Fingolimod
Overview
Description
Fingolimod is a sphingosine-1-phosphate receptor modulator used primarily in the treatment of relapsing-remitting multiple sclerosis. It was approved by the Food and Drug Administration in 2010 and is known for its ability to reduce the frequency of relapses and delay the progression of physical disability in patients with multiple sclerosis .
Preparation Methods
Synthetic Routes and Reaction Conditions: Fingolimod can be synthesized starting from n-octylbenzene and 3-nitropropionic acid. The synthetic route involves a sequence of reactions including Friedel-Crafts acylation, reduction, and double Henry reaction, followed by hydrogenation. This method yields this compound with a yield of 31% and an overall atom economy of 82.7% .
Industrial Production Methods: In industrial settings, this compound is often synthesized using a reverse phase liquid chromatography method. This involves the use of a phosphate buffer, methanol, and acetonitrile as eluents, with separation achieved using a specific column and temperature conditions .
Chemical Reactions Analysis
Types of Reactions: Fingolimod undergoes various chemical reactions, including oxidation, reduction, and substitution. It forms charge transfer complexes with different electron acceptor reagents, such as 7,7,8,8-tetracyanoquinodimethane, tetrachloro 1,4-benzoquinone, and tetracyanoethylene .
Common Reagents and Conditions: The reactions typically involve reagents like orthophosphoric acid, methanol, and acetonitrile under controlled temperature and pH conditions .
Major Products: The major products formed from these reactions include stable anions that can be measured spectrophotometrically .
Scientific Research Applications
Multiple Sclerosis Treatment
Overview : Fingolimod was the first oral disease-modifying therapy approved for relapsing forms of multiple sclerosis. It works by sequestering lymphocytes in lymph nodes, thus reducing their availability to enter the central nervous system and cause inflammation.
Clinical Efficacy :
- Phase III Trials : In pivotal trials like TRANSFORMS and FREEDOMS, this compound demonstrated significant reductions in annual relapse rates compared to interferon beta-1a and placebo groups. The annualized relapse rate was significantly lower in patients treated with this compound (0.5 mg) compared to those on interferon beta-1a (0.17 vs 0.27) .
- Long-term Studies : Extended follow-ups show sustained efficacy over years, maintaining low rates of disease activity and disability progression .
Neurodegenerative Diseases
Recent studies have highlighted this compound's potential beyond multiple sclerosis, particularly in neurodegenerative diseases such as Alzheimer's disease and Huntington's disease.
Huntington's Disease : Research from the University of Barcelona demonstrated that this compound restores hippocampal synaptic plasticity and improves memory function in mouse models of Huntington's disease. This suggests a neuroprotective role that could be further explored in clinical settings .
Alzheimer’s Disease and Parkinson’s Disease : this compound has shown promise in modulating neuroinflammation and may play a role in protecting against neuronal loss associated with these diseases. Its ability to influence pathways related to apoptosis and autophagy could be beneficial in managing neurodegenerative conditions .
Psychiatric Disorders
This compound is being investigated for its potential use as an adjunctive treatment for psychiatric disorders like schizophrenia.
Clinical Trials : A randomized controlled trial indicated that this compound combined with risperidone improved symptoms of schizophrenia more effectively than risperidone alone. The study reported significant improvements in both positive and negative symptoms without major adverse effects .
Cancer Therapy
This compound is being explored for its immunomodulatory effects in cancer treatment.
Mechanisms of Action : It inhibits histone deacetylases and modulates immune responses that could enhance anti-tumor immunity. Research indicates that this compound may induce apoptosis in certain cancer cell lines, suggesting a potential role as an adjunct therapy in oncology .
Safety Profile and Side Effects
This compound is generally well-tolerated; however, there are notable side effects such as macular edema, particularly in patients with a history of uveitis . Regular ophthalmic evaluations are recommended during treatment.
Mechanism of Action
Fingolimod exerts its effects by modulating sphingosine-1-phosphate receptors. It sequesters lymphocytes in lymph nodes, preventing them from contributing to an autoimmune reaction. This modulation reduces the rate of relapses in multiple sclerosis by approximately one-half over a two-year period . The active metabolite, this compound-phosphate, binds to sphingosine-1-phosphate receptors, inducing their downregulation and preventing lymphocyte egress from lymphoid tissues .
Comparison with Similar Compounds
Dimethyl fumarate: Another oral disease-modifying treatment for multiple sclerosis, comparable in effectiveness to Fingolimod.
Sphingosine: A naturally occurring sphingoid base with antimicrobial properties.
Uniqueness: this compound is unique due to its specific mechanism of action involving sphingosine-1-phosphate receptor modulation, which is distinct from other immunosuppressants. Its ability to sequester lymphocytes and prevent their egress from lymphoid tissues sets it apart from other treatments .
Biological Activity
Fingolimod, also known as FTY720, is a sphingosine-1-phosphate (S1P) receptor modulator that has gained prominence as a therapeutic agent for multiple sclerosis (MS). Initially approved in 2010, it was the first oral medication for relapsing forms of MS. Beyond its primary use, recent research has uncovered a variety of biological activities associated with this compound, including antibacterial properties and effects on immune modulation. This article aims to provide a comprehensive overview of the biological activity of this compound, supported by data tables, case studies, and detailed research findings.
This compound acts by binding to S1P receptors, primarily S1P1, which leads to the sequestration of lymphocytes in lymph nodes and reduces their circulation in the bloodstream. This mechanism is pivotal in its efficacy against MS but also underlies its potential effects on other biological systems.
Immune Modulation
This compound's immunomodulatory effects have been extensively studied. It interferes with various immune pathways, including:
- Th2 Response : this compound has been shown to modulate Th2 responses via IL-33/ST2 signaling pathways, indicating its role in allergic and autoimmune conditions .
- Lymphocyte Circulation : By preventing lymphocyte egress from lymph nodes, this compound decreases inflammatory responses in MS patients, leading to reduced lesion formation and brain volume loss .
Antibacterial Activity
Recent studies have highlighted this compound's antibacterial properties, particularly against resistant bacterial strains. A notable study demonstrated that this compound enhances the antibacterial effect of doripenem against carbapenem-resistant Enterobacteriaceae (CREC). The combination treatment significantly reduced the expression of genes associated with antibiotic resistance and bacterial motility .
Key Findings from Antibacterial Studies
- Gene Expression : this compound treatment led to decreased expression of carbapenemase-related genes (bla KPC) and efflux pump-related genes (acrB, acrD), suggesting a mechanism for its antibacterial action .
- Motility Inhibition : this compound inhibited bacterial motility by downregulating motility-related genes (flhD, motA), thereby affecting the bacteria's ability to move and colonize .
Table 1: Summary of Biological Activities of this compound
Case Study 1: Long-term Effects in Multiple Sclerosis
A post-marketing study evaluated the long-term safety and efficacy of this compound in MS patients. The results showed significant reductions in annualized relapse rates and brain volume loss over a 24-month period compared to placebo . The study included diverse patient subgroups based on prior treatment history, confirming this compound's consistent efficacy across different demographics.
Case Study 2: Antibacterial Efficacy
In a laboratory setting, this compound was tested against various bacterial strains, including Staphylococcus aureus and Pseudomonas aeruginosa. The Minimum Inhibitory Concentration (MIC) for this compound was found to be around 15 μM, with certain derivatives showing improved activity against biofilms formed by these pathogens . This highlights the potential for this compound derivatives as novel antibacterial agents.
Efficacy Against Biofilms
Research indicates that this compound not only inhibits planktonic bacterial growth but also affects preformed biofilms. Studies showed that certain derivatives of this compound had lower MICs than the parent compound, suggesting structural modifications can enhance its antibacterial properties without increasing cytotoxicity to human cells .
Table 2: Antibacterial Activity Data
| Compound | Target Bacteria | MIC (μM) | Effect on Biofilm |
|---|---|---|---|
| This compound | S. aureus | 15 | Moderate inhibition |
| Derivative 43 | S. aureus | 10 | Significant inhibition |
| Derivative 55 | Pseudomonas aeruginosa | 5 | Strong inhibition |
Q & A
Basic Research Questions
Q. What experimental models are commonly used to investigate Fingolimod’s mechanism of action in multiple sclerosis (MS)?
this compound’s mechanism is studied using in vitro lymphocyte migration assays and in vivo models like experimental autoimmune encephalomyelitis (EAE). These models assess immune modulation via sphingosine-1-phosphate receptor (S1PR) internalization, which traps lymphocytes in lymph nodes . CNS effects are evaluated using brain tissue analysis to measure direct S1PR modulation on astrocytes and oligodendrocytes .
Q. How are clinical trial endpoints (e.g., relapse rate, MRI activity) standardized in this compound studies?
Phase III trials (e.g., FREEDOMS, TRANSFORMS) use annualized relapse rate (ARR), gadolinium-enhancing MRI lesions, and disability progression (EDSS) as primary endpoints. Standardized protocols include double-blinded designs, placebo or active comparators (e.g., interferon-β), and 24-month follow-ups to assess sustained efficacy .
Q. What methodologies are employed to validate this compound’s pharmacokinetics and hepatic metabolism?
Pharmacokinetic studies use liquid chromatography-mass spectrometry (LC-MS) to measure plasma concentrations, with a focus on CYP4F2-mediated metabolism. Severe hepatic impairment cohorts are monitored for altered exposure using terminal half-life calculations (6–9 days) and dose adjustments .
Advanced Research Questions
Q. How can researchers resolve contradictory findings on this compound’s infection risk in meta-analyses?
Systematic reviews (e.g., Zhao et al., 2021) address heterogeneity by stratifying trials based on patient demographics, prior DMT use, and infection definitions. Sensitivity analyses exclude studies with high bias risk, while subgroup analyses differentiate between herpesviruses (e.g., VZV) and bacterial infections. Pooled odds ratios are adjusted for confounders like lymphocyte count thresholds .
Q. What advanced bioinformatics tools identify this compound’s oncological targets, such as PLK1 in head and neck squamous cell carcinoma (HNSC)?
TCGA data mining combined with Bayesian differential expression analysis (limma R package) identifies PLK1 as a key target. Validation includes in vitro lentiviral PLK1 overexpression in SCC9 cells, CCK-8 proliferation assays, and survival analysis via Kaplan-Meier curves. GSEA confirms enrichment in cell cycle pathways (NES=1.53, P<0.05) .
Q. What methodologies mitigate bias in real-world this compound studies comparing clinical trial vs. practice populations?
Real-world studies (e.g., PANGAEA) use propensity score matching to balance cohorts for disease severity and prior DMT exposure. Sensitivity analyses restrict populations to EU label criteria (highly active MS). Missing data are handled via multiple imputation or observed-case frameworks .
Q. How is this compound’s neuroprotective potential evaluated in Alzheimer’s disease models?
The 3xTg-AD mouse model undergoes Novel Object Location (NOL) tests to assess spatial memory. This compound’s efficacy is quantified by discrimination index improvements (e.g., from 0.2 to 0.5 at 12 months) and Western blot validation of amyloid-β reduction. CNS penetration is confirmed via LC-MS of brain tissue .
Q. What strategies address rebound syndrome after this compound discontinuation in MS patients?
Rebound risk is managed by overlapping this compound cessation with B-cell depleting therapies (e.g., rituximab) or corticosteroids. MRI monitoring at 4-week intervals tracks gadolinium lesion surges (median >30 new lesions). Washout periods are minimized (<2 weeks) to reduce relapse severity .
Q. Methodological Considerations
Q. How are Bayesian statistical frameworks applied in TCGA-based studies of this compound’s anticancer effects?
Bayesian analysis with the limma package identifies differentially expressed genes (|log2FC|≥1, P<0.05). Heatmaps and volcano plots visualize tumor vs. paracancerous tissue differences. Survival significance is validated via log-rank tests (HR=1.34 for PLK1) .
Q. What experimental designs validate this compound’s biomarker-driven efficacy, such as serum Sema4A levels?
Retrospective cohorts stratify MS patients by baseline Sema4A (ELISA-measured). Efficacy endpoints (ARR, EDSS) are compared between high/low Sema4A subgroups. Preclinical validation uses EAE mice injected with recombinant Sema4A-Fc, with this compound response assessed via clinical score reductions .
Properties
IUPAC Name |
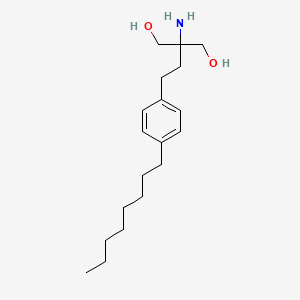
2-amino-2-[2-(4-octylphenyl)ethyl]propane-1,3-diol | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C19H33NO2/c1-2-3-4-5-6-7-8-17-9-11-18(12-10-17)13-14-19(20,15-21)16-22/h9-12,21-22H,2-8,13-16,20H2,1H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
KKGQTZUTZRNORY-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCCCCCCC1=CC=C(C=C1)CCC(CO)(CO)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C19H33NO2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID40167363 | |
| Record name | Fingolimod | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40167363 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
307.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Soluble | |
| Record name | Fingolimod | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Sphingosine‐1‐phosphate (S1P) is an important phospholipid that binds to various G‐protein‐coupled receptor subtypes, which can be identified as S1P1–5R. S1P and the receptors it binds to perform regular functions in the immune, cardiovascular, pulmonary, and nervous systems. S1P can be expressed ubiquitously, playing an important role in regulating inflammation. S1P1R, S1P2R, and S1P3R receptors can be found in the cardiovascular, immune, and central nervous systems. S1P4R is found on lymphocytic and hematopoietic cells, while S1P5R expression is found only on the spleen (on natural killer cells) or in the central nervous system. The active form of the drug, fingolimod phosphate, is a sphingosine 1-phosphate receptor modulator that exerts its mechanism of action in MS by binding to various sphingosine 1-phosphate receptors (1, 3, 4, and 5). It suppresses the exit of lymphocytes from lymph nodes, leading to a lower level of lymphocytes circulating in the peripheral circulation. This reduces the inflammation that is associated with MS. The mechanism of action of fingolimod is not fully understood but may be related to reduced lymphocyte circulation into the central nervous system. Immune modulating treatment such as fingolimod is not typically employed for SARS-CoV-2 pneumonia. Despite this, with the tissue findings of pulmonary edema and hyaline membrane formation, the timely use of immune modulators such as fingolimod can be considered to prevent acute respiratory distress syndrome (ARDS) associated with COVID-19. | |
| Record name | Fingolimod | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
162359-55-9 | |
| Record name | Fingolimod | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=162359-55-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Fingolimod [INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0162359559 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Fingolimod | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Fingolimod | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID40167363 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | FINGOLIMOD | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/3QN8BYN5QF | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Melting Point |
102-107 | |
| Record name | Fingolimod | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08868 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Synthesis routes and methods I
Procedure details







Synthesis routes and methods II
Procedure details





Synthesis routes and methods III
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














