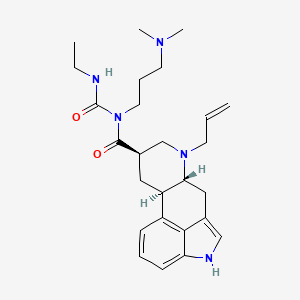
Cabergoline
Overview
Description
Cabergoline is a dopaminergic medication primarily used to treat conditions associated with high levels of prolactin, such as prolactinomas and hyperprolactinemia. It is also used in the management of Parkinson’s disease and other disorders. This compound is an ergot derivative and acts as a potent dopamine D2 receptor agonist .
Preparation Methods
Cabergoline is synthesized from ergoline-8β-carboxylic acid ester through a series of chemical reactions. The process involves protecting the secondary amine and the indole nitrogen functions of ergoline-8β-carboxylic acid C1-4 alkyl esters as carbamate derivatives. The protected compound is then amidated with 3-(dimethylamino)propylamine, reacted with ethyl isocyanate, and the protecting groups are cleaved. Finally, the deprotected secondary amine is reacted with an electrophilic allyl alcohol derivative to obtain this compound .
Chemical Reactions Analysis
Cabergoline undergoes various chemical reactions, including hydrolysis and oxidation. It is highly sensitive to hydrolysis, particularly at the urea moiety and amide group. The alkene bond in this compound is susceptible to oxidation. Common reagents used in these reactions include water for hydrolysis and oxidizing agents for oxidation. The major products formed from these reactions are degradation products identified using infrared and mass spectrometry analyses.
Scientific Research Applications
Treatment of Hyperprolactinemia
Overview : Cabergoline is the first-line treatment for hyperprolactinemia, effectively normalizing prolactin levels in a significant percentage of patients.
Key Findings :
- In a study involving 455 patients, this compound normalized serum prolactin levels in 86% of cases. Specifically, normalization was achieved in 92% of patients with idiopathic hyperprolactinemia or microprolactinoma and 77% in those with macroadenomas .
- Tumor shrinkage was observed in 67% of patients, while visual field abnormalities improved in 70% .
Treatment of Pituitary Adenomas
Overview : this compound not only suppresses hormone production but also induces tumor shrinkage in prolactinomas.
Research Insights :
- Recent studies indicate that this compound may suppress tumor cell proliferation and induce apoptosis through novel mechanisms. This suggests potential applications in treating other tumors beyond pituitary adenomas, including breast cancer and pancreatic neuroendocrine tumors .
Acromegaly Management
Overview : this compound is also utilized in managing acromegaly, particularly in patients who have not undergone radiation therapy.
Clinical Efficacy :
- In non-irradiated patients, this compound normalized insulin-like growth factor 1 (IGF-1) levels in approximately 32% of cases. However, biochemical control (normal IGF-1 and growth hormone levels) was achieved in only 13% .
Potential Use in Other Tumors
Recent research has explored this compound's broader applications in oncology:
- Breast Cancer : Studies suggest that this compound may have anti-tumor effects on breast cancer cells, potentially offering a new therapeutic avenue for treatment .
- Neuroendocrine Tumors : Its efficacy has been investigated for pancreatic neuroendocrine tumors, indicating possible benefits in tumor management .
Case Studies and Clinical Trials
Several case studies illustrate the efficacy and safety profile of this compound:
| Study Type | Patient Population | Outcome |
|---|---|---|
| Retrospective Cohort Study | 455 patients with hyperprolactinemia | 86% normalization of prolactin levels |
| Clinical Trial | Non-irradiated acromegaly patients | 32% normalization of IGF-1 levels |
| Case Series | Patients with breast cancer | Indications of tumor growth suppression |
Mechanism of Action
Cabergoline exerts its effects by stimulating dopamine D2 receptors, which are G-protein coupled receptors associated with Gi proteins. In lactotrophs, stimulation of dopamine D2 receptors inhibits adenylyl cyclase, decreasing intracellular cyclic adenosine monophosphate (cAMP) concentrations and blocking inositol triphosphate (IP3)-dependent release of calcium from intracellular stores. This results in the inhibition of prolactin secretion .
Comparison with Similar Compounds
Cabergoline is often compared with other dopamine agonists such as bromocriptine and pergolide. While all three compounds are used to treat hyperprolactinemia and Parkinson’s disease, this compound has a longer half-life and higher affinity for dopamine D2 receptors, making it more effective and better tolerated by patients . Bromocriptine and pergolide have shorter half-lives and may require more frequent dosing .
Similar Compounds
- Bromocriptine
- Pergolide
- Quinagolide
This compound’s unique properties, such as its long half-life and high receptor affinity, make it a preferred choice for many patients and healthcare providers.
Biological Activity
Cabergoline is a long-acting dopamine agonist primarily known for its efficacy in treating hyperprolactinemia and related disorders. This article explores the biological activity of this compound, focusing on its mechanisms of action, therapeutic applications, and recent research findings.
This compound acts predominantly as an agonist at the dopamine D2 receptors, which are crucial for regulating prolactin secretion from the anterior pituitary gland. The drug's high affinity for D2 receptors enables it to effectively inhibit prolactin release, thus normalizing serum prolactin levels in various clinical settings.
- Dopamine Receptor Binding : this compound exhibits a low affinity for other receptors such as D1, α1- and α2-adrenergic, and serotonin receptors (5-HT1 and 5-HT2), making its action relatively selective for D2 receptors .
- Inhibition of Prolactin Secretion : In vitro studies demonstrate that this compound directly inhibits prolactin secretion by lactotrophs in the pituitary gland, contributing to its therapeutic effects in hyperprolactinemia .
Therapeutic Applications
This compound is utilized in various clinical contexts, including:
-
Hyperprolactinemia :
- A retrospective study involving 455 patients showed that this compound normalized prolactin levels in 86% of cases, with significant improvements noted in visual field abnormalities and tumor shrinkage .
- The median starting dose was 1.0 mg/week, which could be reduced to 0.5 mg/week upon achieving control .
- Acromegaly :
- Cushing's Disease :
Neuroprotective Effects
Recent research suggests that this compound may also possess neuroprotective properties:
- A study indicated that this compound prevents oxidative stress-induced neuronal cell death by reducing extracellular glutamate accumulation and inhibiting ERK1/2 signaling pathways . This suggests potential applications beyond endocrine disorders, possibly extending into neurodegenerative conditions.
Case Studies and Research Findings
Q & A
Basic Research Questions
Q. What experimental models are appropriate for investigating Cabergoline’s dopamine receptor selectivity and mechanism of action?
- Methodological Answer : Utilize in vitro competitive binding assays with radiolabeled dopamine receptors (D2 and D5 subtypes) to measure this compound’s affinity and intrinsic activity. Dose-response curves can quantify receptor activation thresholds. In vivo models (e.g., rodent prolactinoma) should measure serum prolactin suppression as a functional endpoint. Ensure assays include controls for cross-reactivity with other receptors (e.g., serotonin) to confirm specificity .
Q. How can researchers determine the optimal this compound dose for prolactin suppression in hyperprolactinemia?
- Methodological Answer : Conduct dose-escalation studies in animal models or clinical cohorts, measuring serum prolactin levels weekly. Use nonlinear mixed-effects modeling (NONMEM) to correlate dose with hormonal response, adjusting for covariates like body weight and baseline prolactin. Validate findings with pharmacokinetic/pharmacodynamic (PK/PD) simulations to identify minimal effective and maximal tolerated doses .
Q. What biomarkers are validated for monitoring this compound’s therapeutic efficacy in endocrine disorders?
- Methodological Answer : IGF-I normalization (for acromegaly) and prolactin suppression (for hyperprolactinemia) are primary biomarkers. Secondary endpoints include tumor volume reduction (MRI) and patient-reported outcomes (e.g., headache resolution). Ensure assays meet CLIA/CAP certification standards to minimize variability .
Advanced Research Questions
Q. How can researchers reconcile contradictory findings on this compound’s association with valvular heart disease?
- Methodological Answer : Apply multivariate Cox regression in large-scale cohort studies to adjust for confounders (e.g., age, hypertension, cumulative dose). Stratify analyses by dose thresholds (e.g., <3 mg/week vs. ≥3 mg/week) and validate echocardiographic endpoints (e.g., valve thickening) with blinded adjudication. Meta-analyses should assess publication bias via funnel plots and Egger’s test .
Q. What statistical approaches are suitable for analyzing this compound’s synergistic effects with somatostatin analogs in acromegaly?
- Methodological Answer : Use interaction terms in mixed-effects regression models to evaluate additive vs. synergistic effects on IGF-I suppression. Individual patient data (IPD) meta-analyses can pool data from heterogeneous studies, adjusting for baseline IGF-I, tumor size, and prolactin levels. Sensitivity analyses should exclude outliers to ensure robustness .
Q. How to design longitudinal studies evaluating this compound’s long-term neuroendocrine and cardiovascular outcomes?
- Methodological Answer : Implement a prospective, matched cohort design with annual echocardiography and hormonal profiling. Use time-dependent covariates in survival analysis to account for cumulative exposure. Power calculations should assume a 5% annual incidence of subclinical valvulopathy, requiring ≥500 patient-years of follow-up .
Q. Methodological Considerations
- Contradiction Analysis : When studies report conflicting results (e.g., valvulopathy risk), perform subgroup analyses by dose, duration, and population characteristics. Use the Bradford Hill criteria to assess causality .
- Research Design : For preclinical studies, adhere to ARRIVE guidelines for experimental rigor. In clinical research, prioritize randomized adaptive trials for dose-finding and PROBE designs for long-term safety .
Properties
IUPAC Name |
(6aR,9R,10aR)-N-[3-(dimethylamino)propyl]-N-(ethylcarbamoyl)-7-prop-2-enyl-6,6a,8,9,10,10a-hexahydro-4H-indolo[4,3-fg]quinoline-9-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C26H37N5O2/c1-5-11-30-17-19(25(32)31(26(33)27-6-2)13-8-12-29(3)4)14-21-20-9-7-10-22-24(20)18(16-28-22)15-23(21)30/h5,7,9-10,16,19,21,23,28H,1,6,8,11-15,17H2,2-4H3,(H,27,33)/t19-,21-,23-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KORNTPPJEAJQIU-KJXAQDMKSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCNC(=O)N(CCCN(C)C)C(=O)C1CC2C(CC3=CNC4=CC=CC2=C34)N(C1)CC=C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCNC(=O)N(CCCN(C)C)C(=O)[C@@H]1C[C@H]2[C@@H](CC3=CNC4=CC=CC2=C34)N(C1)CC=C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C26H37N5O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
85329-89-1 (diphosphate) | |
| Record name | Cabergoline [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0081409907 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID6022719 | |
| Record name | Cabergoline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6022719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
451.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Insoluble, 6.40e-02 g/L | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The dopamine D2 receptor is a 7-transmembrane G-protein coupled receptor associated with Gi proteins. In lactotrophs, stimulation of dopamine D2 causes inhibition of adenylyl cyclase, which decreases intracellular cAMP concentrations and blocks IP3-dependent release of Ca2+ from intracellular stores. Decreases in intracellular calcium levels may also be brought about via inhibition of calcium influx through voltage-gated calcium channels, rather than via inhibition of adenylyl cyclase. Additionally, receptor activation blocks phosphorylation of p42/p44 MAPK and decreases MAPK/ERK kinase phosphorylation. Inhibition of MAPK appears to be mediated by c-Raf and B-Raf-dependent inhibition of MAPK/ERK kinase. Dopamine-stimulated growth hormone release from the pituitary gland is mediated by a decrease in intracellular calcium influx through voltage-gated calcium channels rather than via adenylyl cyclase inhibition. Stimulation of dopamine D2 receptors in the nigrostriatal pathway leads to improvements in coordinated muscle activity in those with movement disorders. Cabergoline is a long-acting dopamine receptor agonist with a high affinity for D2 receptors. Receptor-binding studies indicate that cabergoline has low affinity for dopamine D1, α1,- and α2- adrenergic, and 5-HT1- and 5-HT2-serotonin receptors. | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
81409-90-7 | |
| Record name | Cabergoline | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=81409-90-7 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cabergoline [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0081409907 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID6022719 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (6aR,9R,10aR)-N-[3-(dimethylamino)propyl]-N-(ethylcarbamoyl)-7-prop-2-enyl-6,6a,8,9,10,10a-hexahydro-4H-indolo[4,3-fg]quinoline-9-carboxamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CABERGOLINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/LL60K9J05T | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
102-104 °C, 102 - 104 °C | |
| Record name | Cabergoline | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00248 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Cabergoline | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















