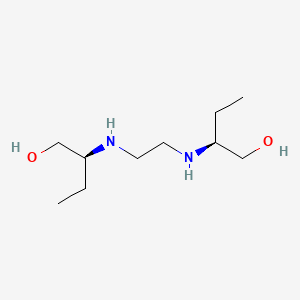
Ethambutol
Overview
Description
Ethambutol is an ethylenediamine derivative that is ethane-1,2-diamine in which one hydrogen attached to each of the nitrogens is sutstituted by a 1-hydroxybutan-2-yl group (S,S-configuration). It is a bacteriostatic antimycobacterial drug, effective against Mycobacterium tuberculosis and some other mycobacteria. It is used (as the dihydrochloride salt) in combination with other antituberculous drugs in the treatment of pulmonary and extrapulmonary tuberculosis; resistant strains of M. tuberculosis are readily produced if this compound is used alone. It has a role as an antitubercular agent, an environmental contaminant and a xenobiotic. It is a member of ethanolamines and an ethylenediamine derivative.
This compound hydrochloride is an antibacterial prescription medicine approved by the U.S. Food and Drug Administration (FDA) for the treatment of active tuberculosis (TB) of the lungs. (Active TB is also called TB disease.)
TB can be an opportunistic infection (OI) of HIV.
This compound is a bacteriostatic agent indicated alongside medications such as [isoniazid], [rifampin], and [pyrazinamide] in the treatment of pulmonary tuberculosis. This compound was first described in the literature in 1961. It was developed out of a need for therapies active against isoniazid resistant strains of Mycobacterium tuberculosis. this compound was granted FDA approval on 6 November 1967.
This compound is an Antimycobacterial.
This compound is a first line but adjunctive antituberculosis medication which is used only in combination with other agents such as isoniazid and rifampin. This compound therapy has been associated with minor, transient and asymptomatic elevations in serum aminotransferase levels, and is a reported but rare cause of clinically apparent acute liver injury.
This compound is a natural product found in Aspergillus sclerotiorum with data available.
This compound is an antibiotic with bacteriostatic, antimicrobial and antitubercular properties. This compound interferes with the biosynthesis of arabinogalactan, a major polysaccharide of the mycobacterial cell wall. It inhibits the polymerization of cell wall arabinan of arabinogalactan and lipoarabinomannan by blocking arabinosyl transferases and induces the accumulation of D-arabinofuranosyl-P-decaprenol, an intermediate in arabinan biosynthesis. This results in halting bacterial growth.
This compound Hydrochloride is the hydrochloride salt form of this compound, an ethylenediamine derivative with antibacterial activity, specifically effective against mycobacteria. Although the exact mechanism of action of this compound hydrochloride is unknown, this compound hydrochloride inhibits the transfer of mycolic acids into the cell wall of bacteria, which impedes bacterial cell growth. This agent may also interfere with RNA synthesis or inhibit other cell metabolism, thereby preventing cell multiplication and causing cell death.
An antitubercular agent that inhibits the transfer of mycolic acids into the cell wall of the tubercle bacillus. It may also inhibit the synthesis of spermidine in mycobacteria. The action is usually bactericidal, and the drug can penetrate human cell membranes to exert its lethal effect. (From Smith and Reynard, Textbook of Pharmacology, 1992, p863)
Mechanism of Action
Target of Action
Ethambutol primarily targets the arabinosyltransferases (embA, embB, and embC) . These enzymes are crucial for the synthesis of the mycobacterial cell wall .
Mode of Action
This compound diffuses into Mycobacterium cells . Once inside the cell, it inhibits the arabinosyltransferases, preventing the formation of the cell wall components arabinogalactan and lipoarabinomannan . This inhibition prevents cell division .
Biochemical Pathways
The inhibition of arabinosyltransferases by this compound affects the peptidoglycan biosynthesis pathway . This pathway is responsible for the production of arabinogalactan and lipoarabinomannan, key components of the mycobacterial cell wall . The disruption of this pathway leads to a decrease in the number of binding sites for mycolic acid, leading to the accumulation of mycolic acid, trehalose monomycolate, and trehalose dimycolate .
Pharmacokinetics
This compound is approximately 75-80% orally bioavailable . A 25 mg/kg oral dose of this compound reaches a Cmax of 2-5 µg/mL, with a Tmax of 2-4 hours . The clearance and volume of distribution were estimated to be 77.4 liters/h and 76.2 liters, respectively . A G/A mutation with regard to CYP1A2 2159 G>A was associated with a 50% reduction in relative bioavailability .
Result of Action
The inhibition of arabinosyltransferases by this compound leads to a decrease in the concentrations of arabinogalactan in the cell wall . This reduces the number of binding sites for mycolic acid, leading to the accumulation of mycolic acid, trehalose monomycolate, and trehalose dimycolate . Reduced levels of lipoarabinomannan may interfere with mycobacterial interaction with host cells .
Action Environment
The action, efficacy, and stability of this compound can be influenced by various environmental factors. It’s important to note that the pharmacokinetics and pharmacodynamics of this compound can be affected by individual patient characteristics, including genetic polymorphisms .
Biochemical Analysis
Biochemical Properties
Ethambutol interacts with three membrane-embedded arabinosyltransferases — EmbA, EmbB, and EmbC — involved in the synthesis of the mycobacterial cell wall . It also targets the glutamate racemase (MurI), a crucial enzyme of phase I peptidoglycan (PG) biosynthesis pathway of Mycobacterium tuberculosis .
Cellular Effects
This compound exerts its effects by inhibiting the formation of cell wall components arabinogalactan and lipoarabinomannan, thereby preventing cell division . Decreased concentrations of arabinogalactan in the cell wall reduces the number of binding sites for mycolic acid, leading to the accumulation of mycolic acid, trehalose monomycolate, and trehalose dimycolate .
Molecular Mechanism
This compound binds to similar domains in EmbB and EmbC and thereby inhibits the arabinosyltransferase activity necessary for creation of components of the mycobacterial cell wall . It acts as a competitive inhibitor of substrate for binding to mycobacterial MurI protein .
Temporal Effects in Laboratory Settings
It is known that this compound’s inhibitory activity against tuberculosis is significant .
Metabolic Pathways
This compound is involved in the metabolic pathways related to the synthesis of the mycobacterial cell wall . It interacts with enzymes like EmbA, EmbB, and EmbC, and glutamate racemase (MurI) .
Biological Activity
Ethambutol (EMB) is a first-line antitubercular agent primarily used in the treatment of tuberculosis (TB). Its biological activity is characterized by its unique mechanism of action, pharmacokinetics, and potential adverse effects, particularly on ocular health. This article delves into the biological activity of this compound, highlighting its mechanisms, pharmacokinetics, case studies, and clinical findings.
This compound exhibits its antitubercular effects primarily through the inhibition of cell wall synthesis in Mycobacterium tuberculosis (M. tuberculosis). It specifically targets the enzyme arabinosyl transferase, which is crucial for the biosynthesis of arabinogalactan, a component of the mycobacterial cell wall. This inhibition leads to impaired cell wall integrity and ultimately bacterial death .
Synergistic Effects with Other Drugs
Recent studies have shown that this compound can enhance the efficacy of other antitubercular agents such as isoniazid (INH). This compound induces the expression of EtbR, a transcriptional repressor that modulates the expression of the inhA gene, thereby increasing INH susceptibility in M. tuberculosis. This synergistic action underscores the importance of combination therapy in TB treatment .
Pharmacokinetics
This compound's pharmacokinetics have been extensively studied, particularly in pediatric populations. A two-compartment model best describes its pharmacokinetics, with first-order elimination characteristics. In a cohort study involving 188 children receiving a median dose of 20.2 mg/kg, it was found that 54.8% were HIV-positive on antiretroviral therapy (ART). The study noted significant variability in plasma concentrations, which can influence therapeutic outcomes .
| Parameter | Value |
|---|---|
| Median Dose | 20.2 mg/kg |
| Age Range | 0.3 - 12.6 years |
| Percentage HIV+ | 54.8% |
Ocular Toxicity
One of the most significant adverse effects associated with this compound is ocular toxicity, particularly optic neuropathy. A retrospective study involving over 4,000 patients treated with this compound reported an incidence rate of 0.5% for this compound-related optic neuropathy (EON). Risk factors identified included older age and smoking status .
Case Study Findings:
- In a cohort of 3 patients with renal TB treated with therapeutic levels of EMB, all experienced severe vision loss; two remained legally blind after long-term follow-up .
- Another study found that among patients diagnosed with EON, cessation of this compound led to visual recovery in approximately 60% within an average recovery time of 6.9 months .
Clinical Implications
The implications of this compound’s biological activity are profound for clinical practice:
- Combination Therapy: The ability to enhance INH efficacy through EtbR modulation suggests that EMB should be used in combination therapies for more effective TB management.
- Monitoring for Toxicity: Given the potential for severe ocular toxicity, regular monitoring of vision is recommended for patients receiving this compound, especially those at higher risk due to age or comorbidities.
Scientific Research Applications
Primary Use in Tuberculosis Treatment
Ethambutol is a cornerstone drug in the multi-drug regimen for treating drug-susceptible pulmonary tuberculosis. It inhibits the synthesis of the bacterial cell wall, making it effective against Mycobacterium tuberculosis. The standard treatment typically combines this compound with other first-line antitubercular drugs such as isoniazid, rifampicin, and pyrazinamide during the intensive phase of therapy .
Pharmacokinetics
Recent studies have demonstrated that this compound effectively penetrates tuberculous lesions. In a rabbit model, it showed a lesion-to-plasma exposure ratio ranging from 9 to 12, indicating significant accumulation in diseased tissues . This property is essential for its efficacy in treating TB, as effective drug concentration at the site of infection is critical.
Emerging Applications in Non-Tuberculous Mycobacterial Infections
This compound has been endorsed for off-label use in treating non-tuberculous mycobacterial infections such as those caused by Mycobacterium avium complex, Mycobacterium kansasii, and Mycobacterium xenopi. Its role in these infections is particularly significant for patients who may have resistance to other first-line agents .
Optic Neuropathy
One notable side effect of this compound is its potential to cause optic neuropathy. A case study documented a patient who developed blurred vision after starting this compound therapy. Despite discontinuation of the drug, the patient's vision did not improve significantly, highlighting the need for monitoring visual function during treatment .
In a broader study involving over 4,000 patients treated with this compound for TB, it was found that approximately 0.5% experienced this compound-induced optic neuropathy. Risk factors included older age and prolonged duration of therapy .
Visual Assessment Studies
Research has shown that even subclinical changes can occur in patients receiving standard doses of this compound. For instance, a study indicated that nearly half of the patients exhibited changes in retinal nerve fiber layer thickness after six months of treatment . These findings suggest that regular ophthalmologic assessments are vital for early detection of potential side effects.
Resistance Patterns and Pharmacodynamics
This compound resistance can emerge, particularly in cases where it is used as monotherapy or when there is co-infection with resistant strains of Mycobacterium tuberculosis. Studies have indicated that efflux-pump mechanisms may contribute to this resistance . Understanding these patterns is crucial for optimizing treatment regimens and minimizing resistance development.
Table: Summary of this compound Applications
| Application Area | Description |
|---|---|
| Tuberculosis Treatment | First-line agent combined with other antitubercular drugs; inhibits cell wall synthesis. |
| Non-Tuberculous Mycobacterial Infections | Off-label use for infections like Mycobacterium avium complex. |
| Optic Neuropathy Monitoring | Risk of vision loss necessitates regular eye examinations during treatment. |
| Resistance Management | Understanding mechanisms to prevent development of drug resistance during therapy. |
Q & A
Basic Research Questions
Q. What experimental models are most appropriate for studying Ethambutol-induced ocular toxicity, and how can confounding variables be controlled?
Methodological Answer:
- Animal Models : Rodent models (e.g., Wistar rats) are widely used due to their physiological similarity to humans in optic nerve responses. Ensure dose equivalence calculations align with human therapeutic ranges (e.g., 15–25 mg/kg/day) .
- Confounding Control : Use sham-treated control groups and monitor variables like zinc/copper levels, as this compound chelates metal ions critical for retinal function . Histopathological scoring by blinded pathologists reduces observer bias .
- Validation : Pair in vivo models with in vitro retinal ganglion cell assays to confirm mechanistic pathways .
Q. How should researchers design studies to evaluate this compound’s reproductive toxicity while adhering to ethical guidelines?
Methodological Answer:
- PICOT Framework : Define Population (e.g., animal models), Intervention (this compound doses mimicking human exposure), Comparison (untreated controls), Outcomes (fertility rates, teratogenicity), and Timeframe (multigenerational studies) .
- Ethical Compliance : Follow GHS Category 1B protocols for reproductive toxicity, including mandatory PPE (gloves, respirators) and locked storage to prevent accidental exposure .
- Data Transparency : Use propensity score matching in retrospective analyses to minimize selection bias, as demonstrated in clinical cohort studies .
Advanced Research Questions
Q. How can contradictory data on this compound’s neurotoxicity mechanisms be resolved through systematic review and meta-analysis?
Methodological Answer:
- Data Synthesis : Conduct a PRISMA-guided review of studies linking this compound to copper/zinc dysregulation versus mitochondrial apoptosis pathways . Use tools like RevMan to calculate pooled effect sizes.
- Contradiction Analysis : Stratify studies by dosage (low vs. high) and exposure duration. For example, acute toxicity may involve metal chelation, while chronic exposure triggers oxidative stress .
- Sensitivity Analysis : Exclude underpowered studies or those with unvalidated biomarkers (e.g., non-blinded histopathology) to refine conclusions .
Q. What methodologies optimize the detection of this compound-resistant Mycobacterium tuberculosis strains in in vitro assays?
Methodological Answer:
- Culture Techniques : Use BACTEC MGIT 960 systems for drug susceptibility testing. Include ethionamide as a control to rule out cross-resistance .
- Molecular Validation : Amplify embB gene mutations (e.g., M306V) via PCR and correlate with minimum inhibitory concentrations (MICs). Use Sanger sequencing for confirmation .
- Statistical Rigor : Apply Kaplan-Meier survival analysis to compare bacterial growth curves between resistant and susceptible strains .
Q. Methodological Challenges and Solutions
Q. How can researchers address limitations in retrospective clinical data when analyzing this compound-associated adverse events?
Methodological Answer:
- Limitation Mitigation : Use propensity score matching to balance covariates (e.g., age, comorbidities) between HRE and HREZ treatment groups .
- Bias Reduction : Employ inverse probability weighting to account for missing data in adverse event reporting .
- Validation : Cross-reference electronic health records with laboratory-confirmed outcomes (e.g., hepatotoxicity via ALT/AST levels) .
Q. What strategies improve the reliability of apoptosis assays in this compound-treated animal models?
Methodological Answer:
- Standardization : Use hematoxylin-eosin (H&E) staining with semi-quantitative histopathological scoring by two independent pathologists .
- Advanced Techniques : Supplement with TUNEL assays to detect DNA fragmentation and caspase-3 immunohistochemistry for apoptotic pathway validation .
- Reproducibility : Archive raw data (e.g., microscopy images) in FAIR-compliant repositories for peer validation .
Q. Data Presentation and Reporting
Q. How should researchers structure appendices to enhance transparency in this compound toxicity studies?
Methodological Answer:
- Appendix Content : Include raw histopathology scores, dose-response curves, and statistical code (e.g., R/Python scripts) .
- Ethical Documentation : Detail Institutional Animal Care and Use Committee (IACUC) approvals and PPE protocols per GHS guidelines .
- Visual Aids : Use heatmaps to illustrate gene mutation frequencies in resistant strains or forest plots for meta-analyses .
Properties
IUPAC Name |
(2S)-2-[2-[[(2S)-1-hydroxybutan-2-yl]amino]ethylamino]butan-1-ol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H24N2O2/c1-3-9(7-13)11-5-6-12-10(4-2)8-14/h9-14H,3-8H2,1-2H3/t9-,10-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
AEUTYOVWOVBAKS-UWVGGRQHSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC(CO)NCCNC(CC)CO | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC[C@@H](CO)NCCN[C@@H](CC)CO | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H24N2O2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8023006, DTXSID901028179 | |
| Record name | Ethambutol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8023006 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (2R*,2'R*)-2,2'-(Ethane-1,2-diyldiimino)dibutan-1-ol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID901028179 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
204.31 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Ethambutol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014474 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Soluble in chloroform, methylene chloride; less soluble in benzene; sparingly soluble in water, 7.58e+00 g/L | |
| Record name | Ethambutol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00330 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ETHAMBUTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3078 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ethambutol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014474 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Ethambutol diffuses into _Mycobacterium_ cells. Once inside the cell, ethambutol inhibits the arabinosyltransferases (embA, embB, and embC), preventing formation of the cell wall components arabinogalactan and lipoarabinomannan, and preventing cell division. Decreased concentrations of arabinogalactan in the cell wall reduces the number of binding sites for mycolic acid, leading to the accumulation of mycolic acid, trehalose monomycolate, and trehalose dimycolate. Lipoarabinomannan is a component of a cell surface molecule involved in the interaction with host cells. Reduced levels of lipoarabinomannan may interfere with mycobacterial interaction with host cells., Ethambutol is bacteriostatic in action. Although the exact mechanism of action has not been fully elucidated, the drug appears to inhibit the synthesis of one or more metabolites in susceptible bacteria resulting in impairment of cellular metabolism, arrest of multiplication, and cell death. Ethambutol is active against susceptible bacteria only when they are undergoing cell division. | |
| Record name | Ethambutol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00330 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ETHAMBUTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3078 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals, WHITE, CRYSTALLINE POWDER | |
CAS No. |
74-55-5, 36697-71-9 | |
| Record name | (+)-Ethambutol | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=74-55-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Ethambutol [INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000074555 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ethambutol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00330 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Ethambutol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8023006 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (2R*,2'R*)-2,2'-(Ethane-1,2-diyldiimino)dibutan-1-ol | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID901028179 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Ethambutol | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.737 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | ETHAMBUTOL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/8G167061QZ | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | ETHAMBUTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3078 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ethambutol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014474 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
171.5-174.5, 87.5-88.8 °C, 88 °C | |
| Record name | Ethambutol | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00330 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ETHAMBUTOL | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3078 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ethambutol | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014474 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















