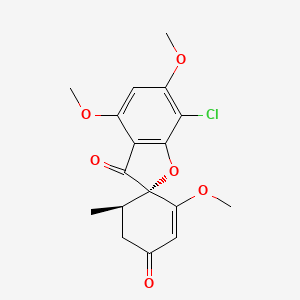
Griseofulvin
Overview
Description
Griseofulvin is a natural antifungal compound first isolated from the mold Penicillium griseofulvum in 1939 . It has been widely used since its commercial introduction in 1959 to treat dermatophyte infections, which are fungal infections affecting the skin, hair, and nails . This compound is known for its ability to inhibit fungal cell mitosis, making it an effective fungistatic agent .
Preparation Methods
Griseofulvin is synthesized through a biosynthetic pathway involving the combination of one acetyl coenzyme A and six malonyl coenzyme A molecules to form a heptaketide backbone . The industrial production of this compound typically involves fermentation processes using Penicillium species . The compound can also be prepared through chemical synthesis, although this method is less common due to the complexity and cost involved .
Chemical Reactions Analysis
Griseofulvin undergoes various chemical reactions, including oxidation, reduction, and substitution reactions. Common reagents used in these reactions include oxidizing agents like hydrogen peroxide and reducing agents such as sodium borohydride . Major products formed from these reactions include desmethylthis compound derivatives, which have been studied for their potential biological activities .
Scientific Research Applications
Griseofulvin is an antifungal polyketide metabolite derived from ascomycetes, initially isolated from Penicillium griseofulvum in 1939 . Since its commercial introduction in 1959, it has been used to treat dermatophyte infections . Beyond its antifungal properties, this compound has gained interest for its potential in disrupting mitosis and cell division in cancer cells, inhibiting hepatitis C virus replication, enhancing ACE2 function, promoting vascular vasodilation, and improving capillary blood flow .
Antifungal Applications
This compound is used as an antifungal drug to treat ringworm and dermatophyte infections in humans and animals . It is also the drug of choice for tinea capitis, onychomycosis, and other superficial fungal infections .
Antitumor Applications
This compound has garnered attention as a potential agent in cancer chemotherapy due to its low toxicity . It inhibits tumor growth and cancer cell proliferation by suppressing spindle microtubule dynamics, inducing mitotic arrest, and causing cell death in multipolar spindles, without harming fibroblasts and keratinocytes with normal centrosome composition . this compound binds to the αβ intra-dimer tubulin interface, leading to mitotic abnormalities like misaligned chromosomes and multipolar spindles, resulting in fragmented nuclei and apoptosis .
Antiviral Applications
This compound may inhibit hepatitis C virus replication by interfering with microtubule polymerization in human cells . Molecular docking analysis suggests that this compound and its derivatives can bind to SARS-CoV-2 main protease, RNA-dependent RNA polymerase (RdRp), and spike protein receptor-binding domain (RBD), suggesting potential inhibitory effects on SARS-CoV-2 entry and viral replication .
Other Applications
This compound is used as a crop protectant in agriculture to prevent fungal colonization and infection .
Anti-malarial Applications
This compound impairs the intraerythrocytic development of P. falciparum . In vitro studies showed that P. falciparum could not grow in human red blood cells from subjects taking this compound . While initial studies showed promise, subsequent clinical trials showed no significant inhibition of parasite growth when subjects were treated with this compound .
Acne Treatment
This compound has been investigated for treating acne vulgaris . Clinical trials have shown improvement in inflammatory acne lesions (papules and pustules) but less significant effects on non-inflamed lesions (comedones, cysts, and nodules) .
Case Study 1: Pediatric Tinea Capitis
A study reported successful treatment of recalcitrant pediatric tinea capitis using this compound . After 10 weeks of treatment, skin lesions nearly disappeared, and remarkable hair regrowth was observed .
Research Finding: Anti-malarial Activity
Ex vivo studies demonstrated that red blood cells (RBCs) collected from subjects who had been orally administered clinical doses of this compound accumulated the drug in levels sufficient to impair parasite growth .
Pharmacokinetic profiling showed that this compound levels in plasma and RBCs peaked within one day of drug intake, with RBC levels (59–143 μg/L) comparable to those required in vitro to inhibit parasite growth . RBCs from a subject given a single 2,000 mg dose inhibited parasite growth for up to 2 days, with parasite growth inhibition gradually declining as plasma and RBC levels of this compound decreased .
Research Finding: Acne Vulgaris Treatment
Mechanism of Action
Griseofulvin exerts its antifungal effects by inhibiting fungal cell mitosis at metaphase . It binds to tubulin, a protein that forms microtubules, and disrupts the formation of the mitotic spindle, preventing the separation of chromosomes during cell division . This action makes the fungal cells unable to replicate and spread . This compound also binds to keratin in human cells, making the keratin resistant to fungal invasion .
Comparison with Similar Compounds
Griseofulvin is often compared to other antifungal agents such as terbinafine and itraconazole . Unlike this compound, which inhibits mitosis, terbinafine works by inhibiting the enzyme squalene epoxidase, leading to the accumulation of toxic squalene in fungal cells . Itraconazole, on the other hand, inhibits the synthesis of ergosterol, an essential component of fungal cell membranes . These differences in mechanisms of action highlight the uniqueness of this compound in targeting fungal cell division .
Similar compounds to this compound include:
- Terbinafine
- Itraconazole
- Fluconazole
- Ketoconazole
This compound’s unique mechanism of action and its ability to bind to keratin make it a valuable antifungal agent, particularly for treating dermatophyte infections .
Biological Activity
Griseofulvin is an antifungal agent derived from the mold Penicillium griseofulvum, primarily used to treat dermatophyte infections. Since its introduction in 1959, it has been recognized for its fungistatic properties, inhibiting fungal growth by interfering with mitosis and cell division. Recent studies have expanded its potential applications, suggesting roles in cancer treatment and viral infections.
This compound exerts its antifungal effects by binding to tubulin, disrupting the mitotic spindle formation during cell division. This action not only inhibits fungal growth but also has implications for human cells, particularly in cancer research. Additionally, molecular docking studies indicate that this compound may bind effectively to proteins involved in the SARS-CoV-2 lifecycle, suggesting potential antiviral properties against COVID-19 .
Table 1: Mechanisms of this compound Action
| Mechanism | Description |
|---|---|
| Tubulin Binding | Disrupts mitotic spindle formation, inhibiting fungal cell division |
| Antiviral Activity | Potential binding to SARS-CoV-2 proteins (main protease, RdRp) |
| Vasodilation | May enhance ACE2 function, contributing to vascular health |
Antifungal Efficacy
This compound is primarily indicated for dermatophyte infections such as tinea capitis and tinea corporis. A meta-analysis comparing this compound to terbinafine showed that both treatments are effective, with this compound being particularly effective against infections caused by Microsporum species .
Case Studies
- Tinea Capitis Treatment : A study involving 175 patients treated with this compound showed a clearance rate of 13.6% among those with tinea corporis caused by Trichophyton rubrum. Side effects were minimal and transient .
- Cancer Research : In a cohort study examining the long-term effects of this compound, one patient developed chronic granulocytic leukemia after treatment for skin infections. This raised concerns regarding potential carcinogenic effects, warranting further investigation into long-term use .
Table 2: Summary of Clinical Findings on this compound
Additional Biological Activities
Recent research has suggested that this compound may have additional biological activities beyond antifungal effects:
- Antiviral Properties : this compound shows promise in inhibiting the replication of hepatitis C virus and SARS-CoV-2, indicating a potential role in treating viral infections .
- Anti-Plasmodial Activity : Studies have shown that this compound can impair the growth of Plasmodium falciparum, the parasite responsible for malaria, through mechanisms involving heme metabolism disruption .
Table 3: Broader Biological Activities of this compound
| Activity | Target Organism/Process | Implications |
|---|---|---|
| Antiviral | Hepatitis C, SARS-CoV-2 | Potential for repurposing in viral therapies |
| Anti-Plasmodial | Plasmodium falciparum | Possible use in malaria treatment |
Q & A
Basic Research Questions
Q. What are the key challenges in optimizing Griseofulvin synthesis for scalable production, and what methodological approaches address them?
this compound synthesis involves a nonreducing polyketide synthase (gsfA) to form the heptaketide backbone, followed by methylation via O-methyltransferases (gsfB/gsfC) . Challenges include low yields in benzophenone intermediate formation and regioselective methylation. Methodological improvements include:
- Enzyme engineering : Modifying gsfA/gfsB activity to enhance catalytic efficiency .
- Chemoenzymatic routes : Combining microbial synthesis with chemical steps (e.g., alcoholysis of griseofulvic acid) to improve scalability .
- Crystallographic analysis : Using X-ray diffraction (space group P41, Z=4) to guide stereochemical control .
Q. How does this compound’s poor aqueous solubility impact preclinical studies, and what formulation strategies mitigate this limitation?
this compound’s solubility in aqueous buffers is ≤8.63·10⁻⁷ mol/L, necessitating advanced formulation techniques :
- Ultramicrosizing : Reduces particle size to 1–2 µm, achieving bioequivalence with lower doses .
- Solid dispersions : Incorporation into PEG matrices increases dissolution rates by 3–5× via fusion/solvent methods .
- Liposomal encapsulation : Enhances GI absorption (2.6× higher Cmax vs. suspensions) . Methodological validation includes FT-IR spectroscopy and DSC to confirm amorphous state stability .
Q. What experimental models are used to study this compound’s antifungal mechanism, and how do they reconcile conflicting data on microtubule disruption vs. nucleic acid synthesis inhibition?
this compound’s fungistatic action involves microtubule binding (α/β-tubulin) and mitotic arrest . Contradictions arise from species-specific effects:
- In vitro fungal models : Microsporum spp. show microtubule depolymerization, while Trichophyton spp. exhibit RNA synthesis inhibition .
- Yeast assays : S. cerevisiae studies reveal SGS1 gene repression, linking mitotic disruption to genomic instability .
- Human cell lines : HeLa cells demonstrate metaphase arrest via suppression of microtubule dynamic instability (IC50 = 20 µM) .
Advanced Research Questions
Q. How can researchers resolve contradictory structure-activity relationship (SAR) data for this compound analogs targeting cancer cells?
Disparate SAR data arise from divergent assays (e.g., fungal vs. mammalian microtubule affinity) . Methodological solutions include:
- 2D NMR/X-ray crystallography : Confirm stereochemical assignments (e.g., 4/6-methoxy positional isomers) .
- Phenotypic screening : Centrosomal clustering inhibition (IC50 = 5 µM) identifies analogs with dual antifungal/anticancer activity .
- Dynamic instability assays : Quantify microtubule rescue/frequency rates to differentiate mechanisms from vinca alkaloids .
Q. What experimental designs are critical for evaluating this compound’s ecological toxicity in agricultural or environmental contexts?
Ecotoxicity studies require multispecies assays:
- Soil microbiota : 16S rRNA sequencing reveals bacterial dominance shifts (bacteria > actinomycetes > fungi) at 1,000 mg/kg .
- Enzyme inhibition : Dose-dependent urease suppression (IC50 = 200 mg/L) and dehydrogenase biphasic effects .
- Plant models : Arabidopsis root elongation assays show minimal phytotoxicity below 200 mg/L .
Q. How does this compound’s pharmacokinetic profile influence its repurposing for oncology, and what combination therapies are methodologically viable?
this compound’s low oral bioavailability (27–72.5%) and hepatic metabolism to 6-DMG limit monotherapy efficacy . Strategies include:
- Adjuvant combinations : Synergy with taxanes (e.g., paclitaxel) via complementary microtubule stabilization/destabilization .
- Nanocarrier systems : PEGylated liposomes improve tumor targeting and reduce hepatic first-pass metabolism .
- Metabolite profiling : LC-MS/MS quantifies 6-DMG levels to optimize dosing schedules .
Q. What advanced techniques elucidate this compound’s species-specific efficacy against dermatophytes (e.g., Microsporum vs. Trichophyton)?
Mechanistic divergence is probed via:
- Proteomic profiling : Tubulin isoform expression differences in Microsporum (β-tubulin dominance) vs. Trichophyton (α-tubulin mutations) .
- Sweat simulation assays : Stratum corneum "wick effect" enhances drug transfer in Microsporum-infected models .
- Comparative genomics : Trichophyton CYP450 upregulation correlates with this compound resistance .
Q. Methodological Notes
Properties
IUPAC Name |
(2S,5'R)-7-chloro-3',4,6-trimethoxy-5'-methylspiro[1-benzofuran-2,4'-cyclohex-2-ene]-1',3-dione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C17H17ClO6/c1-8-5-9(19)6-12(23-4)17(8)16(20)13-10(21-2)7-11(22-3)14(18)15(13)24-17/h6-8H,5H2,1-4H3/t8-,17+/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
DDUHZTYCFQRHIY-RBHXEPJQSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1CC(=O)C=C(C12C(=O)C3=C(O2)C(=C(C=C3OC)OC)Cl)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@@H]1CC(=O)C=C([C@]12C(=O)C3=C(O2)C(=C(C=C3OC)OC)Cl)OC | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C17H17ClO6 | |
| Record name | GRISEOFULVIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20442 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID8020674 | |
| Record name | Griseofulvin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8020674 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
352.8 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Griseofulvin appears as white to pale cream-colored crystalline powder. Odorless or almost odorless. Tasteless. Sublimes without decomposition at 410 °F. (NTP, 1992), Solid | |
| Record name | GRISEOFULVIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20442 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Griseofulvin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014544 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
less than 1 mg/mL at 70 °F (NTP, 1992), SOL IN N,N-DIMETHYLFORMAMIDE @ 25 °C: 12-14 G/100 ML; SLIGHTLY SOL IN ETHANOL, CHLOROFORM, METHANOL, ACETIC ACID, ACETONE, BENZENE, & ETHYL ACETATE; PRACTICALLY INSOL IN WATER & PETROLEUM ETHER, 5.04e-02 g/L | |
| Record name | GRISEOFULVIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20442 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Griseofulvin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00400 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | GRISEOFULVIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1722 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Griseofulvin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014544 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Griseofulvin is fungistatic, however the exact mechanism by which it inhibits the growth of dermatophytes is not clear. It is thought to inhibit fungal cell mitosis and nuclear acid synthesis. It also binds to and interferes with the function of spindle and cytoplasmic microtubules by binding to alpha and beta tubulin. It binds to keratin in human cells, then once it reaches the fungal site of action, it binds to fungal microtubes thus altering the fungal process of mitosis., Fungistatic; griseofulvin inhibits fungal cell mitosis by causing disruption of the mitotic spindle structure, thereby arresting the metaphase of cell division. It is deposited in varying concentrations in the keratin precursor cells of skin, hair, and nails, rendering the keratin resistant to fungal invasion. As the infected keratin is shed, it is replaced with healthy tissue. | |
| Record name | Griseofulvin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00400 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | GRISEOFULVIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1722 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
STOUT OCTAHEDRA OR RHOMBS FROM BENZENE, WHITE TO CREAMY POWDER, COLORLESS CRYSTALLINE SOLID | |
CAS No. |
126-07-8, 2884-22-2 | |
| Record name | GRISEOFULVIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20442 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Griseofulvin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=126-07-8 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | rel-(1′R,6′S)-7-Chloro-2′,4,6-trimethoxy-6′-methylspiro[benzofuran-2(3H),1′-[2]cyclohexene]-3,4′-dione | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=2884-22-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Griseofulvin [USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000126078 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Griseofulvin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00400 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | griseofulvin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=755822 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Griseofulvin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8020674 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Griseofulvin | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.004.335 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | GRISEOFULVIN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/32HRV3E3D5 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | GRISEOFULVIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1722 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Griseofulvin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014544 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
428 °F (NTP, 1992), 220 °C | |
| Record name | GRISEOFULVIN | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20442 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Griseofulvin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00400 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | GRISEOFULVIN | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/1722 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Griseofulvin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014544 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















