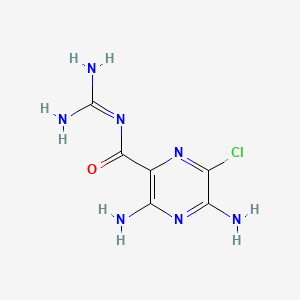
Amiloride
Overview
Description
Amiloride is a pyrazine derivative that is widely used as a potassium-sparing diuretic. It is primarily employed in the treatment of hypertension and congestive heart failure. This compound works by inhibiting sodium reabsorption in the kidneys, which helps to reduce fluid retention and lower blood pressure .
Preparation Methods
Synthetic Routes and Reaction Conditions: Amiloride can be synthesized through a multi-step process involving the reaction of 3,5-diamino-6-chloropyrazine-2-carboxamide with various reagents. One common method involves the reaction of 3,5-diamino-6-chloropyrazine-2-carboxamide with cyanamide under acidic conditions to form this compound .
Industrial Production Methods: Industrial production of this compound typically involves large-scale synthesis using similar reaction conditions as in laboratory synthesis. The process is optimized for yield and purity, often involving purification steps such as recrystallization and chromatography to ensure the final product meets pharmaceutical standards .
Chemical Reactions Analysis
Structural Features Influencing Reactivity
Amiloride’s core structure includes:
-
A pyrazine ring substituted with chlorine (Cl) at C(6)
-
A carbonylguanidinium group at C(2)
-
Primary and secondary amines at C(3) and C(5)
These functional groups enable nucleophilic substitution, charge-transfer interactions, and acid-base reactions. The guanidine group (pKa 8.67) deprotonates in alkaline conditions, altering reactivity .
Substitution Reactions at Pyrazine Ring
Modifications at C(2) and C(5) positions enhance cytotoxic properties. Key findings from derivative synthesis :
| Derivative | Position Modified | Substituent | IC₅₀ (μM) | Key Observation |
|---|---|---|---|---|
| LLC1 | C(5) | Lipophilic chain | 1.2–2.8 | 10× higher potency than HMA |
| LLC3 | C(5) | Lipophilic chain (reduced guanidine) | 5.6–23.1 | 3–23× lower potency than LLC1 |
Critical Insights :
-
Guanidine integrity is essential for cytotoxicity (e.g., LLC1 vs. LLC3) .
-
Lipophilicity (logP) correlates with enhanced membrane permeability and potency .
Charge-Transfer Complexation
This compound forms stable 1:1 charge-transfer complexes with π-acceptors, enabling spectrophotometric quantification :
Reaction with DDQ (Method A)
-
Mechanism : Electron donation from this compound’s guanidine to DDQ.
-
Product : Orange-yellow complex (λₘₐₓ = 476 nm).
Reaction with PCA (Method B)
-
Mechanism : Similar charge transfer, producing a pink complex (λₘₐₓ = 508 nm).
Optimized Conditions :
| Parameter | DDQ (Method A) | PCA (Method B) |
|---|---|---|
| Reagent Concentration | 0.1% | 0.1% |
| Reaction Time | 10 min | 10 min |
| Temperature | 60°C | 25°C |
Stability and Degradation
-
pH Sensitivity : Deprotonation at pH > 8.67 reduces sodium channel inhibition .
-
Photodegradation : Fluorescence (λₑₘ = 420 nm) indicates susceptibility to UV light .
Derivatization for Enhanced Bioactivity
-
C(5) lipophilic chains (e.g., hexamethylene) improve cytotoxicity against breast cancer cells (IC₅₀ = 1.2–2.8 µM) .
-
C(2) modifications (e.g., benzamil) target ion channels but reduce potency if guanidine is altered .
Quantitative Analysis
| Parameter | Method A (DDQ) | Method B (PCA) |
|---|---|---|
| LOD (µg/mL) | 0.766 | 0.014 |
| LOQ (µg/mL) | 2.553 | 0.047 |
| Recovery (%) | 99.4–100.4 | 98.9–101.3 |
Reaction Mechanisms and Selectivity
Scientific Research Applications
Amiloride is a medication primarily known as an anti-kaliuretic and epithelial sodium channel blocker, but it also has various other applications in treating different conditions. It has been used clinically for decades in the management of hypertension and as a pharmacological tool for examining sodium transport .
Scientific Research Applications
Hypertension and Edema
this compound is used to treat hypertension and edema . A study showed that a daily 10 mg dose of this compound taken in the morning with breakfast for four weeks significantly reduced peripheral systolic blood pressure (SBP) by 7.06 ± 2.25 mmHg and diastolic blood pressure (DBP) by 4.35 ± 1.67 mmHg . Central SBP was reduced by 7.68 ± 2.56 mmHg, and central DBP by 4.49 ± 1.78 mmHg . There were also significant increases in flow-mediated dilation (FMD) and a reduction in carotid radial pulse wave velocity (CR-PWV), indicating improved endothelial function and lowered peripheral arterial stiffness .
In a case study, a patient with type 1 diabetes, severe hypertension (200/140 mmHg), progressive edema (of at least 10 L), and overt proteinuria (18.5 g/24 h) was treated with this compound (5 mg/day) in addition to other antihypertensive drugs . This resulted in the resolution of edema, a weight loss of 7 kg, and a reduction in blood pressure (150/100-125/81 mmHg) .
The AMILOR study, a monocentric randomized controlled pilot study, found that this compound had a similar antiedematous effect as furosemide in patients with edema .
Proteinuria
this compound can reduce proteinuria in patients with proteinuric kidney disease . A study involving 12 patients showed that this compound reduced 24-hour urine protein by 38.7% and decreased systolic blood pressure by 12.3 mm Hg .
Cystic Fibrosis
this compound has been researched in the context of cystic fibrosis because of its ability to inhibit epithelial sodium channels (ENaC) .
Pain Reduction
This compound can block proton-induced pain in humans . Simultaneous application of 200 μM this compound reduced acid-evoked pain almost to the pH 7.4 control level . Pretreatment with this compound significantly reduced irritant sensations elicited by citric acid on the dorsal surface of the tongue, but did not reduce capsaicin-evoked oral irritation, suggesting an this compound-sensitive mechanism in acid-induced irritation .
Cancer Treatment
This compound has demonstrated anti-cancer and anti-metastatic potential in vitro and in vivo . It has been shown to inhibit mutagen-induced carcinogenesis, tumor formation, and metastatic progression in rats and mice . These effects are generally attributed to the cytostatic effects of this compound inhibiting sodium hydrogen exchanger-1 (NHE1) and the motility-suppressing effects of urokinase plasminogen activator (uPA) inhibition .
A novel lipophilic C(5) this compound derivative, LLC1, can induce cell death in the single-digit micromolar range, selectively eradicating chemoresistant breast tumor cells .
Other Applications
This compound-based ligands have been identified as small molecules . this compound has been found to be ineffective at all doses tested, implicating opioid receptors, GC-C, and sodium and TRP channel activation as possible pharmacological reference compounds .
Data Table
Case Studies
Case Study 1: Hypertension and Edema
A 38-year-old male with type 1 diabetes developed severe hypertension (200/140 mmHg), progressive edema (at least 10 L), and overt proteinuria (18.5 g/24 h) despite being treated with five antihypertensive drugs . The addition of this compound at 5 mg/day to his treatment regimen resulted in the resolution of edema, a weight loss of 7 kg, and a reduction in blood pressure to 125/81 mmHg . The patient's 24-hour urinary sodium excretion increased (127-165 mmol/day), his eGFR decreased (41-29 mL/min), and his plasma potassium concentration increased (4.6-7.8 mmol/L) .
Case Study 2: Proteinuria
In a study of 12 patients with proteinuric kidney disease, this compound was administered at 5 mg twice daily . The results showed a 38.7% reduction in 24-hour urine protein and a 12.3 mm Hg decrease in systolic blood pressure . These findings suggest that this compound can be effective in reducing proteinuria and improving blood pressure control in patients with kidney disease .
Mechanism of Action
Amiloride works by inhibiting the epithelial sodium channels (ENaC) in the distal convoluted tubules and collecting ducts of the kidneys. This inhibition reduces sodium reabsorption and promotes the excretion of sodium and water, while sparing potassium. The molecular targets of this compound include the ENaC, which plays a crucial role in regulating sodium and potassium balance in the body .
Comparison with Similar Compounds
Triamterene: Another potassium-sparing diuretic with a similar mechanism of action.
Spironolactone: A potassium-sparing diuretic that works by antagonizing aldosterone receptors.
Eplerenone: Similar to spironolactone but with fewer side effects.
Uniqueness of Amiloride: this compound is unique in its specific inhibition of the ENaC, which makes it particularly effective in conditions where sodium retention is a problem. Unlike spironolactone and eplerenone, this compound does not affect aldosterone receptors, which reduces the risk of hormonal side effects .
Biological Activity
Amiloride is a potassium-sparing diuretic primarily used in the treatment of hypertension and heart failure. It selectively inhibits epithelial sodium channels (ENaC), leading to various biological effects beyond its diuretic action. This article explores the biological activity of this compound, focusing on its mechanisms of action, therapeutic applications, and recent research findings.
This compound's primary mechanism involves the inhibition of ENaC located in the distal nephron, lung, and colon. By blocking these channels, this compound reduces sodium reabsorption and promotes potassium retention, which is crucial in managing conditions like hypertension and edema. The molecular structure of ENaC consists of three subunits (α, β, and γ), and mutations in these subunits can lead to disorders such as Liddle syndrome, where ENaC activity is abnormally high .
Key Mechanisms:
- Inhibition of ENaC: Reduces sodium reabsorption.
- Impact on Acid-Sensing Ion Channels (ASICs): Involved in various physiological processes.
- Interaction with Na+/H+ Antiporters and Na+/Ca2+ Exchangers: Affects intracellular pH and calcium transport during cellular activation .
Therapeutic Applications
This compound has been investigated for several therapeutic uses beyond its traditional role as a diuretic:
- Hypertension Management: It is often combined with other diuretics to enhance blood pressure control.
- Edema Treatment: this compound has shown effectiveness in treating resistant edema in patients with conditions like nephrotic syndrome .
- Bone Health: Recent studies indicate that this compound inhibits osteoclastogenesis, suggesting potential applications in treating bone loss-related diseases .
- Cancer Research: this compound and its derivatives are being explored for their cytotoxic properties against various cancer cell lines, showing promise in targeting chemoresistant tumors .
Case Study 1: Resistant Edema
A study involving a 38-year-old male patient with type 1 diabetes revealed that the addition of this compound (5 mg/day) to his treatment regimen resolved severe edema and significantly reduced blood pressure from 200/140 mmHg to 150/100 mmHg after several days. The patient's urinary sodium excretion increased, indicating effective diuretic action .
Case Study 2: Osteoclastogenesis Inhibition
Research demonstrated that this compound effectively inhibited RANKL-induced osteoclast differentiation in vitro. The study showed a concentration-dependent decrease in TRAP-positive multinucleated cells, highlighting its potential as a therapeutic agent for diseases like osteoporosis .
Recent Research Findings
Recent studies have expanded the understanding of this compound's biological activity:
- Antiedematous Effects: The AMILOR study compared this compound with furosemide for treating edema. While both showed similar effects in reducing extracellular water over time, further research is needed to establish this compound's definitive role .
- Cancer Therapeutics: Investigations into this compound derivatives have revealed enhanced cytotoxicity against breast cancer cells while sparing normal cells. This specificity may be due to the modulation of lysosomal function and sodium-hydrogen exchanger inhibition .
Comparative Data Table
Q & A
Basic Research Questions
Q. What are the primary molecular targets of amiloride, and how do these interactions influence experimental design in sodium transport studies?
this compound primarily inhibits epithelial sodium channels (ENaC) and acid-sensing ion channel-1 (ASIC-1), which are critical in sodium homeostasis and cellular signaling. Methodologically, researchers often use in vitro electrophysiology (e.g., whole-cell patch clamp) to quantify this compound’s inhibitory effects on ion currents . For ENaC studies, experimental designs should account for tissue-specific expression (e.g., renal vs. airway epithelia) and validate target specificity using competitive antagonists or knockout models .
Q. How can researchers standardize this compound solution preparation to address solubility challenges in physiological studies?
this compound’s limited solubility in isotonic solutions (e.g., 0.3% NaCl) necessitates careful preparation. A common protocol involves preparing a 10 mmol/L stock at pH 6.4, stored in dark glass vials at room temperature to prevent flocculation. If precipitation occurs, warming the solution to 50°C and manual agitation can restore homogeneity. Researchers must report osmolality (e.g., 116 mOsm for 10 mmol/L this compound vs. 303 mOsm for frusemide) and validate stability via UV spectrophotometry or LC-MS .
Q. What statistical approaches are recommended for analyzing this compound’s dose-response effects in airway resistance studies?
Repeated-measures ANOVA is suitable for comparing pre- and post-treatment outcomes (e.g., FEV1 changes). For dose-response curves, non-linear regression models (e.g., log[inhibitor] vs. response) should include 95% confidence intervals. In studies with heterogeneous responses (e.g., "LOW" vs. "HIGH" this compound responders), cluster analysis or stratification by baseline parameters (e.g., sodium intake) can reduce variability .
Advanced Research Questions
Q. How can computational docking studies guide the design of this compound analogs with enhanced specificity for ASIC-1?
Molecular docking software (e.g., AutoDock Vina) predicts binding affinities between this compound and ASIC-1’s extracellular domains. Researchers should prioritize analogs with lower free energy scores (ΔG < -7 kcal/mol) and validate predictions via electrophysiology. For example, substituting the guanidinium group with hydrophobic moieties may improve potency. Post-docking, mutagenesis of predicted interaction sites (e.g., residue 299 in CVB3 polymerase) can confirm mechanistic relevance .
Q. What methodologies resolve conflicting data on this compound’s role in tumor microenvironment modulation?
Discrepancies in MMP3/MMP9 inhibition (e.g., reduced activity in lung tumors vs. no effect in glioblastoma) may arise from pH-dependent drug stability. Multivariate curve resolution alternating least squares (MCR-ALS) can deconvolute photodegradation products under varying pH (4.0–9.0) and irradiation conditions. Complementary LC-MS identifies degradation byproducts (e.g., hydroxylated derivatives), ensuring observed effects are drug-specific rather than artifact-driven .
Q. How does this compound synergize with existing therapies in relapsed multiple myeloma, and what experimental models validate this?
In xenograft models, this compound (5–20 mg/kg daily) synergizes with dexamethasone by disrupting spliceosomal machinery (e.g., downregulating SF3B1). RNA-Seq and isoform-level analysis (e.g., Cufflinks) quantify alternative splicing events. Mechanistic validation requires immunofluorescence for nuclear speckle disassembly and p53 pathway activation via immunoblotting (e.g., phospho-p53 Ser15). Studies must include TP53-mutant cell lines to assess resistance mechanisms .
Q. Methodological Considerations
Q. How should researchers address variability in this compound responses across epithelial tissues?
Stratify samples by transporter expression (e.g., Ussing chamber measurements of trans-epithelial resistance). For airway studies, differentiate between apical vs. basolateral ENaC activity using polarized cell cultures. In renal research, urinary this compound concentration assays (via HPLC) correlate pharmacokinetics with sodium excretion .
Q. What analytical frameworks are optimal for studying this compound’s pH-dependent photodegradation in drug formulation research?
Combine kinetic and acid-base titration data via MCR-ALS with hard modeling constraints. Equilibrium constraints resolve acid-base species (e.g., protonated vs. deprotonated this compound), while kinetic constraints model degradation pathways. Rate constants (k) estimated via Arrhenius plots at 25–45°C inform storage guidelines .
Properties
IUPAC Name |
3,5-diamino-6-chloro-N-(diaminomethylidene)pyrazine-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C6H8ClN7O/c7-2-4(9)13-3(8)1(12-2)5(15)14-6(10)11/h(H4,8,9,13)(H4,10,11,14,15) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XSDQTOBWRPYKKA-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1(=C(N=C(C(=N1)Cl)N)N)C(=O)N=C(N)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C6H8ClN7O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
17440-83-4 (hydrochloride), 2016-88-8 (anhydrous hydrochloride) | |
| Record name | Amiloride [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0002609463 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID9043853 | |
| Record name | Amiloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9043853 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
229.63 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Amiloride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014732 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Slightly soluble, 1.22e+00 g/L | |
| Record name | Amiloride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00594 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Amiloride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014732 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Amiloride works by inhibiting sodium reabsorption in the distal convoluted tubules and collecting ducts in the kidneys by binding to the amiloride-sensitive sodium channels. This promotes the loss of sodium and water from the body, but without depleting potassium. Amiloride exerts its potassium sparing effect through the inhibition of sodium reabsorption at the distal convoluted tubule, cortical collecting tubule and collecting duct; this decreases the net negative potential of the tubular lumen and reduces both potassium and hydrogen secretion and their subsequent excretion. Amiloride is not an aldosterone antagonist and its effects are seen even in the absence of aldosterone. | |
| Record name | Amiloride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00594 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
2609-46-3, 2016-88-8 | |
| Record name | Amiloride | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=2609-46-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Amiloride [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0002609463 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Amiloride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00594 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Amiloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9043853 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Amiloride | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.018.205 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | AMILORIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/7DZO8EB0Z3 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Amiloride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014732 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
240.5-241.5, 240 °C | |
| Record name | Amiloride | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00594 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Amiloride | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014732 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















