Cyclizine
Overview
Description
Cyclizine is a piperazine-derivative antihistamine used primarily as an antiemetic and antivertigo agent. It is commonly employed to prevent and treat nausea, vomiting, and dizziness associated with motion sickness and vertigo . This compound was discovered in 1947 and is included in the World Health Organization’s List of Essential Medicines .
Preparation Methods
Cyclizine can be synthesized through various methods. One common synthetic route involves the Eschweiler–Clarke methylation of diphenylmethylpiperazine . Another method includes the reaction of benzhydryl bromide with 1-methylpiperazine in acetonitrile to form the hydrobromide salt of the drug . Industrial production methods often focus on optimizing yield and purity, with specific reaction conditions tailored to achieve these goals .
Chemical Reactions Analysis
Cyclizine undergoes several types of chemical reactions, including:
Substitution: The compound can participate in substitution reactions, particularly involving the piperazine ring.
Hydrolysis: this compound can be hydrolyzed under acidic or basic conditions to yield its constituent components.
Common reagents and conditions used in these reactions include oxidizing agents like potassium permanganate and acidic or basic hydrolysis conditions. The major products formed from these reactions depend on the specific reagents and conditions employed.
Scientific Research Applications
Pharmacological Properties
Cyclizine is primarily recognized for its antiemetic properties, particularly in the prevention and treatment of motion sickness. It effectively alleviates symptoms such as nausea, vomiting, and dizziness associated with vestibular disorders. Additionally, this compound exhibits antihistaminic , anticholinergic , and central nervous system depressant effects, contributing to its therapeutic efficacy in various conditions.
Key Pharmacological Effects
- Antiemetic : Prevents nausea and vomiting.
- Antivertigo : Manages vertigo symptoms.
- Anti-inflammatory : Exhibits potential anti-inflammatory effects (as detailed in recent studies) .
- Cytotoxicity : Induces apoptosis in macrophages, revealing insights into cellular mechanisms .
Motion Sickness
This compound is widely used to prevent and treat motion sickness. Its effectiveness is attributed to its ability to depress labyrinth excitability and vestibular stimulation, impacting the chemoreceptor trigger zone in the medulla .
Management of Vertigo
The compound is also employed in the management of vertigo associated with inner ear disorders. Its central anticholinergic properties help mitigate symptoms effectively .
Postoperative Nausea
In clinical settings, this compound is administered intravenously for managing postoperative nausea and vomiting, particularly in pediatric populations .
Research Applications
Recent studies have explored this compound's potential beyond its traditional uses:
Anti-inflammatory Research
Research indicates that this compound derivatives can reduce vascular permeability and acute inflammation. However, they may not significantly affect chronic inflammatory responses .
Cytotoxicity Studies
A study demonstrated that this compound induces cytotoxicity and apoptosis in RAW264.7 macrophages through both intrinsic and extrinsic pathways. This highlights its potential implications in immune response modulation .
Case Study 1: this compound in Poisoning Management
A clinical case examined the use of this compound in patients experiencing drug-induced hypotension due to poisoning. The study emphasized the importance of careful monitoring and appropriate dosing to manage symptoms effectively .
Case Study 2: Pediatric Use
In a pediatric setting, this compound was successfully utilized for managing postoperative nausea in children aged 5 years and older, demonstrating its safety profile when administered correctly .
Data Tables
Mechanism of Action
Cyclizine exerts its effects primarily through its antihistaminic and anticholinergic properties. It acts as a histamine H1 receptor antagonist, blocking the action of histamine in the brain, which helps prevent nausea and vomiting . Additionally, this compound has central anticholinergic properties that depress labyrinth excitability and vestibular stimulation, affecting the medullary chemoreceptor trigger zone . This combination of actions contributes to its effectiveness in managing motion sickness and vertigo.
Comparison with Similar Compounds
Cyclizine is often compared with other antihistamines and antiemetics, such as:
Meclizine: Similar to this compound, meclizine is used to treat vertigo and motion sickness.
Dimenhydrinate (Dramamine): Another common antiemetic, dimenhydrinate is used for motion sickness but has a different side effect profile and may be less effective in some cases.
This compound’s unique combination of antihistaminic and anticholinergic properties, along with its relatively low incidence of drowsiness, sets it apart from these similar compounds .
Biological Activity
Cyclizine is a piperazine derivative primarily used as an antiemetic for the prevention and treatment of nausea and vomiting, particularly in postoperative settings and palliative care. Its biological activity extends beyond antiemetic effects, encompassing cytotoxicity, apoptosis induction, and pro-inflammatory responses. This article explores the various biological activities of this compound, supported by research findings, data tables, and case studies.
Antiemetic Effects
this compound has been shown to significantly reduce nausea and vomiting. In clinical trials, it decreased the risk of nausea by 65% and vomiting by 55% compared to placebo in postoperative patients . Its effectiveness is comparable to other antiemetics such as ondansetron . The compound acts primarily through antagonism of histamine H1 receptors in the central nervous system.
Cytotoxicity and Apoptosis
Recent studies indicate that this compound exhibits cytotoxic effects on macrophages. At a concentration of 500 μM, this compound induced approximately 70% cytotoxicity and 30% apoptosis in RAW264.7 macrophage cells. This effect was mediated through both intrinsic and extrinsic apoptotic pathways, involving mitochondrial dysfunction and upregulation of death receptors . The study demonstrated that this compound activated caspases 3, 8, and 9 while altering the expression of apoptotic regulatory proteins Bcl2 and Bad .
Pro-inflammatory Responses
this compound has also been implicated in enhancing pro-inflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6. The compound stimulates the phosphorylation of nuclear factor-κB (NFκB), leading to increased expression of these cytokines . This suggests that while this compound may be beneficial in treating nausea, it could also contribute to inflammatory processes under certain conditions.
Pharmacokinetics
The pharmacokinetic profile of this compound reveals important insights into its biological activity. Following administration, peak plasma concentrations are typically reached within 2-3 hours. For a 50 mg oral dose, peak concentrations are approximately 70 μg/L . this compound has a terminal elimination half-life ranging from 13.5 to 25 hours depending on the route of administration .
| Parameter | Value |
|---|---|
| Peak Plasma Concentration | ~70 μg/L (after 50 mg oral dose) |
| Elimination Half-Life | 13.5 - 25 hours |
| Volume of Distribution | ~16.5 L/kg |
| Clearance | ~0.87 L/h/kg |
Case Studies
Palliative Care
In a study examining real-world use in palliative care settings, approximately 75% of patients reported benefits from this compound for nausea management. However, about one-third experienced tolerable side effects . This highlights the importance of evaluating individual patient responses when considering this compound as a treatment option.
Postoperative Nausea and Vomiting (PONV)
A randomized controlled trial involving 960 women undergoing day-case surgery found that combining this compound with granisetron reduced the incidence of PONV more effectively than either drug alone (17% vs. 23% for this compound alone) . This suggests potential benefits in combination therapy for managing nausea in surgical patients.
Q & A
Basic Research Questions
Q. What experimental models validate cyclizine’s antiemetic efficacy, and what methodological limitations exist in historical studies?
- Answer : this compound’s antiemetic efficacy is primarily supported by a 1954 study using an emesis model in animals, demonstrating inhibition of motion-induced vomiting . However, this study lacked dose-dependency analysis, selectivity assessments, and modern pharmacokinetic profiling (e.g., no hERG channel or ECG data). Current research should prioritize:
- Dose-response curves in validated emesis models (e.g., cisplatin-induced vomiting in ferrets).
- Mechanistic studies using histamine H1 receptor knockout models to isolate this compound’s antihistamine vs. anticholinergic effects .
Q. How does this compound interact with the central nervous system (CNS), and what safety pharmacology gaps persist?
- Answer : Limited safety data from pre-1990s studies show this compound reduces spontaneous motor activity at low doses (5 mg/kg IP) but increases hyperactivity at higher doses (20 mg/kg IP) in rodents . Critical gaps include:
- Electrophysiological assays (e.g., hERG inhibition, QT prolongation risks).
- Neuroinflammatory profiling (e.g., cytokine modulation in microglia).
- Methodology : Use in vitro hERG assays and in vivo EEG monitoring to assess CNS excitation/sedation thresholds .
Advanced Research Questions
Q. What molecular mechanisms underlie this compound’s potentiation of opioid analgesia, and how can these interactions be quantified?
- Answer : this compound enhances opioid-induced antinociception in rodents via histaminergic pathway modulation, independent of direct receptor binding . Key methods:
- Isobolographic analysis to quantify synergistic interactions with morphine/pentazocine.
- Microdialysis in CNS regions (e.g., nucleus tractus solitarius) to measure histamine/β-endorphin release .
Q. How does this compound induce pancreatic toxicity in preclinical models, and what translational relevance exists for human glucose metabolism?
- Answer : Oral this compound (≥75 mg/kg/day) in rats causes pancreatic β-cell damage, hyperglycemia, and insulin dysregulation . Research strategies:
- Transcriptomic analysis of this compound-treated pancreatic islets (e.g., RNA-seq for apoptosis markers like BAX/Bcl-2).
- Human relevance : Compare this compound’s effects on insulin secretion in iPSC-derived β-cells vs. rodent models .
Q. What experimental approaches resolve contradictions in this compound’s reproductive toxicity data across species?
- Answer : Published studies in rats, mice, and rabbits report resorptions and malformations at variable NOELs (no observed effect levels) without maternal toxicity . Solutions:
- Systematic review of species-specific placental transfer rates (e.g., LC-MS/MS quantification of this compound/northis compound in fetal tissues).
- Dose standardization using allometric scaling to human-equivalent doses .
Q. Methodological Guidance
Q. How can researchers address this compound’s pharmacokinetic variability across species and administration routes?
- Answer : this compound’s bioavailability differs markedly between IV (humans) and oral (dogs), with major metabolite northis compound lacking antihistaminic activity . Strategies:
- Species-specific metabolic profiling : Use liver microsomes from humans, dogs, and rats to compare CYP450-mediated demethylation.
- Route optimization : Conduct crossover trials in animal models comparing IV, IM, and oral bioavailability via LC-MS/MS .
Q. What analytical techniques validate this compound’s metabolic pathways and impurity profiles in biological matrices?
- Answer :
- Basic : GC-MS for urinary northis compound detection (limit of quantification: 10 ng/mL) .
- Advanced : UPLC-PDA for resolving this compound from toxic impurities (e.g., diphenylmethane derivatives) in plasma .
- ADMET prediction : Use Schrödinger’s QikProp to model this compound’s blood-brain barrier permeability .
Q. Critical Research Gaps
- Mechanistic clarity : this compound’s inhibition of hepatic estrone sulfotransferase and murine MAO-B requires human enzyme kinetic studies .
- Proinflammatory effects : this compound activates Akt-NFκB in macrophages, elevating TNF-α and IL-6—implications for chronic inflammation in overdose cases .
Properties
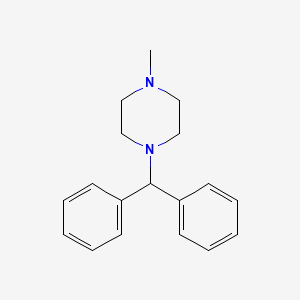
IUPAC Name |
1-benzhydryl-4-methylpiperazine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C18H22N2/c1-19-12-14-20(15-13-19)18(16-8-4-2-5-9-16)17-10-6-3-7-11-17/h2-11,18H,12-15H2,1H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
UVKZSORBKUEBAZ-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1CCN(CC1)C(C2=CC=CC=C2)C3=CC=CC=C3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C18H22N2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4022864 | |
| Record name | Cyclizine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4022864 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
266.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Cyclizine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015307 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
INSOL IN ETHER 1 G IN ABOUT 115 ML ALC /HYDROCHLORIDE/, SOL IN ALCOHOL & CHLOROFORM; SLIGHTLY SOL IN WATER, FREELY SOL IN ETHER, 7.52e-02 g/L | |
| Record name | Cyclizine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01176 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CYCLIZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3309 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cyclizine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015307 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Vomiting (emesis) is essentially a protective mechanism for removing irritant or otherwise harmful substances from the upper GI tract. Emesis or vomiting is controlled by the vomiting centre in the medulla region of the brain, an important part of which is the chemotrigger zone (CTZ). The vomiting centre possesses neurons which are rich in muscarinic cholinergic and histamine containing synapses. These types of neurons are especially involved in transmission from the vestibular apparatus to the vomiting centre. Motion sickness principally involves overstimulation of these pathways due to various sensory stimuli. Hence the action of cyclizine which acts to block the histamine receptors in the vomiting centre and thus reduce activity along these pathways. Furthermore since cyclizine possesses anti-cholinergic properties as well, the muscarinic receptors are similarly blocked., .../IT SEEMS/ THAT STIMULATION OF VESTIBULAR APPARATUS IS NECESSARY & SUFFICIENT...& THAT VESTIBULAR CEREBELLAR MIDBRAIN "INTEGRATIVE VOMITING CENTER" & MEDULLARY CHEMORECEPTIVE TRIGGER ZONE ARE...INVOLVED /IN MOTION SICKNESS/. IT IS...PROBABLE THAT EFFECTIVE ANTIHISTAMINES EXERT.../ACTION/ IN THESE CENTERS. /ANTIHISTAMINE/ | |
| Record name | Cyclizine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01176 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CYCLIZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3309 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
CRYSTALS FROM PETROLEUM ETHER, WHITE OR CREAMY WHITE CRYSTALLINE POWDER | |
CAS No. |
82-92-8 | |
| Record name | Cyclizine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=82-92-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cyclizine [USP:INN:BAN:DCF] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000082928 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cyclizine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01176 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CYCLIZINE | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=756710 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | CYCLIZINE | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=26608 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Cyclizine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4022864 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Cyclizine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.001.314 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CYCLIZINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/QRW9FCR9P2 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | CYCLIZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3309 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cyclizine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015307 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
105.5-107.5, 105.5 TO 107.5 °C | |
| Record name | Cyclizine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01176 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CYCLIZINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3309 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















