Itraconazole
Overview
Description
Itraconazole is a triazole antifungal agent used to treat a variety of fungal infections, including aspergillosis, blastomycosis, coccidioidomycosis, histoplasmosis, and paracoccidioidomycosis . It was first synthesized in the early 1980s and approved for medical use in the United States in 1992 . This compound is known for its broad-spectrum activity against fungal pathogens and is available in oral and intravenous formulations .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of itraconazole involves multiple steps, starting with the preparation of key intermediates. One common method includes the reaction of 1-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-yl)ethanone with 1,3-dioxolane to form a dioxolane intermediate. This intermediate is then reacted with piperazine to yield this compound .
Industrial Production Methods: In industrial settings, this compound is prepared by dissolving a mixture of this compound and L-ascorbic acid in a mixed solvent of dichloromethane and methanol. Hydroxypropyl methylcellulose and pluronic F-127 are added, followed by dichloromethane to obtain a raw material solution. This solution is then subjected to supercritical fluid crystallization using carbon dioxide, resulting in composite particles with decreased particle size and increased bioavailability .
Chemical Reactions Analysis
Types of Reactions: Itraconazole undergoes several types of chemical reactions, including oxidation, reduction, and substitution.
Common Reagents and Conditions:
Reduction: Specific reduction reactions of this compound are less common in the literature.
Substitution: Substitution reactions involving this compound typically occur during its synthesis rather than in its metabolic pathways.
Major Products Formed: The major metabolites of this compound include hydroxy-itraconazole, keto-itraconazole, and N-desalkyl-itraconazole .
Scientific Research Applications
Systemic Fungal Infections
Itraconazole is primarily indicated for the treatment of several serious fungal infections, including:
- Blastomycosis : Effective in both pulmonary and extrapulmonary forms.
- Histoplasmosis : Particularly useful for chronic cavitary pulmonary disease and disseminated forms.
- Aspergillosis : Recommended for patients intolerant to or refractory to amphotericin B therapy .
Superficial Fungal Infections
This compound is also used to treat superficial fungal infections, notably:
- Onychomycosis : The treatment has a cure rate of approximately 63% .
- Seborrheic Dermatitis : Although not FDA-approved for this indication, this compound has shown efficacy when administered in pulse therapy .
Prophylactic Use
This compound is utilized as a prophylactic agent in immunocompromised patients, including those with:
- HIV/AIDS
- Patients undergoing chemotherapy
- Individuals who have received organ transplants
The broad-spectrum coverage and safety profile make this compound a preferred choice for preventing systemic fungal infections in these populations .
Oncological Applications
Recent research has highlighted this compound's potential in cancer treatment:
- Inhibition of Tumor Growth : this compound has been shown to inhibit the Hedgehog signaling pathway, which is involved in cancer cell proliferation. Studies indicate that this compound can induce autophagy-mediated cell death in colon cancer cells, leading to significant tumor growth suppression in animal models .
- Enhanced Chemosensitivity : A clinical retrospective study found that combining this compound with platinum-based chemotherapy improved progression-free survival and overall survival rates in ovarian cancer patients compared to chemotherapy alone .
Formulation and Bioavailability
The pharmacological advancements surrounding this compound have led to the development of various formulations:
- Oral Solutions and Intravenous Formulations : These formulations enhance solubility and bioavailability, making this compound accessible for patients with systemic fungal infections who may have variable absorption from capsule formulations .
Efficacy in Immunocompromised Patients
A study involving patients with HIV demonstrated that those receiving this compound prophylaxis had significantly lower incidences of systemic fungal infections compared to those who did not receive prophylaxis.
Cancer Treatment Trials
Clinical trials exploring the use of this compound in combination with standard chemotherapy regimens have shown promising results, particularly in improving patient outcomes in ovarian cancer.
Summary Table of Applications
| Application Area | Specific Uses | FDA Approval Status |
|---|---|---|
| Systemic Fungal Infections | Blastomycosis, Histoplasmosis, Aspergillosis | Yes |
| Superficial Fungal Infections | Onychomycosis, Seborrheic Dermatitis | Partial (Onychomycosis only) |
| Prophylaxis | HIV/AIDS, Chemotherapy, Organ Transplants | Yes |
| Oncology | Tumor growth inhibition | Research Phase |
Mechanism of Action
Itraconazole exerts its antifungal effects by inhibiting the enzyme lanosterol 14α-demethylase, a cytochrome P450 enzyme involved in the conversion of lanosterol to ergosterol. Ergosterol is a crucial component of fungal cell membranes. By inhibiting this enzyme, this compound disrupts the synthesis of ergosterol, leading to increased membrane permeability and ultimately fungal cell death .
Comparison with Similar Compounds
Itraconazole is often compared with other triazole antifungal agents such as fluconazole, voriconazole, and posaconazole:
Fluconazole: this compound has a broader spectrum of activity, particularly against Aspergillus species, which fluconazole does not cover.
Voriconazole: Voriconazole has a broader spectrum of activity than this compound but is associated with more severe side effects.
Posaconazole: Posaconazole is similar to this compound in terms of spectrum but has better bioavailability and fewer drug interactions.
This compound’s unique properties, such as its broad-spectrum activity and potential use in cancer treatment, make it a valuable compound in both clinical and research settings.
Q & A
Basic Research Questions
Q. What chromatographic methods are validated for simultaneous quantification of itraconazole with other antifungals, and how are critical parameters optimized?
- Methodological Answer : Reverse-phase high-performance liquid chromatography (RP-HPLC) with a Zorbax Eclipse C18 column is recommended. Optimize parameters (e.g., mobile phase pH, flow rate) using a 2³ factorial design to assess resolution, tailing, and theoretical plates. Validation includes robustness testing via fractional factorial design . For example, Roshdy et al. (2021) resolved this compound alongside fluconazole and terbinafine by analyzing interactions between independent variables (e.g., acetonitrile percentage) and responses (e.g., resolution R1–R4) .
Q. How should researchers design in vitro release studies for this compound-loaded nanoparticles to ensure reproducibility?
- Methodological Answer : Use phosphate-buffered saline (pH 6.8) at 37°C with sink conditions. Employ a minimum of five replicates (n=5) and report mean ± SEM. For example, in vitro release profiles of this compound from PLGA nanoparticles (F4, F7, F8) showed sustained release over 24 hours, with formulation variables (e.g., PLGA concentration) impacting release kinetics .
Q. What are the standard protocols for assessing this compound’s antifungal activity in preclinical models?
- Methodological Answer : Use broth microdilution assays per CLSI guidelines (e.g., MIC determination against Aspergillus or Candida). Include controls for pH-dependent solubility and serum protein binding, as this compound’s efficacy is influenced by physiological conditions. Liu et al. (2011) demonstrated improved antifungal activity of pyridine-substituted analogues under standardized testing .
Advanced Research Questions
Q. How can response surface methodology (RSM) optimize this compound multiparticulate systems for enhanced dissolution and reduced adhesion?
- Methodological Answer : Apply central composite design (CCD) to variables like drug-carrier mass ratio and core weight gain. Use response variables (e.g., accumulative dissolution rate, adhesion rate) to generate polynomial equations. For example, Sadalge et al. (2024) optimized this compound pellets using CCD, validated by SEM and XRD to confirm amorphous dispersion .
Q. What experimental design principles are critical for formulating this compound-loaded PLGA nanoparticles with high encapsulation efficiency?
- Methodological Answer : Employ a 2³ factorial design to evaluate PLGA concentration (X1), benzyl benzoate (X2), and drug loading (X3). Use ANOVA to identify significant factors (e.g., PLGA% impacts particle size, F-test p < 0.05) and derive regression models for response optimization (e.g., ITRAe = 250.5 + 25.3X1 – 18.7X2) .
Q. How can pharmacokinetic-pharmacodynamic (PK-PD) modeling resolve contradictions in this compound’s concentration-dependent antitumor effects?
- Methodological Answer : Perform noncompartmental PK analysis to quantify plasma/tumor this compound levels. Correlate with pharmacodynamic endpoints (e.g., tumor perfusion via MRI, cytokine levels) using Spearman rank correlation. Gerber et al. (2020) linked higher tumor this compound concentrations to reduced Ktrans (perfusion) and IL1b/GM-CSF levels (ρ = -0.71, p < 0.05) .
Q. What statistical strategies address robustness in HPLC method validation for this compound impurity profiling?
- Methodological Answer : Use fractional factorial design (e.g., 2⁴⁻¹) to test factors like column temperature and mobile phase composition. Assess system suitability parameters (e.g., tailing factor, theoretical plates). Kasagić et al. (2013) validated a method for this compound impurities B/F, confirming robustness via controlled variability in retention times (<2% RSD) .
Q. How should window-of-opportunity trials be designed to evaluate this compound’s antivascular effects in non-small cell lung cancer (NSCLC)?
- Methodological Answer : Administer this compound (300 mg BID) pre-surgically for 10–14 days. Use dynamic contrast-enhanced MRI for tumor perfusion (Ktrans) and collect paired biopsies for biomarker analysis (e.g., microvessel density). Gerber et al. (2020) demonstrated dose-dependent reductions in tumor volume and perfusion, validated by metabolic profiling .
Q. Key Methodological Considerations
- Experimental Design : Prioritize factorial designs (e.g., 2³, CCD) over one-factor-at-a-time approaches to capture interactions .
- Statistical Analysis : Use ANOVA for factorial experiments and Spearman correlations for nonparametric PK-PD relationships .
- Characterization Techniques : SEM, XRD, and DSC are critical for confirming drug-polymer interactions in formulations .
Properties
CAS No. |
84625-61-6 |
|---|---|
Molecular Formula |
C35H38Cl2N8O4 |
Molecular Weight |
705.6 g/mol |
IUPAC Name |
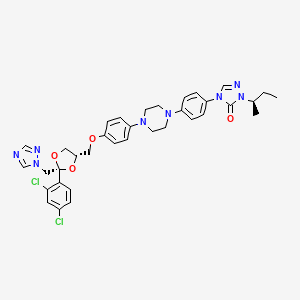
2-butan-2-yl-4-[4-[4-[4-[[(2R)-2-(2,4-dichlorophenyl)-2-(1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-1,2,4-triazol-3-one |
InChI |
InChI=1S/C35H38Cl2N8O4/c1-3-25(2)45-34(46)44(24-40-45)29-7-5-27(6-8-29)41-14-16-42(17-15-41)28-9-11-30(12-10-28)47-19-31-20-48-35(49-31,21-43-23-38-22-39-43)32-13-4-26(36)18-33(32)37/h4-13,18,22-25,31H,3,14-17,19-21H2,1-2H3/t25?,31?,35-/m0/s1 |
InChI Key |
VHVPQPYKVGDNFY-WSTHBRJPSA-N |
impurities |
Impurities: 4-[4-[4-(4-methoxyphenyl)piperazin-1-yl]phenyl]-2-[(1RS)-1-methylpropyl]-2,4-dihydro-3H-1,2,4-triazol-3-one; 4-[4-[4-[4-[[cis-2-(2,4-dichlorophenyl)-2-(4H-1,2,4-triazol-4-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-[(1RS)-1-methylpropyl]-2,4-dihydro-3H-1,2,4-triazol-3-one; 4-[4-[4-[4-[[cis-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-propyl-2,4-dihydro-3H-1,2,4-triazol-3-one; 4-[4-[4-[4-[[cis-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-(1-methylethyl)-2,4-dihydro-3H-1,2,4-triazol-3-one; 4-[4-[4-[4-[[trans-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-[(1RS)-1-methylpropyl]-2,4-dihydro-3H-1,2,4-triazol-3-one; 2-butyl-4-[4-[4-[4-[[cis-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2,4-dihydro-3H-1,2,4-triazol-3-one; 4-[4-[4-[4-[[cis-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy]phenyl]piperazin-1-yl]phenyl]-2-[[cis-2-(2,4-dichlorophenyl)-2-(1H-1,2,4-triazol-1-ylmethyl)-1,3-dioxolan-4-yl]methyl]-2,4-dihydro-3H-1,2,4-triazol-3-one. |
SMILES |
CCC(C)N1C(=O)N(C=N1)C2=CC=C(C=C2)N3CCN(CC3)C4=CC=C(C=C4)OCC5COC(O5)(CN6C=NC=N6)C7=C(C=C(C=C7)Cl)Cl |
Isomeric SMILES |
CCC(C)N1C(=O)N(C=N1)C2=CC=C(C=C2)N3CCN(CC3)C4=CC=C(C=C4)OCC5CO[C@](O5)(CN6C=NC=N6)C7=C(C=C(C=C7)Cl)Cl |
Canonical SMILES |
CCC(C)N1C(=O)N(C=N1)C2=CC=C(C=C2)N3CCN(CC3)C4=CC=C(C=C4)OCC5COC(O5)(CN6C=NC=N6)C7=C(C=C(C=C7)Cl)Cl |
Appearance |
Solid powder |
Color/Form |
Solid Crystals from toluene |
melting_point |
168-170 166.2 °C |
Key on ui other cas no. |
84625-61-6 84604-65-9 |
physical_description |
Solid |
Pictograms |
Irritant |
Purity |
>98% (or refer to the Certificate of Analysis) |
shelf_life |
>2 years if stored properly |
solubility |
Practically insoluble in water and dilute acidic solutions |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
Itraconazole, R51211, Orungal, Oriconazole, Sporanox, Itraconazolum, Itraconazol, Itrizole |
vapor_pressure |
2.6X10-20 mm Hg at 25 °C (est) |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















