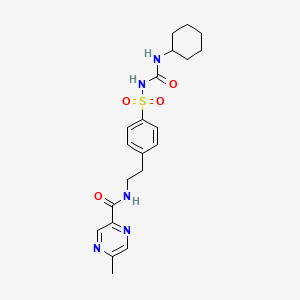
Glipizide
Overview
Description
Glipizide is an oral hypoglycemic agent belonging to the second-generation sulfonylurea drug class. It is primarily used to control blood sugar levels in patients with type 2 diabetes mellitus. This compound works by stimulating the pancreas to release insulin and increasing tissue sensitivity to insulin . It was first introduced in 1984 and is available under various brand names, including Glucotrol .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of glipizide involves several steps, starting from 4-[2-(5-methylpyrazine-2-carboxamido)ethyl]benzenesulfonamide. The key steps include chlorination, followed by reaction with cyclohexylamine in the presence of an iron catalyst and carbon monoxide . This method is simple and practicable, providing a new process route for the synthesis of this compound .
Industrial Production Methods: In industrial settings, this compound is often prepared using solvent evaporation methods to enhance its solubility. Solid dispersions of this compound are prepared using polymers such as polyvinyl pyrrolidone (PVP) and polyethylene glycol (PEG). The drug-to-polymer ratios and the conditions for solvent evaporation are optimized to achieve the desired solubility and dissolution rates .
Chemical Reactions Analysis
Types of Reactions: Glipizide undergoes various chemical reactions, including oxidation, reduction, and substitution. The compound is known to decompose upon heating, leading to the formation of 5-methyl-N-[2-(4-sulphamoylphenyl)ethyl]pyrazine-2-carboxamide .
Common Reagents and Conditions: Common reagents used in the reactions involving this compound include iron catalysts, carbon monoxide, and cyclohexylamine . The decomposition process involves gas evolution, including cyclohexanamine and carbon dioxide .
Major Products: The major products formed from the decomposition of this compound include 5-methyl-N-[2-(4-sulphamoylphenyl)ethyl]pyrazine-2-carboxamide .
Scientific Research Applications
Primary Treatment for Type 2 Diabetes
- Indication : Glipizide is indicated for adults with type 2 diabetes as an adjunct to diet and exercise. It is often prescribed when glycemic control is inadequate with lifestyle modifications alone .
- Combination Therapy : It can be used in conjunction with other antidiabetic medications like metformin, especially in cases where monotherapy does not achieve desired HbA1c levels .
Efficacy in Glycemic Control
- In clinical studies, this compound has shown significant reductions in fasting blood glucose and HbA1c levels when used as part of a comprehensive diabetes management plan. A meta-analysis indicated that this compound effectively reduced HbA1c by approximately 1-2% compared to placebo .
Cardiovascular Outcomes
- Recent studies have evaluated the cardiovascular safety of this compound, particularly in patients with a high risk of cardiovascular disease. While older sulfonylureas were associated with increased cardiovascular risks, newer studies suggest that this compound may have a neutral effect on cardiovascular outcomes, making it a safer option for patients with comorbid conditions .
Case Study 1: Efficacy in Elderly Patients
A study involving elderly patients with type 2 diabetes demonstrated that this compound effectively lowered blood glucose levels while maintaining a favorable safety profile. The patients showed improved glycemic control without significant adverse effects over a six-month period .
Case Study 2: Combination Therapy
In another case study, a patient initially treated with metformin alone was switched to a combination of metformin and this compound after failing to achieve target HbA1c levels. The addition of this compound led to a significant reduction in HbA1c from 8.5% to 6.9% within three months, highlighting its effectiveness as an adjunct therapy .
Side Effects
While this compound is generally well-tolerated, it can cause several side effects:
- Hypoglycemia : The most common adverse effect, particularly if meals are skipped or if the patient engages in excessive physical activity without adjusting their medication .
- Gastrointestinal Issues : Nausea, vomiting, and diarrhea can occur but are usually mild and transient .
- Weight Gain : Some patients may experience weight gain due to increased insulin secretion and decreased glucose utilization by tissues .
Mechanism of Action
Glipizide exerts its effects by sensitizing the beta cells of the pancreatic islets of Langerhans to insulin response. It partially blocks potassium channels among beta cells, leading to cell depolarization and the opening of voltage-gated calcium channels. The resulting calcium influx encourages insulin release from beta cells . This mechanism helps in lowering blood glucose levels by promoting insulin secretion and increasing tissue sensitivity to insulin .
Comparison with Similar Compounds
Similar Compounds: Glipizide is often compared with other sulfonylurea drugs such as glimepiride and glyburide. It is also compared with non-sulfonylurea drugs like metformin and incretin mimetics such as semaglutide .
Uniqueness: Compared to other sulfonylureas, this compound has a rapid absorption and onset of action with the shortest half-life and duration of action. This reduces the risk of long-lasting hypoglycemia, which is often observed with other blood glucose-lowering agents . Additionally, this compound’s unique chemical structure, which includes a non-polar side chain, enhances its hypoglycemic potency .
Biological Activity
Glipizide is an oral hypoglycemic agent belonging to the sulfonylurea class, primarily used in the management of type 2 diabetes mellitus (T2DM). Its biological activity involves several mechanisms that contribute to its efficacy in lowering blood glucose levels. This article explores the pharmacodynamics, pharmacokinetics, clinical efficacy, and potential interactions of this compound, supported by data tables and case studies.
This compound functions as an insulin secretagogue , stimulating insulin release from pancreatic beta cells. The primary mechanism involves:
- Binding to Sulfonylurea Receptors : this compound binds to the sulfonylurea receptor (SUR1) on the beta-cell membrane, leading to the closure of ATP-sensitive potassium channels. This causes depolarization of the cell membrane and opening of voltage-gated calcium channels, resulting in increased intracellular calcium levels and subsequent insulin secretion .
- Extrapancreatic Effects : Apart from stimulating insulin release, this compound enhances glucose uptake in peripheral tissues such as skeletal muscle and adipose tissue. It also inhibits hepatic glucose output and lipolysis, thereby contributing to overall glucose homeostasis .
Pharmacokinetics
The pharmacokinetic profile of this compound is characterized by:
- Absorption : this compound is rapidly absorbed after oral administration, with peak plasma concentrations occurring approximately 1-2 hours post-dose.
- Metabolism : It undergoes extensive hepatic metabolism primarily via cytochrome P450 enzymes (CYP2C9), producing inactive metabolites that are excreted in urine.
- Half-Life : The elimination half-life ranges from 2 to 4 hours, necessitating multiple daily doses for sustained effect .
Clinical Efficacy
Numerous clinical studies have assessed the efficacy of this compound in managing T2DM. A notable study involved two multicenter clinical trials assessing an extended-release formulation (GITS) of this compound:
| Dosage (mg) | Fasting Plasma Glucose Reduction (mg/dl) | HbA1c Reduction (%) |
|---|---|---|
| 5 | -57 | -1.50 |
| 10 | -65 | -1.70 |
| 20 | -74 | -1.82 |
| Placebo | No significant change | No significant change |
The results indicated that all doses significantly reduced fasting plasma glucose (FPG) and HbA1c compared to placebo, with maximal efficacy observed at doses of 20 mg for FPG and 5 mg for HbA1c .
Case Studies
A multi-center assessment involving 592 patients demonstrated that this compound effectively controlled blood glucose levels in both treatment-naïve and previously treated patients. The study reported:
- A rapid decrease in post-prandial blood glucose levels within the first week.
- A mean increase in body weight of approximately 1 kg over 12 weeks.
- High patient tolerance with only a small percentage (4.7%) experiencing side effects, primarily mild hypoglycemia .
Pharmacogenomic Considerations
Pharmacogenomic studies have highlighted genetic factors influencing this compound efficacy. For instance, variations in genes related to drug metabolism can affect individual responses to this compound treatment, emphasizing the need for personalized medicine approaches in diabetes management .
Herb-Drug Interactions
Recent research has explored potential interactions between this compound and herbal compounds. For example, Andrographis paniculata (APE) was shown to enhance the pharmacokinetic parameters of this compound in diabetic rats, suggesting a synergistic effect that could improve therapeutic outcomes . Conversely, certain herbs may inhibit its metabolism, leading to altered bioavailability and efficacy.
Q & A
Basic Research Questions
Q. What experimental methodologies are recommended for quantifying glipizide in pharmacokinetic studies?
High-performance liquid chromatography (HPLC) with UV detection is widely used due to its specificity and sensitivity. Key parameters include:
- Column : C18 reverse-phase (e.g., 250 mm × 4.6 mm, 5 µm particle size).
- Mobile phase : Acetonitrile-phosphate buffer (pH 3.0) in a 40:60 ratio.
- Flow rate : 1.0 mL/min.
- Detection wavelength : 225 nm. Validation should follow ICH guidelines for linearity (1–50 µg/mL), precision (RSD <2%), and recovery (>95%) .
Q. How should preclinical studies be designed to evaluate this compound’s mechanism of action in glucose regulation?
- In vitro : Use pancreatic β-cell lines (e.g., INS-1) to measure insulin secretion under varying glucose concentrations (5–25 mM) with this compound (1–100 nM).
- In vivo : Employ streptozotocin-induced diabetic rodent models, administering this compound (2.5–10 mg/kg) and monitoring blood glucose via glucometers at 0, 30, 60, and 120 minutes post-dose. Include control groups (vehicle and healthy animals) and validate results with Western blotting for SUR1 receptor expression .
Q. What statistical approaches are appropriate for analyzing this compound’s efficacy in clinical trials?
Use ANOVA for comparing HbA1c reduction across treatment arms (e.g., this compound vs. placebo), followed by post-hoc Tukey tests. For time-series glucose data, mixed-effects models account for intra-patient variability. Sample size calculations should assume a 0.5% HbA1c difference with 80% power and α=0.05 .
Advanced Research Questions
Q. How can contradictory findings on this compound’s cardiovascular risks be resolved in meta-analyses?
- Data stratification : Separate studies by patient comorbidities (e.g., CVD history) and this compound dosage (≤10 mg/day vs. >10 mg/day).
- Sensitivity analysis : Exclude trials with high attrition rates (>20%) or unblinded designs.
- Meta-regression : Investigate confounding variables like concomitant metformin use. Tools like Cochrane’s ROBINS-I should assess bias .
Q. What in silico strategies are effective for predicting this compound-drug interactions at the CYP2C9 enzyme?
- Molecular docking : Use AutoDock Vina to simulate this compound binding to CYP2C9 (PDB ID: 1OG5).
- Pharmacophore modeling : Identify critical interaction sites (e.g., sulfonylurea moiety).
- Machine learning : Train random forest models on datasets like DrugBank to predict inhibition constants (Ki). Validate with in vitro microsomal assays .
Q. How do genetic polymorphisms (e.g., CYP2C9*3) impact this compound pharmacokinetics across diverse populations?
- Study design : Recruit cohorts stratified by CYP2C9 genotypes (e.g., *1/*1, *1/*3).
- PK parameters : Calculate AUC0–∞, Cmax, and t1/2 via non-compartmental analysis (WinNonlin).
- Statistical modeling : Use NONMEM for population PK analysis, incorporating covariates like BMI and renal function. Ethical considerations: Ensure informed consent addresses genetic data privacy .
Q. Methodological Guidance
Q. What criteria define a robust literature review for identifying gaps in this compound research?
- Search strategy : Combine terms (e.g., “this compound AND pharmacokinetics NOT metformin”) across PubMed, Scopus, and Web of Science, limited to 2010–2025.
- Screening : PRISMA flowchart to document inclusion/exclusion.
- Quality assessment : Apply Newcastle-Ottawa Scale for observational studies. Highlight understudied areas (e.g., long-term neurocognitive effects) .
Q. How can researchers optimize this compound formulation stability in dissolution studies?
- Accelerated stability testing : Store tablets at 40°C/75% RH for 6 months. Monitor degradation products via LC-MS.
- Dissolution media : Use pH 1.2 (HCl), 4.5 (acetate), and 6.8 (phosphate) to simulate gastrointestinal conditions.
- Kinetic modeling : Apply Weibull equations to predict shelf-life .
Q. Data Presentation & Reproducibility
Q. What are the best practices for reporting this compound clinical trial data to ensure reproducibility?
- Raw data : Deposit in repositories like ClinicalTrials.gov or Dryad.
- Preprocessing : Document outlier removal criteria (e.g., values beyond ±3 SD).
- Visualization : Use Forest plots for meta-analyses and heatmaps for gene expression correlations. Follow CONSORT guidelines for flow diagrams and EQUATOR Network standards .
Q. How should conflicting in vitro vs. in vivo this compound efficacy data be reconciled?
- Dose translation : Apply allometric scaling (e.g., body surface area adjustment) between cell culture (µM) and animal models (mg/kg).
- Tissue distribution : Measure this compound concentrations in pancreatic tissue via LC-MS/MS.
- Mechanistic studies : Use knockout mice (e.g., SUR1−/−) to isolate target effects .
Properties
IUPAC Name |
N-[2-[4-(cyclohexylcarbamoylsulfamoyl)phenyl]ethyl]-5-methylpyrazine-2-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C21H27N5O4S/c1-15-13-24-19(14-23-15)20(27)22-12-11-16-7-9-18(10-8-16)31(29,30)26-21(28)25-17-5-3-2-4-6-17/h7-10,13-14,17H,2-6,11-12H2,1H3,(H,22,27)(H2,25,26,28) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
ZJJXGWJIGJFDTL-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=CN=C(C=N1)C(=O)NCCC2=CC=C(C=C2)S(=O)(=O)NC(=O)NC3CCCCC3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C21H27N5O4S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID0040676 | |
| Record name | Glipizide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0040676 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
445.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Glipizide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015200 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
>66.8 [ug/mL] (The mean of the results at pH 7.4), 1.64e-02 g/L | |
| Record name | SID855947 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Glipizide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01067 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Glipizide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015200 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Type 2 diabetes mellitus (T2DM) is a chronic metabolic disorder with increasing prevalence worldwide. Characterized by higher-than-normal levels of blood glucose, T2DM is a complex disorder that arises from the interaction between genetic, environmental and behavioral risk factors. Insulin is a peptide hormone that plays a critical role in regulating blood glucose levels. In response to high blood glucose levels, insulin promotes the uptake of glucose into the liver, muscle cells, and fat cells for storage. Although there are multiple events occurring that lead to the pathophysiology of T2DM, the disorder mainly involves insulin insensitivity as a result of insulin resistance, declining insulin production, and eventual failure of beta cells of pancreatic islets that normally produce insulin. Early management with lifestyle intervention, such as controlled diet and exercise, is critical in reducing the risk of long-term secondary complications, such as cardiovascular mortality. Glipizide, like other sulfonylurea drugs, is an insulin secretagogue, which works by stimulating the insulin release from the pancreatic beta cells thereby increasing the plasma concentrations of insulin. Thus, the main therapeutic action of the drug depends on the functional beta cells in the pancreatic islets. Sulfonylureas bind to the sulfonylurea receptor expressed on the pancreatic beta-cell plasma membrane, leading to the closure of the ATP-sensitive potassium channel and reduced potassium conductance. This results in depolarization of the pancreatic beta cell and opening of the voltage-sensitive calcium channels, promoting calcium ion influx. Increased intracellular concentrations of calcium ions in beta cells stimulates the secretion, or exocytosis, of insulin granules from the cells. Apart from this main mechanism of action, the blood-glucose-lowering effect of glipizide involves increased peripheral glucose utilization via stimulating hepatic gluconeogenesis and by increasing the number and sensitivity of insulin receptors. | |
| Record name | Glipizide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01067 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
29094-61-9 | |
| Record name | Glipizide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=29094-61-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Glipizide [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0029094619 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Glipizide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01067 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | glipizide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759120 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Glipizide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0040676 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Glipizide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.044.919 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | GLIPIZIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/X7WDT95N5C | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Glipizide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015200 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
200-203, 208 - 209 °C | |
| Record name | Glipizide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01067 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Glipizide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015200 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















