Delamanid
Overview
Description
Delamanid is a nitro-dihydro-imidazooxazole derivative used primarily in the treatment of multidrug-resistant tuberculosis (MDR-TB). It is known for its potent in vitro and in vivo antitubercular activity against both drug-susceptible and drug-resistant strains of Mycobacterium tuberculosis . This compound is marketed under the trade name Deltyba and is approved for use in several countries, including Japan and those in the European Union .
Preparation Methods
Synthetic Routes and Reaction Conditions: Delamanid is synthesized through a multi-step process involving the formation of the imidazooxazole core followed by the introduction of various functional groups. The key steps include:
- Formation of the imidazooxazole ring.
- Introduction of the nitro group.
- Attachment of the trifluoromethoxyphenoxy and piperidinylphenoxy groups.
Industrial Production Methods: Industrial production of this compound involves large-scale synthesis using optimized reaction conditions to ensure high yield and purity. The process includes:
- Use of high-pressure homogenization for emulsification.
- Spray-drying to form a dried powder.
- Ensuring stability through nanoencapsulation to enhance aqueous dissolution kinetics .
Chemical Reactions Analysis
Types of Reactions: Delamanid undergoes various chemical reactions, including:
Reduction: The nitro group is reduced to an amine.
Oxidation: The imidazooxazole ring can undergo oxidation under specific conditions.
Substitution: Functional groups on the imidazooxazole ring can be substituted with other groups.
Common Reagents and Conditions:
Reduction: Catalytic hydrogenation using palladium on carbon.
Oxidation: Use of oxidizing agents like potassium permanganate.
Substitution: Nucleophilic substitution reactions using appropriate nucleophiles.
Major Products:
- Reduction of the nitro group leads to the formation of an amine derivative.
- Oxidation of the imidazooxazole ring can produce various oxidized derivatives.
- Substitution reactions yield a variety of substituted imidazooxazole compounds .
Scientific Research Applications
Delamanid has a wide range of scientific research applications, including:
Chemistry: Used as a model compound for studying nitroimidazole derivatives.
Biology: Investigated for its effects on Mycobacterium tuberculosis and other bacterial strains.
Medicine: Primarily used in the treatment of multidrug-resistant tuberculosis. .
Industry: Employed in the development of new antitubercular drugs and formulations.
Mechanism of Action
Delamanid exerts its effects by inhibiting the synthesis of methoxy- and keto-mycolic acids, which are essential components of the mycobacterial cell wall. It is a prodrug that requires activation by the mycobacterial F420 coenzyme system, including the deazaflavin-dependent nitroreductase. This activation leads to the generation of reactive nitrogen species, which disrupt the synthesis of mycolic acids and ultimately kill the bacteria .
Comparison with Similar Compounds
Pretomanid: Another nitroimidazole derivative used in the treatment of tuberculosis.
Isoniazid: A first-line antitubercular drug that inhibits mycolic acid synthesis.
Rifampicin: An antibiotic that inhibits bacterial RNA synthesis.
Comparison:
Delamanid vs. Pretomanid: this compound exhibits greater in vitro potency against multidrug-resistant and extensively drug-resistant tuberculosis strains compared to pretomanid.
This compound vs. Isoniazid: While both inhibit mycolic acid synthesis, this compound has a distinct mechanism involving the F420 coenzyme system.
This compound vs. Rifampicin: this compound targets mycolic acid synthesis, whereas rifampicin inhibits RNA synthesis, making them complementary in combination therapy
This compound’s unique mechanism of action and its efficacy against drug-resistant strains make it a valuable addition to the arsenal of antitubercular drugs.
Biological Activity
Delamanid, a novel compound from the nitro-dihydro-imidazooxazole class, has emerged as a significant agent in the fight against multidrug-resistant tuberculosis (MDR-TB). Its unique mechanism of action, coupled with potent biological activity against various strains of Mycobacterium tuberculosis (MTB), positions it as a valuable addition to existing tuberculosis therapies.
This compound primarily functions by inhibiting the synthesis of mycolic acids, crucial components of the mycobacterial cell wall. This inhibition is mediated through the activation of this compound by the enzyme deazaflavin-dependent nitroreductase (Ddn), which facilitates its conversion into a reactive intermediate that disrupts mycolic acid production .
Key Mechanistic Insights:
- Pro-drug Activation : this compound is a pro-drug that requires metabolic activation by Ddn, which is specific to mycobacteria and not present in human cells, thereby minimizing potential toxicity .
- Inhibition of Mycolic Acid Synthesis : The drug targets methoxy-mycolic and keto-mycolic acid synthesis pathways, critical for mycobacterial cell wall integrity .
- Selective Activity : The selective activation mechanism helps explain this compound's efficacy against mycobacteria while avoiding genotoxic effects in humans .
In Vitro Studies
This compound has demonstrated potent antibacterial activity in vitro against both drug-susceptible and drug-resistant strains of MTB. The minimum inhibitory concentration (MIC) ranges from 0.006 to 0.024 μg/ml, indicating high potency . Notably, studies reveal:
- No Cross-Resistance : this compound does not exhibit cross-resistance with first-line anti-TB drugs such as rifampicin and isoniazid, making it an effective option for patients who have failed previous treatments .
- Post-Antibiotic Effect : It shows a significant post-antibiotic effect on intracellular organisms after pulsed therapy, comparable to rifampicin .
Clinical Trials
Clinical investigations have confirmed this compound's efficacy in real-world settings:
- Early Bactericidal Activity : A study involving smear-positive TB patients showed that this compound significantly reduced colony-forming units (CFUs) over a 14-day treatment period, with higher doses correlating with greater reductions .
- Randomized Trials : In a placebo-controlled trial on MDR-TB patients, this compound combined with an optimized background regimen resulted in higher sputum culture conversion rates compared to placebo .
Case Studies
A review of clinical outcomes indicated that treatment with this compound for six months led to improved patient outcomes and reduced mortality rates among MDR-TB patients .
Summary of Research Findings
| Study Type | Findings |
|---|---|
| In Vitro Studies | MIC of 0.006-0.024 μg/ml; no cross-resistance with first-line drugs; significant post-antibiotic effect observed. |
| Clinical Trials | Significant reduction in CFUs; higher sputum culture conversion rates in treated groups. |
| Case Studies | Improved outcomes and reduced mortality in patients treated with this compound alongside optimized regimens. |
Resistance Mechanisms
Despite its effectiveness, resistance to this compound can occur. Mutations in coenzyme F420 genes have been implicated as mechanisms for resistance in mycobacteria . Understanding these resistance pathways is crucial for developing strategies to mitigate treatment failures.
Q & A
Basic Research Questions
Q. What is the molecular mechanism of Delamanid’s antimycobacterial activity, and how can this inform experimental design?
this compound inhibits mycolic acid synthesis by targeting the F420-dependent deazaflavin nitroreductase (Ddn) system in Mycobacterium tuberculosis (Mtb), disrupting cell wall integrity . To validate this mechanism experimentally:
- Use in vitro assays with F420-deficient Mtb strains to confirm target specificity.
- Combine lipidomic profiling (e.g., HPLC) to quantify mycolic acid inhibition .
- Reference clinical pharmacokinetic data (e.g., protein binding >99%, half-life 30–38 hours) to optimize dosing in animal models .
Q. How should researchers design studies to evaluate this compound’s efficacy in multidrug-resistant TB (MDR-TB) patients?
- Cohort selection : Prioritize participants with baseline sputum culture positivity and documented MDR/RR-TB resistance, excluding those with concurrent CYP3A4 inducer use (e.g., rifampicin) to avoid pharmacokinetic interference .
- Outcome metrics : Use two consecutive negative cultures ≥15 days apart as the primary endpoint for culture conversion .
- Control for confounders : Adjust for variables like prior TB drug exposure, comorbidities, and regimen changes using inverse probability weighting .
Q. What are the key considerations for interpreting this compound’s clinical trial data, particularly conflicting results on culture conversion?
- Example : In the endTB cohort, adding this compound to 3-drug regimens did not significantly improve 2-month culture conversion (49.6% vs. 55.6% in controls) but reduced mortality with prolonged use .
- Methodological resolution : Conduct time-to-event analysis to assess delayed conversion and stratify by treatment duration (e.g., ≥6 months vs. shorter courses) .
Advanced Research Questions
Q. How can this compound’s pharmacokinetic variability be addressed in preclinical-to-clinical translation?
- Challenge : this compound’s absorption increases 3-fold with high-fat meals, complicating dosing consistency .
- Solutions :
- Standardize fed/fasted conditions in animal studies and correlate with plasma exposure (AUC/MIC ratios).
- Use physiologically based pharmacokinetic (PBPK) modeling to predict human outcomes from nonclinical data .
Q. What experimental strategies can elucidate this compound’s resistance mechanisms?
- Hypothesis : Mutations in F420 biosynthetic genes (e.g., fgd, fbiA) reduce prodrug activation .
- Methods :
- Perform whole-genome sequencing of post-treatment Mtb isolates to identify resistance-associated variants.
- Validate using in vitro susceptibility testing with isogenic mutant strains .
Q. How should researchers evaluate this compound’s synergy with other anti-TB drugs in combination regimens?
- Approach :
- Use checkerboard assays or time-kill curves to quantify synergy with bedaquiline, linezolid, or pretomanid .
- Monitor for QT prolongation when combining with other CYP3A4-metabolized drugs (e.g., clofazimine) via electrocardiogram in phase II trials .
Q. How can contradictory findings on this compound’s efficacy be resolved in meta-analyses?
- Case : Early studies reported 70–95% culture conversion with this compound, while later trials showed marginal benefits .
- Resolution :
- Apply GRADE criteria to assess bias risk, heterogeneity, and publication bias.
- Perform subgroup analyses by region (e.g., high vs. low TB burden) and resistance profiles .
Q. Methodological Rigor and Reproducibility
Q. What steps ensure reproducibility in this compound-related studies?
- Data transparency : Publish raw culture conversion timelines and regimen adjustments in supplementary materials .
- Protocol adherence : Follow WHO guidelines for MDR-TB treatment duration and outcome definitions (e.g., "cured" vs. "completed") .
- Statistical rigor : Pre-specify endpoints and adjust for immortal time bias in observational studies using Cox models .
Q. How can researchers adapt preclinical models for this compound’s extrapulmonary TB applications?
- Model selection : Use murine or rabbit models of TB meningitis or osteomyelitis to assess blood-brain barrier penetration .
- Metrics : Compare this compound concentrations in infected tissues (e.g., lymph nodes) via LC-MS/MS .
Q. Tables for Key Data
Table 1. This compound Pharmacokinetic Parameters
| Parameter | Value | Source |
|---|---|---|
| Protein binding | >99% | |
| Half-life | 30–38 hours | |
| Metabolic pathway | CYP3A4-mediated hydrolysis | |
| Food effect (AUC) | 3-fold increase with high-fat |
Table 2. Clinical Outcomes from Key this compound Studies
| Study Design | Culture Conversion (2-month) | Mortality (≥6 months) | Source |
|---|---|---|---|
| endTB Observational | 49.6% (this compound) vs. 55.6% | 1.0% (this compound) | |
| Phase IIb Trial | 45.4% (this compound) vs. 29.6% | 8.3% (Control) |
Properties
IUPAC Name |
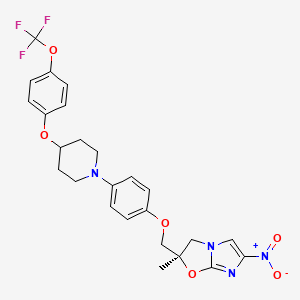
(2R)-2-methyl-6-nitro-2-[[4-[4-[4-(trifluoromethoxy)phenoxy]piperidin-1-yl]phenoxy]methyl]-3H-imidazo[2,1-b][1,3]oxazole | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C25H25F3N4O6/c1-24(15-31-14-22(32(33)34)29-23(31)38-24)16-35-18-4-2-17(3-5-18)30-12-10-20(11-13-30)36-19-6-8-21(9-7-19)37-25(26,27)28/h2-9,14,20H,10-13,15-16H2,1H3/t24-/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
XDAOLTSRNUSPPH-XMMPIXPASA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1(CN2C=C(N=C2O1)[N+](=O)[O-])COC3=CC=C(C=C3)N4CCC(CC4)OC5=CC=C(C=C5)OC(F)(F)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@@]1(CN2C=C(N=C2O1)[N+](=O)[O-])COC3=CC=C(C=C3)N4CCC(CC4)OC5=CC=C(C=C5)OC(F)(F)F | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C25H25F3N4O6 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID60218326 | |
| Record name | Delamanid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID60218326 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
534.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
Delamanid is a prodrug that requires biotransformation via via the mycobacterial F420 coenzyme system, including the deazaflavin dependent nitroreductase (Rv3547), to mediate its antimycobacterial activity against both growing and nongrowing mycobacteria. Mutations in one of five coenzyme F420 genes, _fgd, Rv3547, fbiA, fbiB, and fbiC_ has been proposed as the mechanism of resistance to delamanid. Upon activation, the radical intermediate formed between delamanid and desnitro-imidazooxazole derivative is thought to mediate antimycobacterial actions via inhibition of methoxy-mycolic and keto-mycolic acid synthesis, leading to depletion of mycobacterial cell wall components and destruction of the mycobacteria. Nitroimidazooxazole derivative is thought to generate reactive nitrogen species, including nitrogen oxide (NO). However unlike isoniazid, delamanid does not alpha-mycolic acid. | |
| Record name | Delamanid | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11637 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
681492-22-8 | |
| Record name | Delamanid | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=681492-22-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Delamanid [USAN:INN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0681492228 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Delamanid | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11637 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Delamanid | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID60218326 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | DELAMANID | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/8OOT6M1PC7 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















