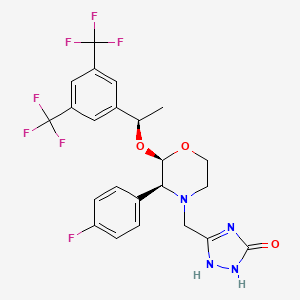
Aprepitant
Overview
Description
Aprepitant is a medication primarily used to prevent chemotherapy-induced nausea and vomiting, as well as postoperative nausea and vomiting . It belongs to the class of neurokinin-1 receptor antagonists and works by blocking the action of substance P, a neuropeptide associated with vomiting . This compound is marketed under the brand name Emend, among others .
Preparation Methods
Synthetic Routes and Reaction Conditions: Aprepitant is synthesized through a multi-step process involving several key intermediates. The synthesis typically starts with the preparation of a morpholine derivative, which is then subjected to various chemical reactions, including alkylation, acylation, and cyclization . The final product is obtained through purification and crystallization steps to ensure high purity and yield .
Industrial Production Methods: In industrial settings, the production of this compound involves optimizing the reaction conditions to maximize yield and minimize impurities. Techniques such as high-performance liquid chromatography (HPLC) and gas chromatography (GC) are used to monitor the reaction progress and ensure the quality of the final product . Additionally, advanced formulation techniques, such as nanoprecipitation and solid dispersion, are employed to enhance the solubility and bioavailability of this compound .
Chemical Reactions Analysis
Types of Reactions: Aprepitant undergoes various chemical reactions, including oxidation, reduction, and substitution . These reactions are essential for modifying the chemical structure and improving the pharmacological properties of the compound.
Common Reagents and Conditions: Common reagents used in the synthesis of this compound include alkylating agents, acylating agents, and reducing agents . The reactions are typically carried out under controlled conditions, such as specific temperatures, pressures, and pH levels, to ensure optimal yields and minimal side reactions .
Major Products Formed: The major products formed from the chemical reactions of this compound include its various intermediates and the final active pharmaceutical ingredient (API) . These products are characterized using techniques such as nuclear magnetic resonance (NMR) spectroscopy, mass spectrometry (MS), and infrared (IR) spectroscopy to confirm their structures and purities .
Scientific Research Applications
Chemotherapy-Induced Nausea and Vomiting (CINV)
Overview
Aprepitant is predominantly used in combination with other antiemetic agents to manage CINV, especially in patients receiving highly emetogenic chemotherapy. The drug's efficacy has been established through multiple clinical trials.
Efficacy Data
A systematic review highlighted the effectiveness of this compound in preventing CINV. In a phase III study, patients receiving this compound demonstrated a complete response rate of 83.2% compared to 71.3% in the control group (P < 0.05) .
| Study | Treatment Group | Complete Response Rate (%) | Control Group Response Rate (%) |
|---|---|---|---|
| Hesketh et al., 2003 | This compound + standard therapy | 83.2 | 71.3 |
| Poli-Bigelli et al., 2003 | This compound + standard therapy | 62 | 52 |
Antitumor Effects
Mechanism of Action
Recent studies have indicated that this compound may possess antitumor properties. Research has shown that it induces apoptosis in tumor cells and can reduce tumor volume in animal models .
Case Studies
In preclinical trials involving human osteosarcoma and hepatoblastoma xenografts, this compound significantly decreased tumor size when administered alongside conventional therapies .
Dermatological Applications
Topical Use
this compound has been explored for its topical application to mitigate side effects associated with other cancer treatments, such as erlotinib-induced dermatitis and hair loss. In a study involving rats, topical administration of this compound resulted in up to a 70% reduction in neutrophil activity related to inflammation caused by erlotinib .
| Treatment Group | Neutrophil Activity Reduction (%) |
|---|---|
| Control | Baseline |
| Low Dose | 24% reduction |
| High Dose | 70% reduction |
Potential Use in Other Conditions
Pruritus Management
Emerging research suggests that this compound may be effective in treating pruritus (itching) associated with various conditions, particularly in cancer patients .
Antiviral Properties
Studies have also hinted at the antiviral effects of this compound, suggesting its potential use against certain viral infections by modulating immune responses .
Mechanism of Action
Aprepitant exerts its effects by selectively blocking the neurokinin-1 (NK1) receptors in the brain . These receptors are activated by substance P, a neuropeptide involved in the vomiting reflex . By inhibiting the binding of substance P to NK1 receptors, this compound effectively prevents the transmission of signals that trigger nausea and vomiting . This mechanism of action makes this compound a valuable adjunct therapy in combination with other antiemetic agents, such as serotonin (5-HT3) receptor antagonists and corticosteroids .
Comparison with Similar Compounds
Aprepitant is often compared with other antiemetic agents, such as ondansetron and dexamethasone . While ondansetron is a serotonin (5-HT3) receptor antagonist and dexamethasone is a corticosteroid, this compound is unique in its mechanism of action as an NK1 receptor antagonist . This distinct mechanism allows this compound to complement traditional antiemetic drugs and enhance the control of chemotherapy-induced nausea and vomiting . Other similar compounds include fosthis compound, a prodrug of this compound, which is converted to this compound in the body and shares the same mechanism of action .
Biological Activity
Aprepitant is a neurokinin-1 (NK-1) receptor antagonist primarily used for the prevention of chemotherapy-induced nausea and vomiting (CINV). Recent research has expanded its potential applications, particularly in oncology, where it has shown promise as an anticancer agent. This article delves into the biological activity of this compound, highlighting its mechanisms of action, efficacy in clinical studies, and emerging roles in cancer therapy.
This compound functions by selectively blocking the NK-1 receptors in the central nervous system, which are activated by substance P, a neuropeptide involved in the vomiting reflex. By inhibiting these receptors, this compound effectively reduces both acute and delayed phases of CINV induced by chemotherapeutic agents such as cisplatin .
Recent studies have revealed additional mechanisms through which this compound exerts its effects:
- Induction of Immunogenic Cell Death : this compound has been shown to activate the anticipatory unfolded protein response (a-UPR), leading to necrotic cell death that stimulates immune responses. This process involves the release of damage-associated molecular patterns (DAMPs), such as HMGB1 and ATP, which enhance macrophage migration and activation .
- Calcium Release and Stress Response Activation : The drug appears to trigger calcium release from the endoplasmic reticulum, resulting in hyperactivation of stress response pathways that contribute to its anticancer effects .
Chemotherapy-Induced Nausea and Vomiting (CINV)
A meta-analysis encompassing 23 randomized controlled trials with 7,956 patients demonstrated that this compound-containing regimens significantly improved complete response rates for CINV compared to standard therapies. The results indicated:
- Complete Response Rates : 50.3% in standard therapy vs. 66.4% and 70.5% in this compound 40/25 mg and 125/80 mg groups respectively .
- Safety Profile : No significant differences in severe adverse events were noted between groups; however, higher incidences of fatigue and hiccups were observed in the this compound group .
Case Studies
A case study reported successful outcomes using this compound for refractory postoperative nausea and vomiting, suggesting its utility beyond traditional applications .
Efficacy Against Cancer Cells
Recent preclinical studies have established that this compound not only alleviates nausea but also induces cell death in various cancer types:
- Breast Cancer Models : this compound treatment resulted in typical features of necrotic cell death, including cell swelling and membrane rupture. This mode of action was confirmed through both 2D cell culture and 3D organoid models .
Summary of Key Trials
| Study Type | Population | Treatment Regimen | Complete Response Rate | Adverse Events |
|---|---|---|---|---|
| Phase II | Japanese cancer patients | This compound + standard therapy | 70.5% (highest) | Similar to standard therapy |
| Meta-analysis | 7,956 patients | This compound-containing regimens | Significantly improved vs. control | Higher fatigue incidence |
| Case Study | Post-operative patients | This compound for refractory nausea | Improvement noted | Not specified |
Q & A
Q. Basic: How are randomized controlled trials (RCTs) designed to evaluate aprepitant's efficacy in chemotherapy-induced nausea and vomiting (CINV)?
RCTs for this compound typically employ a double-blind, placebo-controlled design with standardized antiemetic regimens. Key endpoints include "complete response" (no emesis/no rescue therapy) over 5 days post-chemotherapy. For example, in cisplatin-based chemotherapy, this compound (125 mg orally on day 1; 80 mg on days 2–3) is combined with ondansetron and dexamethasone, while the control group receives standard dual therapy . Statistical analysis uses logistic regression models to compare outcomes, with significance thresholds at p < 0.05. Sample sizes are calculated to ensure power (e.g., 260 patients per group in Study 052) .
Q. Basic: What preclinical models validate this compound's pharmacokinetics and brain penetration?
Ferrets are a validated preclinical model due to human-like NK1 receptor pharmacology. Studies administer this compound orally (1–3 mg/kg) and measure plasma and brain concentrations via LC-MS. For instance, brain cortex levels reach 80–150 ng/g 24 hours post-dose, correlating with sustained antiemetic efficacy against cisplatin-induced emesis . Plasma clearance (~1.5 mL/min/kg) and metabolite profiling (parent compound dominance) confirm this compound’s stability and primary role in efficacy .
Q. Advanced: How can conflicting pharmacokinetic data in patient subpopulations be resolved?
Conflicting data, such as age-related effects on this compound clearance in Japanese patients, require meta-analysis and covariate adjustment. For example, one study found no correlation between age and plasma exposure (n = 44), while another (n = 315) reported mild age-dependent clearance changes . Methodological solutions include:
- Stratified randomization in trials.
- Population pharmacokinetic modeling to quantify covariates (e.g., CYP3A4 activity).
- Sensitivity analyses excluding outliers .
Q. Advanced: What methodologies elucidate this compound's off-target mechanisms, such as nSMase2 inhibition?
Virtual screening (e.g., molecular docking) identifies this compound’s interaction with nSMase2, validated via:
- Cell-free assays : Measure enzyme activity inhibition (IC50 values).
- Cell-dependent assays : Quantify exosome suppression in HCT116 cells (e.g., 15 µM this compound reduces exosome release without cytotoxicity) .
- Structural dynamics : RMSD plots confirm stable this compound–nSMase2 binding . Complementary techniques like Western blotting assess downstream biomarkers (e.g., ceramide levels).
Q. Basic: How are drug-drug interaction (DDI) risks assessed for this compound?
DDI studies focus on CYP3A4-mediated metabolism:
- In vitro assays : Human liver microsomes quantify this compound’s inhibition/induction potential.
- Clinical PK studies : Co-administration with CYP3A4 substrates (e.g., dexamethasone) or inhibitors (e.g., ketoconazole) to measure AUC changes .
- Population PK models : Adjust dosing for patients on concurrent CYP3A4 modulators .
Q. Advanced: How is this compound repurposed for exosome inhibition in cancer therapy?
Repurposing strategies involve:
- Dose-response assays : Test this compound (1–50 µM) in cell lines (e.g., HCT116) to determine IC50 for nSMase2.
- Functional validation : Measure exosome markers (CD63, Alix) via ELISA or nanoparticle tracking analysis .
- In vivo models : Xenograft studies assessing tumor exosome suppression and metastasis .
Q. Basic: How is statistical power determined in this compound trials?
Sample size calculations use the formula:
where p = pooled proportion, α = 0.05, and power (1–β) = 80%. For example, a study with 200 patients per group achieved 95% confidence to detect a 20% efficacy difference .
Q. Advanced: What mechanistic studies validate this compound's anti-inflammatory effects via NF-κB?
In LPS-induced macrophages:
- ROS measurement : Fluorescent probes (e.g., DCFH-DA) quantify oxidative stress suppression.
- Cytokine profiling : ELISA for TNF-α, IL-6, and MCP-1.
- Western blotting : Assess NF-κB pathway proteins (e.g., p65 phosphorylation, IκBα degradation) .
Q. Basic: How are animal models standardized for this compound reproducibility?
- Species selection : Ferrets for emesis studies (NK1 receptor homology); rodents for PK/PD.
- Dosing consistency : Fixed mg/kg ratios (e.g., 3 mg/kg in ferrets).
- Endpoint alignment : Emesis frequency, brain/plasma ratios, and receptor occupancy (autoradiography in gerbils) .
Q. Advanced: How are structure-activity relationship (SAR) studies conducted for this compound analogues?
Properties
IUPAC Name |
3-[[(2R,3S)-2-[(1R)-1-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)morpholin-4-yl]methyl]-1,4-dihydro-1,2,4-triazol-5-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C23H21F7N4O3/c1-12(14-8-15(22(25,26)27)10-16(9-14)23(28,29)30)37-20-19(13-2-4-17(24)5-3-13)34(6-7-36-20)11-18-31-21(35)33-32-18/h2-5,8-10,12,19-20H,6-7,11H2,1H3,(H2,31,32,33,35)/t12-,19+,20-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
ATALOFNDEOCMKK-OITMNORJSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C1=CC(=CC(=C1)C(F)(F)F)C(F)(F)F)OC2C(N(CCO2)CC3=NNC(=O)N3)C4=CC=C(C=C4)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@H](C1=CC(=CC(=C1)C(F)(F)F)C(F)(F)F)O[C@@H]2[C@@H](N(CCO2)CC3=NNC(=O)N3)C4=CC=C(C=C4)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C23H21F7N4O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3049047 | |
| Record name | Aprepitant | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3049047 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
534.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Aprepitant | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014811 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Practically insoluble, 1.94e-02 g/L | |
| Record name | Aprepitant | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00673 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Aprepitant | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014811 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Aprepitant has been shown in animal models to inhibit emesis induced by cytotoxic chemotherapeutic agents, such as cisplatin, via central actions. Animal and human Positron Emission Tomography (PET) studies with Aprepitant have shown that it crosses the blood brain barrier and occupies brain NK1 receptors. Animal and human studies show that Aprepitant augments the antiemetic activity of the 5-HT3-receptor antagonist ondansetron and the corticosteroid ethasone and inhibits both the acute and delayed phases of cisplatin induced emesis. | |
| Record name | Aprepitant | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00673 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
170729-80-3 | |
| Record name | Aprepitant | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=170729-80-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Aprepitant [USAN:INN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0170729803 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Aprepitant | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00673 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Aprepitant | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=748825 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Aprepitant | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3049047 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Aprepitant | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | APREPITANT | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/1NF15YR6UY | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Aprepitant | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014811 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















