record name | Benznidazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9046570 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
260.25 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
>39 [ug/mL] (The mean of the results at pH 7.4) | |
| Record name | SID56323658 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
Mechanism of Action |
Benznidazole is thought to be reduced to various electrophilic metabolites by nitroreductases present in *Trypanosoma cruzi*. These metabolites likely bind to proteins, lipids, DNA, and RNA resulting in damage to these macromolecules. Benznidazole has been found to increase trypanosomal death through interferon-γ which is likely present in increased amounts due to inflammation caused by macromolecule damage. DNA in parasites affected by benznidazole has been found to undergo extensive unpacking with overexpression of DNA repair proteins supporting the idea of DNA damage contributing to the mechanism of the drug. | |
| Record name | Benznidazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11989 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
22994-85-0 | |
| Record name | Benznidazole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=22994-85-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Benznidazole [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0022994850 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Benznidazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11989 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BENZNIDAZOLE | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=299972 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Benznidazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9046570 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | N-Benzyl-2-nitro-1H-imidazole-1-acetamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | BENZNIDAZOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/YC42NRJ1ZD | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Melting Point |
190-192 | |
| Record name | Benznidazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11989 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















