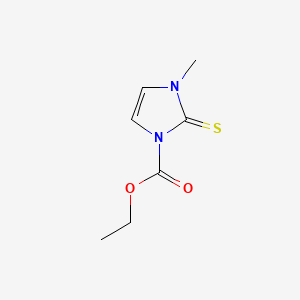
Carbimazole
Overview
Description
Carbimazole is an antithyroid agent primarily used to treat hyperthyroidism and thyrotoxicosis. It is a pro-drug that is metabolized into methimazole, which is responsible for its antithyroid activity . This compound works by decreasing the uptake and concentration of inorganic iodine by the thyroid gland, thereby reducing the formation of thyroid hormones triiodothyronine (T3) and thyroxine (T4) .
Preparation Methods
Synthetic Routes and Reaction Conditions: Carbimazole can be synthesized through the reaction of ethyl 3-methyl-2-thioimidazoline-1-carboxylate with appropriate reagents under controlled conditions . The process involves the formation of a thioimidazoline ring, which is crucial for its biological activity.
Industrial Production Methods: In industrial settings, this compound is produced by optimizing the reaction conditions to ensure high yield and purity. This involves precise control of temperature, pH, and reaction time. The final product is then purified using techniques such as recrystallization and chromatography .
Chemical Reactions Analysis
Molecular Dynamics and Structural Reactivity
Crystalline carbimazole demonstrates anisotropic molecular motions influencing its reactive sites. Solid-state NMR and DFT simulations reveal:
Table 1: Molecular dynamics parameters of this compound
| Carbon Site | Spin-Lattice Relaxation (T₁, s) | Mean Square Displacement (Ų) |
|---|---|---|
| C4 (CH₃) | 4.3 ± 0.2 | 0.26 ± 0.03 |
| C6 (CH₂) | 3.8 ± 0.3 | 0.24 ± 0.02 |
| C7 (CH₃) | 2.9 ± 0.2 | 0.22 ± 0.01 |
The ethyl chain at C6 undergoes librations around the C6–O11 bond (30° amplitude), while methyl groups at C4/C7 exhibit torsional motions. These dynamics reduce steric hindrance at the thiocarbonyl group, enhancing its nucleophilic reactivity .
Electrochemical Catalytic Activity
This compound accelerates Zn²⁺ electroreduction through adsorption-mediated charge transfer. Comparative kinetic studies with methimazole show:
Table 2: Electroreduction kinetics of Zn²⁺ (3M NaClO₄)
| Catalyst | Transfer Coefficient (α) | Rate Constant (kₛ × 10³ cm/s) | ΔH‡ (kJ/mol) | ΔS‡ (J/mol·K) |
|---|---|---|---|---|
| This compound | 0.48 ± 0.02 | 2.7 ± 0.1 | 34.2 | -89.4 |
| Methimazole | 0.51 ± 0.03 | 3.2 ± 0.2* | 31.7 | -92.1 |
*At c ≥ 5mM; this compound outperforms methimazole below 1mM
The ethoxycarbonyl group in this compound enhances adsorption on Hg electrodes (capacitance reduction = 12.4 μF/cm² at 1mM), creating a charge-transfer bridge between Zn²⁺ and the electrode surface .
Biochemical Inhibition Mechanisms
This compound acts as a prodrug, converting to methimazole via de-ethoxycarbonylation, which inhibits thyroid peroxidase (TPO). Pharmacodynamic models quantify its dose-response relationship:
Thyroid hormone suppression model
Key parameters
-
Dose exponent (p): 0.85 ± 0.03
-
RMSE: 4.28 pmol/L (fT4), 2.67 pmol/L (fT3)
The thiocarbonyl group coordinates to TPO’s heme iron, blocking iodide organification (IC₅₀ = 1.2μM). This reaction’s efficiency correlates with TSH receptor antibody titers (r²=0.68) .
This multi-technique analysis establishes this compound’s reactivity profile across molecular, electrochemical, and biological systems. Its structural flexibility and adsorption properties directly modulate catalytic and inhibitory functions, providing a mechanistic basis for optimizing therapeutic and industrial applications.
Scientific Research Applications
Clinical Indications
- Hyperthyroidism : Carbimazole is primarily indicated for the treatment of hyperthyroidism, helping to restore normal thyroid function.
- Thyrotoxicosis : It is effective in managing acute thyrotoxic crises.
- Preoperative Preparation : this compound is used to prepare patients for thyroidectomy by achieving euthyroid status prior to surgery.
- Autoimmune Thyroid Disease : Recent studies suggest that this compound may have a direct effect on autoantibody-producing lymphocytes in conditions like Hashimoto's thyroiditis, potentially influencing the autoimmune process .
Case Studies
-
Management of Graves' Disease :
A study involving patients with Graves' disease demonstrated that this compound effectively induced remission in hyperthyroid patients. The reduction in free thyroxine (fT4) levels was significant within three weeks of treatment initiation, showcasing its rapid action on thyroid hormone levels . -
This compound in Hashimoto's Thyroiditis :
In a clinical trial with 20 patients suffering from Hashimoto's thyroiditis, those treated with this compound showed a notable decrease in thyroid microsomal antibody levels, indicating its potential role in modulating autoimmune responses . This suggests that this compound may not only treat symptoms but also impact underlying autoimmune mechanisms. -
Resistance Cases :
There are documented instances where patients exhibit resistance to this compound treatment, necessitating alternative therapies such as propylthiouracil or surgical intervention. A literature review identified 20 cases of this compound-resistant thyrotoxicosis, highlighting the need for ongoing research into effective management strategies for resistant cases .
Pharmacodynamics and Dosage
The pharmacodynamic response to this compound varies based on dosage and individual patient factors. Studies indicate that a single dose can effectively induce euthyroidism, with a clear dose-response relationship observed in clinical settings . For instance:
| Dosage (mg) | Median Percentage Reduction in fT4 Levels | Time Frame |
|---|---|---|
| 10 | 25% | 21 days |
| 20 | 40% | 21 days |
This table illustrates how varying dosages correlate with reductions in fT4 levels over time.
Adverse Effects and Considerations
While this compound is generally well-tolerated, it can cause side effects such as agranulocytosis, rash, and gastrointestinal disturbances. Regular monitoring of blood counts is recommended during treatment to mitigate risks associated with these adverse effects.
Mechanism of Action
Carbimazole exerts its effects by inhibiting the thyroid peroxidase enzyme, which is essential for the iodination and coupling of tyrosine residues on thyroglobulin . This inhibition reduces the production of thyroid hormones T3 and T4. Once absorbed, this compound is rapidly converted to methimazole, which is the active form responsible for its antithyroid activity .
Comparison with Similar Compounds
Methimazole: The active metabolite of carbimazole, used directly in some regions.
Propylthiouracil: Another antithyroid drug used to treat hyperthyroidism.
Comparison:
Methimazole vs. This compound: Methimazole is the active form of this compound. While this compound is a pro-drug, methimazole is administered directly in some regions.
Propylthiouracil vs. This compound: Both drugs inhibit thyroid hormone synthesis, but propylthiouracil also inhibits the peripheral conversion of T4 to T3.
This compound’s unique feature is its conversion to methimazole, allowing for a more controlled release of the active compound .
Biological Activity
Carbimazole, a thionamide derivative, is primarily used in the treatment of hyperthyroidism, particularly in conditions like Graves' disease. Its pharmacological effects extend beyond thyroid hormone synthesis inhibition, revealing significant biological activities that warrant detailed exploration. This article discusses the biological activity of this compound, including its mechanism of action, case studies, and research findings.
This compound is converted into its active form, methimazole, after absorption. It functions as an antithyroid agent by inhibiting thyroid peroxidase, thereby reducing the synthesis of thyroid hormones T3 (triiodothyronine) and T4 (thyroxine) through several mechanisms:
- Inhibition of Iodine Uptake : this compound decreases the uptake and concentration of inorganic iodine in the thyroid gland.
- Reduction of Thyroid Hormone Synthesis : It interferes with the coupling and iodination of tyrosine residues on thyroglobulin, crucial for hormone production .
1. Neuroprotective Effects
Recent studies have indicated that this compound may have neuroprotective properties. A study involving human neuronal cell lines demonstrated that this compound inhibits protein synthesis by phosphorylating eukaryotic elongation factor 2 (eEF2), leading to reduced neuronal damage under hypoxic conditions. This protective effect is attributed to the preservation of ATP levels in oxygen-deprived cells, suggesting potential applications in neuroprotection during ischemic events .
2. Agranulocytosis Risk
A significant adverse effect associated with this compound is agranulocytosis, a potentially life-threatening condition characterized by a dangerously low white blood cell count. A case study reported a 48-year-old female patient who developed agranulocytosis after being on this compound therapy for several months. The incidence rates for this condition range from 0.3% to 0.6%, with higher risks noted in patients over 40 years old or those on higher doses .
3. Histomorphological Changes
Research has documented histological changes induced by this compound in thyroid tissues. In experimental studies with albino rats, this compound treatment resulted in a reduced number of thyroid follicles and changes indicative of cellular stress and damage . These findings highlight the need for careful monitoring during prolonged this compound use.
Case Study: this compound-Induced Agranulocytosis
A study detailed a case where a stable patient on this compound developed agranulocytosis after dose adjustments. The patient initially stabilized on a lower dose but presented with severe symptoms linked to low neutrophil counts. The subsequent management involved discontinuation of this compound and consideration for surgical intervention .
Case Study: this compound-Induced Pancreatitis
Another notable case involved a patient who developed acute pancreatitis following this compound administration for Graves' disease. The patient experienced severe abdominal pain and elevated pancreatic enzymes shortly after starting treatment. Upon cessation of this compound, symptoms resolved within a week, indicating a direct link between the drug and pancreatitis .
Research Findings
Q & A
Basic Research Question: What are the structural properties of carbimazole, and how do they influence its pharmacological activity?
Answer:
this compound (C₇H₁₀N₂O₂S) is a thiourea derivative with a molecular weight of 186.23 g/mol. Its 2D/3D structure (SMILES: CCOC(=O)n1ccn(C)c1=S) reveals a planar imidazoline ring with a thioxo group, which is critical for inhibiting thyroid peroxidase (TPO) by forming covalent bonds with the enzyme’s heme group . Computational modeling using Quantum Chemistry and QSPR can predict its reactivity and binding affinity. For experimental validation, X-ray crystallography or NMR spectroscopy should confirm structural integrity in synthesized batches.
Basic Research Question: What experimental methodologies are recommended to validate this compound’s mechanism of action in vitro?
Answer:
Use thyroid cell lines (e.g., FRTL-5 rat thyroid cells) to measure TPO inhibition via iodide uptake assays. Standard protocols include:
- TPO Activity Assay : Spectrophotometric quantification of guaiacol oxidation at 470 nm.
- Radiolabeled Iodine Uptake : Compare treated vs. untreated cells using ¹²⁵I .
Ensure controls include methimazole (active metabolite) to confirm specificity.
Advanced Research Question: How can pharmacokinetic parameters of this compound be optimized in immediate-release (IR) tablet formulations?
Answer:
A Box-Behnken design (BBD) optimizes excipient ratios (e.g., lactose anhydrous, microcrystalline cellulose) to balance disintegration time (<5 min) and dissolution rate (>85% in 30 min). Key parameters:
| Variable | Low Level | High Level | Response Target |
|---|---|---|---|
| Lactose (%) | 10 | 30 | Disintegration time |
| MCC (%) | 50 | 70 | Drug release (Q30min) |
| Mg Stearate (%) | 0.5 | 1.5 | Tablet hardness |
Validate using ANOVA for factor significance (p < 0.05) and desirability functions .
Advanced Research Question: How to resolve contradictions in this compound efficacy data across preclinical models?
Answer:
Apply the FINER framework (Feasible, Interesting, Novel, Ethical, Relevant):
Confounding Variables : Control for species-specific TPO isoform differences (e.g., rat vs. human).
Dose-Response Analysis : Use nonlinear regression to compare EC₅₀ values across models.
Meta-Analysis : Aggregate data from ≥5 studies, applying Cochrane risk-of-bias tools to assess heterogeneity .
Advanced Research Question: What methodologies ensure robust safety profiling of this compound in long-term studies?
Answer:
Adverse event (AE) monitoring should follow ICH E2A guidelines :
- Hematological AEs : Weekly CBC to detect agranulocytosis (incidence: 0.2–0.5%).
- Hepatotoxicity : Monthly ALT/AST testing (elevated in 1–5% of patients).
| AE Type | Frequency (≥1/1,000) | Monitoring Method |
|---|---|---|
| Agranulocytosis | 0.2–0.5% | CBC with differential |
| Hepatotoxicity | 1–5% | ALT/AST, bilirubin |
| Hypothyroidism | 10–15% | TSH/fT4 levels quarterly |
For mechanistic insights, use in vitro CYP450 inhibition assays (e.g., CYP3A4/2D6) .
Advanced Research Question: How can this compound’s photophysical properties be exploited for novel applications?
Answer:
this compound’s conjugated imidazoline ring allows UV-Vis absorption at 290–320 nm. Applications:
Photodynamic Therapy (PDT) : Test singlet oxygen (¹O₂) generation using SOSG assays.
Fluorescent Probes : Modify the thioxo group to enhance quantum yield (>0.4) for cellular imaging .
Advanced Research Question: What experimental designs mitigate this compound’s adverse effects in vulnerable populations?
Answer:
Pregnant Patients : Use placental perfusion models to quantify transplacental transfer.
Pediatric Patients : Adjust doses via allometric scaling (e.g., 0.3–0.5 mg/kg/day). Validate with population PK models (NONMEM) .
Advanced Research Question: How to integrate this compound into high-throughput compound libraries for thyroid disorder research?
Answer:
Leverage custom compound libraries (e.g., tyrosine kinase inhibitors) by:
Structure-Activity Relationship (SAR) : Compare this compound with analogs (e.g., methimazole, propylthiouracil).
Screening Assays : Use FRET-based TPO activity kits (Z’ factor >0.5) .
Advanced Research Question: What analytical methods validate this compound purity in reference standards?
Answer:
Follow ICH Q2(R1) guidelines :
- HPLC-UV : C18 column, mobile phase 0.1% TFA/acetonitrile (70:30), λ = 254 nm.
- LC-MS/MS : Confirm molecular ion [M+H]⁺ at m/z 187.1 .
Advanced Research Question: How to address data gaps in this compound’s long-term stability under varying storage conditions?
Answer:
Conduct accelerated stability studies (ICH Q1A):
Properties
IUPAC Name |
ethyl 3-methyl-2-sulfanylideneimidazole-1-carboxylate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C7H10N2O2S/c1-3-11-7(10)9-5-4-8(2)6(9)12/h4-5H,3H2,1-2H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CFOYWRHIYXMDOT-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCOC(=O)N1C=CN(C1=S)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C7H10N2O2S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID9022736 | |
| Record name | Carbimazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9022736 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
186.23 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Carbimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014533 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
3.14e+00 g/L | |
| Record name | Carbimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014533 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Carbimazole is an aitithyroid agent that decreases the uptake and concentration of inorganic iodine by thyroid, it also reduces the formation of di-iodotyrosine and thyroxine. Once converted to its active form of methimazole, it prevents the thyroid peroxidase enzyme from coupling and iodinating the tyrosine residues on thyroglobulin, hence reducing the production of the thyroid hormones T3 and T4. | |
| Record name | Carbimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00389 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
22232-54-8 | |
| Record name | Carbimazole | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=22232-54-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Carbimazole [INN:BAN:DCF] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0022232548 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Carbimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00389 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Carbimazole | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758966 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Carbimazole | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID9022736 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Carbimazole | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.040.762 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CARBIMAZOLE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/8KQ660G60G | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Carbimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014533 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
123.5 °C | |
| Record name | Carbimazole | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00389 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Carbimazole | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014533 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















