Domperidone
Overview
Description
Domperidone is a dopamine receptor antagonist used primarily as an antiemetic and prokinetic agent. It is effective in treating nausea, vomiting, and gastrointestinal disorders such as gastroparesis. This compound works by blocking dopamine receptors in the gut and the chemoreceptor trigger zone, which helps to increase gastrointestinal motility and reduce symptoms of nausea .
Preparation Methods
Domperidone can be synthesized through the coupling reaction of two benzimidazolone derivatives. The synthetic routes involve the cyclization of o-phenylenediamine with carbonyl reagents followed by coupling with 1,3-dihalopropane. Another route involves the coupling reaction of o-halo or o-amino substituted nitrobenzene with 1,3-disubstituted propane followed by reduction and cyclization. The latter route avoids the production of di-substituted by-products and has higher reaction selectivity .
Industrial production methods for this compound tablets involve direct powder compression of the raw material. The process includes pulverizing the raw material, mixing it with excipients such as carboxymethyl starch sodium, pregelatinized starch, microcrystalline cellulose, and magnesium stearate, and then tabletting the mixture .
Chemical Reactions Analysis
Domperidone undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized under specific conditions, leading to the formation of oxidized derivatives.
Reduction: Reduction reactions can convert nitro groups to amino groups, which are essential in the synthesis of this compound intermediates.
Substitution: Substitution reactions, such as halogenation, can modify the chemical structure of this compound, leading to different derivatives.
Common reagents used in these reactions include carbonyl reagents, 1,3-dihalopropane, and reducing agents. The major products formed from these reactions are benzimidazolone derivatives and substituted nitrobenzene derivatives .
Scientific Research Applications
Introduction to Domperidone
This compound is a dopamine-2 receptor antagonist primarily used as an antiemetic and prokinetic agent. It acts on the gastrointestinal tract to enhance gastric motility and alleviate nausea and vomiting. Unlike metoclopramide, this compound does not penetrate the blood-brain barrier significantly, which minimizes neurological side effects and enhances its safety profile for long-term use . This article explores the diverse applications of this compound in clinical settings, supported by comprehensive data tables and case studies.
Gastrointestinal Disorders
Gastroparesis and Nonulcer Dyspepsia
This compound has been effectively utilized in managing gastroparesis, a condition characterized by delayed gastric emptying. A study involving patients with idiopathic gastric stasis demonstrated that while motility was unchanged, symptom scores significantly improved in those treated with this compound compared to placebo . This highlights its role in symptomatic relief rather than direct motility enhancement.
Gastroesophageal Reflux Disease (GERD)
Recent research indicates that combining this compound with proton pump inhibitors (PPIs) may provide superior outcomes in GERD management. A systematic review showed that this combination significantly reduced reflux episodes and improved symptom scores compared to PPIs alone . The efficacy of this compound in this context is further illustrated in the following table:
| Study | Population | Treatment Duration | Treatment Regimen | Results |
|---|---|---|---|---|
| Bediwy 2014 | Children | 12 weeks | This compound 0.5 mg/kg + Esomeprazole 2 mg/kg | Improved symptom scores |
| Huiying 2020 | Adults | 1 month | This compound 10 mg + Omeprazole 20 mg | Significant reduction in heartburn score |
| Jianzhong 2019 | Children | 2 months | This compound 10 mg + Omeprazole 20 mg | Enhanced symptom relief |
Lactation Support
This compound is frequently prescribed off-label to stimulate milk production in breastfeeding women. A case study documented a nursing mother who successfully increased her milk supply using this compound, although she experienced withdrawal symptoms upon discontinuation after prolonged use . This application underscores the compound's utility beyond gastrointestinal disorders.
Neurological Applications
While primarily indicated for gastrointestinal issues, this compound has been investigated for its potential benefits in treating certain neurological conditions associated with nausea and vomiting, such as migraines. Its efficacy in these contexts remains an area of ongoing research.
Safety Profile and Side Effects
This compound is generally well-tolerated, with a favorable safety profile due to its limited central nervous system penetration. Common side effects include gastrointestinal disturbances such as abdominal cramps or diarrhea. However, serious adverse effects are rare when used at recommended doses .
Case Studies on Safety
A notable case involved a breastfeeding woman who experienced withdrawal symptoms after abrupt cessation of this compound following eight months of use. Symptoms included tachycardia and anxiety, indicating that prolonged exposure may lead to tolerance and subsequent withdrawal challenges .
Mechanism of Action
Domperidone exerts its effects by blocking dopamine D2 and D3 receptors in the gut and the chemoreceptor trigger zone. This blockade increases gastrointestinal peristalsis and accelerates gastric emptying. Additionally, this compound causes the release of prolactin by blocking dopamine receptors in the pituitary gland, which is outside the blood-brain barrier .
Comparison with Similar Compounds
Domperidone is often compared with other dopamine receptor antagonists such as metoclopramide. Unlike metoclopramide, this compound has a lower propensity to cross the blood-brain barrier, resulting in fewer central nervous system side effects. This makes this compound a preferred choice for patients who are sensitive to neurological side effects .
Similar compounds include:
Metoclopramide: Another dopamine receptor antagonist used for similar indications but with a higher risk of central nervous system side effects.
Cisapride: A prokinetic agent that also enhances gastrointestinal motility but has been associated with serious cardiac side effects.
This compound’s unique ability to selectively block peripheral dopamine receptors while minimizing central nervous system side effects distinguishes it from other similar compounds .
Biological Activity
Domperidone is an antiemetic agent primarily used to treat nausea and vomiting. It acts as a peripheral dopamine D2 receptor antagonist, which influences gastrointestinal motility and has been explored for various therapeutic applications beyond its conventional uses. This article delves into the biological activity of this compound, summarizing its mechanisms, pharmacological effects, and recent research findings.
This compound selectively blocks dopamine D2 and D3 receptors located in the gastrointestinal tract and the chemoreceptor trigger zone (CTZ) in the brain. By inhibiting these receptors, this compound increases gastric motility and accelerates gastric emptying, which helps alleviate symptoms of nausea and vomiting. Additionally, it stimulates prolactin release from the pituitary gland, which may have immunomodulatory effects .
Pharmacological Effects
The pharmacological effects of this compound can be summarized as follows:
- Antiemetic Activity : Reduces nausea and vomiting.
- Gastrointestinal Motility : Enhances gastric emptying and intestinal transit.
- Prolactin Stimulation : Increases prolactin levels, potentially influencing immune responses.
Recent Research Findings
-
Antiviral Activity Against SARS-CoV-2 :
A recent clinical trial investigated the efficacy of this compound in reducing viral load in patients with mild-to-moderate COVID-19. The study found that while this compound exhibited in vitro antiviral activity against SARS-CoV-2, it did not significantly reduce viral load compared to placebo in clinical settings. Adverse events were reported in both groups, with no hospitalizations occurring . -
Antitumor Activity in Breast Cancer :
Research has shown that this compound exhibits cytotoxic effects on triple-negative breast cancer (TNBC) cell lines (BT-549 and CAL-51). The study concluded that this compound could be a potential therapeutic agent for TNBC due to its ability to induce apoptosis in cancer cells . -
Effects on Orthostatic Hypotension :
This compound has been evaluated for its effectiveness in managing orthostatic hypotension, particularly in Parkinson's disease patients. It was noted to help improve symptoms associated with this condition, although further studies are needed to establish its safety profile .
Case Study 1: this compound for COVID-19
A double-blind clinical trial conducted in Spain involved 173 outpatients with mild-to-moderate COVID-19 who received either 30 mg/day of this compound or a placebo for seven days. The primary endpoint was the reduction of viral load measured by RT-qPCR. While there was a statistically significant reduction in viral load at day 4 compared to baseline, the difference between treatment and placebo groups was not significant at later time points .
Case Study 2: this compound in Breast Cancer
In vitro studies on TNBC cells demonstrated that treatment with this compound resulted in significant cytotoxicity. The mechanism was linked to the induction of apoptosis via modulation of signaling pathways associated with cell survival and death .
Summary of Biological Activities
| Activity Type | Description |
|---|---|
| Antiemetic | Reduces nausea and vomiting through D2 receptor antagonism |
| Gastrointestinal Motility | Enhances gastric emptying and intestinal transit |
| Prolactin Stimulation | Increases prolactin levels, potentially enhancing immune function |
| Antiviral | Exhibits in vitro activity against SARS-CoV-2; unclear clinical efficacy |
| Antitumor | Induces apoptosis in TNBC cells |
| Orthostatic Hypotension Management | Improves symptoms related to orthostatic hypotension |
Q & A
Basic Research Questions
Q. What is the molecular mechanism of Domperidone as a dopamine D2 receptor antagonist in gastrointestinal motility?
this compound selectively blocks peripheral dopamine D2 receptors, increasing gastric motility by antagonizing dopamine-mediated inhibition of gastric emptying. It enhances lower esophageal sphincter pressure (LESP) and promotes antral peristalsis without affecting gastrin release or gastric acid secretion. Experimental validation involves in vitro muscle contraction assays and receptor binding studies using radiolabeled ligands .
Q. What are the current gaps in clinical efficacy data for this compound in pediatric populations?
Limited studies exist on this compound’s efficacy in pediatric gastroesophageal reflux disease (GERD). Most trials involve older children or combine this compound with other agents, complicating isolation of its effects. A well-designed comparative trial against other prokinetics is needed, prioritizing safety monitoring for QT prolongation risks observed in adults .
Q. How is this compound’s bioavailability affected by co-administered drugs?
this compound is metabolized via CYP3A4; concurrent use of CYP3A4 inhibitors (e.g., ketoconazole) elevates plasma levels. Pharmacokinetic studies require validated HPLC methods with fluorescence detection (e.g., LLOQ: 0.5 ng/mL) to quantify this compound in human plasma and assess drug-drug interactions .
Advanced Research Questions
Q. How does this compound modulate the MEK/ERK/STAT3 signaling pathway in colorectal cancer (CRC) cells?
this compound downregulates DRD2 expression, reducing β-arrestin2-MEK complex formation in HCT116 cells. Immunoprecipitation and immunoblotting (Fig. 4B-C) show selective inhibition of MEK/ERK/STAT3 signaling, impacting proliferation and apoptosis. Experimental designs should include siRNA-mediated DRD2 knockdown to confirm pathway specificity .
Q. What methodological approaches are used to assess this compound-induced apoptosis via mitochondrial superoxide in triple-negative breast cancer (TNBC) models?
Apoptosis is quantified via annexin V/PI staining (flow cytometry) and caspase cleavage (immunoblotting). Mitochondrial superoxide levels are measured using MitoSOX Red, with scavengers like Mito-TEMPO (100 nM) to validate ROS-dependent mechanisms. Dose-response curves (IC50: ~50 µM) and mitochondrial membrane potential assays (JC-1 staining) further characterize cytotoxicity .
Q. How can statistical methods address data contradictions in this compound studies with combined therapies?
When assumptions for parametric tests (e.g., ANOVA) are violated (non-normal distribution, heterogeneous variance), non-parametric alternatives (Mann-Whitney U, Kruskal-Wallis) are recommended. For time-series data in pharmacological studies, mixed-effects models account for repeated measures and missing data .
Q. What particle dispersion methods optimize this compound particle size analysis for formulation development?
Wet dispersion with laser diffraction (Bettersizer 2600) provides accurate particle size distribution by minimizing aggregation. Key parameters include ultrasonic dispersing time (≥2 min) and stirring speed (2,500 rpm). Comparative dry dispersion studies (air pressure: 0.5–1.0 bar) validate reproducibility .
Q. Experimental Design & Data Analysis
Q. How to design in vitro studies evaluating this compound’s impact on cell cycle progression?
Flow cytometry (propidium iodide staining) quantifies cell cycle phases (subG1, G2/M arrest). Immunoblotting for cyclins (A, B, D) and CDKs, alongside inhibitors (p21, p27), identifies regulatory mechanisms. TNBC models (e.g., CAL-51 cells) show polyploidy induction (26.9% at 50 µM), requiring mitotic index validation .
Q. What kinetic models best describe this compound release from gastroretentive formulations?
Zero-order (F = k₀t) and Korsmeyer-Peppas models (Mt/M∞ = k·tⁿ) fit dissolution data. Higuchi diffusion models are less suitable for floating tablets, where matrix erosion dominates. R² >0.95 and AIC/BIC criteria guide model selection .
Q. How to mitigate confounding variables in this compound’s prokinetic efficacy studies?
Control for anticholinergic co-therapies (reduce motility) and gastric pH (antacids lower bioavailability). Randomize cohorts by CYP3A4 genotype to account for metabolic variability. Placebo-controlled crossover designs enhance internal validity .
Properties
IUPAC Name |
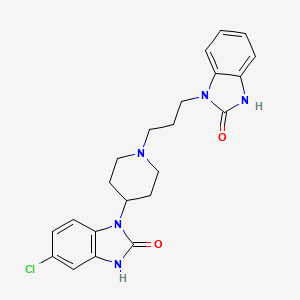
6-chloro-3-[1-[3-(2-oxo-3H-benzimidazol-1-yl)propyl]piperidin-4-yl]-1H-benzimidazol-2-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H24ClN5O2/c23-15-6-7-20-18(14-15)25-22(30)28(20)16-8-12-26(13-9-16)10-3-11-27-19-5-2-1-4-17(19)24-21(27)29/h1-2,4-7,14,16H,3,8-13H2,(H,24,29)(H,25,30) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
FGXWKSZFVQUSTL-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CN(CCC1N2C3=C(C=C(C=C3)Cl)NC2=O)CCCN4C5=CC=CC=C5NC4=O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H24ClN5O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
83898-65-1 (maleate (1:1)), 99497-03-7 (maleate), 83898-65-1 (maleate salt/solvate) | |
| Record name | Domperidone [USAN:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0057808669 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID1045116 | |
| Record name | Domperidone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1045116 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
425.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Domperidone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015315 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
50.4 [ug/mL] (The mean of the results at pH 7.4), 9.25e-02 g/L | |
| Record name | SID855562 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Domperidone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01184 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Domperidone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015315 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Domperidone acts as a gastrointestinal emptying (delayed) adjunct and peristaltic stimulant. The gastroprokinetic properties of domperidone are related to its peripheral dopamine receptor blocking properties. Domperidone facilitates gastric emptying and decreases small bowel transit time by increasing esophageal and gastric peristalsis and by lowering esophageal sphincter pressure. Antiemetic: The antiemetic properties of domperidone are related to its dopamine receptor blocking activity at both the chemoreceptor trigger zone and at the gastric level. It has strong affinities for the D2 and D3 dopamine receptors, which are found in the chemoreceptor trigger zone, located just outside the blood brain barrier, which - among others - regulates nausea and vomiting | |
| Record name | Domperidone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01184 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
57808-66-9 | |
| Record name | Domperidone | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=57808-66-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Domperidone [USAN:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0057808669 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Domperidone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01184 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | domperidone | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759575 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | domperidone | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=299589 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Domperidone | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1045116 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Domperidone | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.055.408 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DOMPERIDONE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/5587267Z69 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Domperidone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015315 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
242.5 °C | |
| Record name | Domperidone | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01184 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Domperidone | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015315 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













