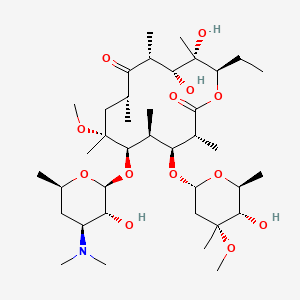
Clarithromycin
Overview
Description
Clarithromycin is a macrolide antibiotic derived from erythromycin. It is widely used to treat various bacterial infections, including strep throat, pneumonia, skin infections, Helicobacter pylori infection, and Lyme disease . This compound works by inhibiting bacterial protein synthesis, making it an effective treatment for a broad range of bacterial infections .
Preparation Methods
Clarithromycin is synthesized from erythromycin through a series of chemical reactions. The process involves oximation, etherification, silanization, methylation, and reduction hydrolysis . The industrial production of this compound typically involves the following steps:
Oximation Reaction: Erythromycin thiocyanate undergoes an oximation reaction.
Etherification Reaction: The oxime is then etherified.
Silanization Reaction: The etherified product is subjected to silanization.
Methylation Reaction: The silanized product undergoes methylation.
Reduction Hydrolysis: Finally, the product is reduced and hydrolyzed to obtain this compound.
Chemical Reactions Analysis
Clarithromycin undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under specific conditions.
Reduction: Reduction reactions are part of its synthesis process.
Substitution: Substitution reactions can occur, particularly during its synthesis.
Common reagents used in these reactions include organic solvents, acids, and bases. The major products formed from these reactions are intermediates that lead to the final product, this compound .
Scientific Research Applications
Antibacterial Applications
Clarithromycin's Role in Treating Bacterial Infections:
this compound is primarily prescribed for treating a variety of bacterial infections, including:
- Respiratory Tract Infections: Effective against pathogens causing pneumonia and bronchitis.
- Skin Infections: Used for treating skin and soft tissue infections.
- Helicobacter pylori Eradication: Often part of combination therapy to eradicate H. pylori in peptic ulcer disease.
Table 1: Common Indications for this compound Use
| Indication | Pathogen/Condition | Dosage Form |
|---|---|---|
| Community-acquired pneumonia | Streptococcus pneumoniae | Oral: 500 mg |
| Acute exacerbation of bronchitis | Haemophilus influenzae | Oral: 500 mg |
| Skin and soft tissue infections | Staphylococcus aureus | Oral: 500 mg |
| H. pylori eradication | H. pylori | Oral: 500 mg (part of triple therapy) |
Oncological Applications
Recent studies have indicated that this compound may have antineoplastic properties, particularly in hematologic malignancies.
Mechanisms of Action:
- Immunomodulatory Effects: this compound has shown the ability to modulate immune responses by affecting cytokine production, particularly interleukin-6 and tumor necrosis factor-alpha, which are crucial in cancer progression.
- Combination Therapies: It has been successfully used in combination with other chemotherapeutic agents to enhance efficacy against multiple myeloma and non-small cell lung cancer.
Case Study: Multiple Myeloma Treatment
A study demonstrated that this compound combined with standard chemotherapy improved patient outcomes by reducing myeloma growth factors and enhancing natural killer cell activity .
Table 2: Clinical Studies on this compound in Oncology
| Study Reference | Cancer Type | Findings |
|---|---|---|
| Hamada et al. (1995) | Various Tumors | Induction of cytotoxic macrophages |
| Mikasa et al. | Non-small cell lung cancer | Increased median survival time |
| Sakamoto et al. | Non-small cell lung cancer | Enhanced NK cell activity post-treatment |
Respiratory Conditions
This compound's anti-inflammatory properties make it beneficial in treating chronic respiratory conditions.
Applications in Asthma and COPD:
- Asthma Management: It has been shown to reduce airway inflammation and improve lung function when used alongside standard asthma medications.
- Chronic Obstructive Pulmonary Disease (COPD): this compound may reduce exacerbations by modulating inflammatory cytokines.
Table 3: Efficacy of this compound in Respiratory Conditions
| Condition | Study Reference | Outcome |
|---|---|---|
| Chronic Asthma | Kraft et al. | Reduced TNF-alpha mRNA levels |
| COPD Exacerbations | Zeng et al. | Decreased frequency of exacerbations |
Emerging Applications
COVID-19 Research:
this compound has been investigated for its potential role in treating COVID-19 pneumonia due to its anti-inflammatory properties and effects on viral replication pathways . The CAME COVID study aimed to evaluate its efficacy in patients with mild pneumonia not requiring oxygen support.
Neuronal Effects:
Recent findings suggest this compound may increase neuronal excitability, indicating potential applications in neurological recovery processes such as emergence from anesthesia or treatment of certain cognitive impairments .
Mechanism of Action
Clarithromycin exerts its effects by binding to the bacterial 50S ribosomal subunit. This binding inhibits peptidyl transferase activity and interferes with amino acid translocation during the translation and protein assembly process. As a result, bacterial protein synthesis is inhibited, leading to a decrease in bacterial growth and ultimately helping the body’s immune system to eliminate the infection .
Comparison with Similar Compounds
Clarithromycin is similar to other macrolide antibiotics such as erythromycin and azithromycin. it has unique properties that make it distinct:
Erythromycin: This compound is a 6-O-methyl derivative of erythromycin, which enhances its stability and bioavailability.
Azithromycin: While both are macrolides, this compound has a broader spectrum of activity and is more effective against certain bacterial strains.
Similar compounds include:
This compound’s unique chemical structure and enhanced stability make it a valuable antibiotic in the treatment of various bacterial infections.
Biological Activity
Clarithromycin is a macrolide antibiotic widely used for treating various bacterial infections. It is known for its effective antimicrobial properties, particularly against Gram-positive bacteria. This article delves into the biological activity of this compound, exploring its mechanisms of action, pharmacokinetics, clinical efficacy, and safety profile.
This compound exerts its antibacterial effects primarily by inhibiting bacterial protein synthesis. It achieves this by binding to the 23S rRNA component of the 50S ribosomal subunit, effectively blocking the translocation of aminoacyl-tRNA and halting polypeptide synthesis. This action is similar to that of erythromycin but demonstrates greater potency against certain pathogens.
- Metabolism : this compound is metabolized in the liver to 14-hydroxy this compound, an active metabolite that enhances its antimicrobial activity. Both compounds work synergistically against susceptible organisms .
Pharmacokinetics
This compound is well-absorbed when taken orally and exhibits good tissue penetration, particularly in lung tissues where it achieves concentrations significantly higher than those in serum. The pharmacokinetic profile includes:
- Half-life : Approximately 3 to 7 hours.
- Volume of Distribution : Not specifically quantified but noted for extensive distribution into tissues .
Clinical Efficacy
This compound has been evaluated in several clinical settings, demonstrating effectiveness against a range of bacterial infections. A notable study compared the efficacy of this compound with penicillin V for treating pharyngitis caused by Group A β-haemolytic streptococci (GABHS). The results indicated:
- Clinical Cure Rates : 88.1% for this compound vs. 92.4% for penicillin.
- Bacteriological Eradication Rates : 82.8% for this compound vs. 83.6% for penicillin.
- Adverse Events : Similar incidence rates between both treatment groups, with most events being mild or moderate .
Comparative Potency
In vitro studies have shown that this compound possesses greater potency and a longer post-antibiotic effect (PAE) compared to azithromycin against Streptococcus pneumoniae. The minimum inhibitory concentrations (MICs) indicate that this compound is effective at lower concentrations than many other antibiotics in its class.
| Organism | This compound MIC (µg/mL) | Azithromycin MIC (µg/mL) |
|---|---|---|
| Streptococcus pneumoniae | ≤ 0.25 | 0.5 - 2 |
| Staphylococcus aureus | 0.06 - >32 | >32 |
Safety Profile and Cardiovascular Outcomes
While this compound is generally well-tolerated, there have been concerns regarding its cardiovascular safety profile. Studies have shown mixed results regarding its association with increased cardiovascular events:
- One study indicated a potential reduction in cardiovascular risk among patients treated with this compound over three months; however, another study found no significant difference in event rates compared to placebo among patients undergoing coronary artery bypass grafting .
- A population-based study highlighted an increased risk of myocardial infarction associated with this compound use compared to amoxicillin .
Case Studies and Research Findings
Several case studies have documented the efficacy of this compound in treating various infections:
- Chronic Obstructive Pulmonary Disease (COPD) : A randomized controlled trial demonstrated that patients receiving this compound had fewer exacerbations compared to those on placebo.
- Helicobacter pylori Eradication : this compound is commonly used in combination therapy for H. pylori eradication, showing high success rates when paired with other antibiotics and proton pump inhibitors.
Q & A
Basic Research Questions
Q. What validated bioanalytical methods are recommended for quantifying clarithromycin in human plasma, and how do they ensure regulatory compliance?
- Methodological Answer : Ultra-performance liquid chromatography tandem mass spectrometry (UPLC-MS/MS) and LCMS/MS are widely validated for this compound quantification, achieving sensitivity limits of 0.1–5 ng/mL. Key validation parameters per FDA (2018) and EMA (2012) guidelines include linearity (R² > 0.99), precision (CV < 15%), and accuracy (85–115% recovery). Internal standards like deuterated this compound improve reproducibility .
Q. What in vitro models are optimal for assessing this compound’s antibacterial efficacy against atypical pathogens like Mycobacterium avium complex (MAC)?
- Methodological Answer : Broth microdilution (MIC90 determination) and time-kill assays under acidic pH (mimicking macrophage phagolysosomes) are standard. Synergy testing with ethambutol or rifabutin uses fractional inhibitory concentration indices (FICI ≤ 0.5 indicates synergy). Ensure nutrient-rich media (e.g., Middlebrook 7H9) for MAC growth .
Q. How should researchers design pharmacokinetic (PK) studies to account for this compound’s pH-dependent solubility and variable bioavailability?
- Methodological Answer : Use crossover designs with controlled gastric pH (e.g., proton-pump inhibitor coadministration) to isolate solubility effects. Population PK modeling with nonlinear mixed-effects (NLME) software (e.g., NONMEM) can quantify covariates like CYP3A4 polymorphisms. Sparse sampling strategies validated by Bayesian estimation reduce patient burden .
Advanced Research Questions
Q. How can contradictory findings on this compound’s modulation of GABAergic signaling (e.g., PET imaging vs. behavioral outcomes) be reconciled methodologically?
- Methodological Answer : Discrepancies may arise from dose-dependent receptor occupancy or indirect modulation. Employ [¹¹C]flumazenil PET to quantify GABAA receptor binding in vivo alongside ex vivo autoradiography in rodent models. Behavioral assays (e.g., rotarod for motor coordination) should use dose-escalation protocols to correlate receptor occupancy with functional outcomes .
Q. What experimental strategies optimize detection of this compound’s off-target effects in transcriptomic studies?
- Methodological Answer : Use RNA sequencing (RNA-seq) with paired-end reads (≥30M per sample) to capture low-abundance transcripts. Pathway enrichment analysis (e.g., DAVID, GSEA) identifies dysregulated pathways (e.g., NF-κB). Validate hits via qPCR with housekeeping genes (e.g., GAPDH, ACTB) and orthogonal methods like CRISPR interference .
Q. How can population pharmacokinetic (PopPK) modeling address this compound’s variability in critically ill patients with renal/hepatic impairment?
- Methodological Answer : Integrate covariates (e.g., eGFR, albumin levels) into a PopPK model using nonlinear mixed-effects modeling. Monte Carlo simulations (10,000 iterations) predict probability of target attainment (PTA) for AUC/MIC ratios >25. Apply Bayesian forecasting with therapeutic drug monitoring (TDM) to individualize dosing .
Q. Data Analysis & Contradiction Resolution
Q. What statistical approaches resolve conflicting reports on this compound’s anti-inflammatory effects in chronic pulmonary diseases?
- Methodological Answer : Meta-regression (random-effects models) can identify confounding variables (e.g., baseline CRP levels, concurrent corticosteroids). Subgroup analysis stratified by disease severity (e.g., FEV1% predicted) and trial duration (>6 months vs. shorter) clarifies effect heterogeneity. Sensitivity analysis excludes studies with high risk of bias (Cochrane RoB 2.0 tool) .
Q. How should researchers validate this compound’s proposed role in mitigating antibiotic resistance via efflux pump inhibition?
- Methodological Answer : Use in vitro models with overexpression of macrolide efflux pumps (e.g., mefA in Streptococcus pneumoniae). Compare MIC values with/without efflux inhibitors (e.g., reserpine). Transcriptomic analysis (RT-qPCR) quantifies pump gene expression. In vivo validation requires murine infection models with bioluminescent pathogens for real-time efficacy monitoring .
Q. Ethical & Methodological Considerations
Q. What ethical safeguards are critical when studying this compound in vulnerable populations (e.g., pediatric or immunocompromised patients)?
- Methodological Answer : Obtain informed consent/assent with age-appropriate documentation. Use centralized TDM to minimize blood draw volumes. Protocol pre-registration (ClinicalTrials.gov ) ensures transparency. Include independent data safety monitoring boards (DSMBs) for interim analyses .
Q. How can researchers mitigate batch-to-batch variability in this compound formulations during preclinical studies?
- Methodological Answer : Source USP-certified reference standards for calibration. Perform HPLC-UV purity checks (≥95%) and quantify 14-(R)-hydroxythis compound impurities (<2%). Use stability-indicating methods (ICH Q1A guidelines) under accelerated conditions (40°C/75% RH) to ensure formulation consistency .
Properties
IUPAC Name |
(3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-6-[(2S,3R,4S,6R)-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-14-ethyl-12,13-dihydroxy-4-[(2R,4R,5S,6S)-5-hydroxy-4-methoxy-4,6-dimethyloxan-2-yl]oxy-7-methoxy-3,5,7,9,11,13-hexamethyl-oxacyclotetradecane-2,10-dione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C38H69NO13/c1-15-26-38(10,45)31(42)21(4)28(40)19(2)17-37(9,47-14)33(52-35-29(41)25(39(11)12)16-20(3)48-35)22(5)30(23(6)34(44)50-26)51-27-18-36(8,46-13)32(43)24(7)49-27/h19-27,29-33,35,41-43,45H,15-18H2,1-14H3/t19-,20-,21+,22+,23-,24+,25+,26-,27+,29-,30+,31-,32+,33-,35+,36-,37-,38-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
AGOYDEPGAOXOCK-KCBOHYOISA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC1C(C(C(C(=O)C(CC(C(C(C(C(C(=O)O1)C)OC2CC(C(C(O2)C)O)(C)OC)C)OC3C(C(CC(O3)C)N(C)C)O)(C)OC)C)C)O)(C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC[C@@H]1[C@@]([C@@H]([C@H](C(=O)[C@@H](C[C@@]([C@@H]([C@H]([C@@H]([C@H](C(=O)O1)C)O[C@H]2C[C@@]([C@H]([C@@H](O2)C)O)(C)OC)C)O[C@H]3[C@@H]([C@H](C[C@H](O3)C)N(C)C)O)(C)OC)C)C)O)(C)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C38H69NO13 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3022829 | |
| Record name | Clarithromycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3022829 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
748.0 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Clarithromycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015342 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
2.17e-01 g/L | |
| Record name | Clarithromycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01211 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Clarithromycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015342 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Clarithromycin is first metabolized to 14-OH clarithromycin, which is active and works synergistically with its parent compound. Like other macrolides, it then penetrates bacteria cell wall and reversibly binds to domain V of the 23S ribosomal RNA of the 50S subunit of the bacterial ribosome, blocking translocation of aminoacyl transfer-RNA and polypeptide synthesis. Clarithromycin also inhibits the hepatic microsomal CYP3A4 isoenzyme and P-glycoprotein, an energy-dependent drug efflux pump., Clarithromycin usually is bacteriostatic, although it may be bactericidal in high concentrations or against highly susceptible organisms. Bactericidal activity has been observed against Streptococcus pyogenes, S. pneumoniae, Haemophilus influenzae, and Chlamydia trachomatis. Clarithromycin inhibits protein synthesis in susceptible organisms by penetrating the cell wall and binding to 50S ribosomal subunits, thereby inhibiting translocation of aminoacyl transfer-RNA and inhibiting polypeptide synthesis. The site of action of clarithromycin appears to be the same as that of erythromycin, clindamycin, lincomycin, and chloramphenicol. | |
| Record name | Clarithromycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01211 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Clarithromycin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8055 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Colorless needles from chloroform + diisopropyl ether (1:2) ... Also reported as crystals from ethanol | |
CAS No. |
81103-11-9, 116836-41-0 | |
| Record name | Clarithromycin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=81103-11-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Clarithromycin [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0081103119 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | (14R)-14-Hydroxyclarithromycin | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0116836410 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Clarithromycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01211 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | clarithromycin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758704 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Clarithromycin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3022829 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Erythromycin, 6-O-methyl | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.119.644 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Clarithromycin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CLARITHROMYCIN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/H1250JIK0A | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Clarithromycin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8055 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Clarithromycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015342 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
217-220 °C (decomposes) ... Also reported as mp 222-225 °C, 217 - 220 °C | |
| Record name | Clarithromycin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01211 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Clarithromycin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8055 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Clarithromycin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015342 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















