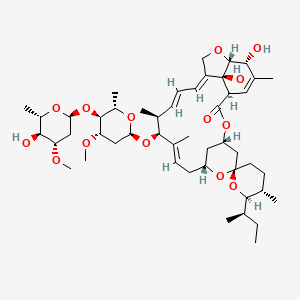
Ivermectin B1a
Overview
Description
Ivermectin B1a is a broad-spectrum antiparasitic agent belonging to the avermectin family of medications. It was discovered in 1975 and initially used in veterinary medicine to prevent and treat heartworm and acariasis. Approved for human use in 1987, ivermectin is used to treat infestations such as head lice, scabies, river blindness (onchocerciasis), strongyloidiasis, trichuriasis, ascariasis, and lymphatic filariasis .
Preparation Methods
Ivermectin B1a is synthesized from the fermentation products of the bacterium Streptomyces avermitilis. The preparation involves several steps:
Protection of Hydroxyl Groups: The 4’'-position hydroxyl and 5
Biological Activity
Ivermectin B1a, a derivative of avermectin, is a broad-spectrum antiparasitic agent recognized for its multifaceted biological activities. This article delves into its biological mechanisms, therapeutic applications, and the latest research findings, supported by data tables and case studies.
Overview of this compound
Ivermectin is primarily composed of two homologs: 22,23-dihydroavermectin B1a (HB1a) and 22,23-dihydroavermectin B1b (HB1b). The compound is known for its efficacy against various parasites, including nematodes and arthropods, and has recently gained attention for its antiviral properties against several viruses, including SARS-CoV-2.
This compound exhibits its biological activity through several mechanisms:
- Antiparasitic Activity : The primary target of ivermectin is the glutamate-gated chloride channels in invertebrates. This interaction leads to paralysis and death of the parasites .
- Antiviral Activity : Ivermectin has been shown to inhibit the nuclear import of viral proteins, which is crucial for the replication of viruses such as HIV-1 and Zika virus. It disrupts the interaction between importin proteins and viral nuclear localization signals .
- Immunomodulation : The compound also has immunomodulatory effects, influencing host immune responses .
Antiviral Efficacy Against SARS-CoV-2
Recent studies have highlighted ivermectin's potential against SARS-CoV-2. A comparative analysis showed that while HB1b has a higher affinity for viral structures, HB1a interacts more effectively with host structures. This differential behavior suggests that both homologs could be strategically utilized in antiviral therapies .
Table 1: Comparative Binding Affinity of Ivermectin Homologs
| Homolog | Target Structure | Binding Affinity |
|---|---|---|
| HB1a | Host proteins | High |
| HB1b | Viral proteins | Higher |
Case Studies
A notable case reported the use of ivermectin in a patient with disseminated strongyloidiasis. The patient received a combination of oral and subcutaneous ivermectin due to severe gastrointestinal complications. The treatment resulted in significant clinical improvement, showcasing ivermectin's effectiveness even in critical conditions .
Clinical Applications
This compound is widely used in treating:
- Onchocerciasis : Often referred to as river blindness.
- Lymphatic Filariasis : A parasitic disease caused by filarial worms.
- Scabies : A skin infestation caused by mites.
Scientific Research Applications
Antiparasitic Applications
Ivermectin B1a is primarily recognized for its efficacy against various parasitic infections. It is the treatment of choice for onchocerciasis (river blindness) and is also effective against other nematodes such as strongyloidiasis, ascariasis, and trichuriasis. The compound works by enhancing the permeability of cell membranes in parasites, leading to paralysis and death.
Table 1: Antiparasitic Efficacy of this compound
| Parasite | Infection | Efficacy |
|---|---|---|
| Onchocerca volvulus | Onchocerciasis | Highly effective |
| Strongyloides stercoralis | Strongyloidiasis | Effective |
| Ascaris lumbricoides | Ascariasis | Effective |
| Trichuris trichiura | Trichuriasis | Effective |
Antiviral Activity
Recent studies have indicated that this compound exhibits antiviral properties against several viruses, including SARS-CoV-2, the virus responsible for COVID-19. Research suggests that it may inhibit viral replication and modulate inflammatory responses.
Case Study: Ivermectin in COVID-19 Treatment
A systematic review of randomized controlled trials revealed that Ivermectin significantly reduced mortality rates and improved clinical outcomes in patients with COVID-19. One notable trial involved administering Ivermectin to family members of infected individuals, resulting in a statistically significant decrease in symptomatic cases (7.4% vs. 58.4%, P < 0.001) among treated participants .
Anticancer Potential
This compound has shown promising anticancer activity, particularly in colorectal cancer models. It has been observed to inhibit cell proliferation and induce apoptosis in cancer cells by promoting tubulin polymerization.
Table 2: Anticancer Effects of this compound
| Cancer Cell Line | IC50 Value (µM) | Mechanism of Action |
|---|---|---|
| HCT-116 | 30 | Promotes tubulin polymerization |
| SW480 | Not specified | Induces apoptosis via Caspase activation |
A study demonstrated that this compound significantly inhibited the growth of HCT-116 cells with an IC50 value of 30 µM, suggesting its potential as a microtubule-targeting agent for cancer therapy .
Q & A
Basic Research Questions
Q. How can researchers reliably identify and quantify Ivermectin B1a in mixed samples (e.g., commercial ivermectin formulations)?
Methodological Answer:
- Use reversed-phase HPLC with UV detection (e.g., C18 column, mobile phase: acetonitrile/water gradient). Quantify based on retention times and peak areas calibrated against certified reference standards (e.g., 22,23-dihydroavermectin B1a vs. B1b) .
- Validate the method using parameters such as limit of detection (LOD: ~0.1 µg/mL), limit of quantification (LOQ: ~0.5 µg/mL), and recovery rates (≥95%) .
Q. What is the primary mechanism of action of this compound in parasitic infections?
Methodological Answer:
- Conduct electrophysiological assays on nematode neurons to measure glutamate-gated chloride channel (GluCl) currents. Compare dose-response curves between this compound and its analogs (e.g., abamectin B1a) .
- Use gene knockout models (e.g., C. elegans GluCl mutants) to confirm target specificity .
Q. What analytical methods are suitable for distinguishing this compound from structurally similar analogs (e.g., avermectin B1a)?
Methodological Answer:
- Employ high-resolution mass spectrometry (HRMS) to differentiate based on molecular weight (B1a: 875.09 Da vs. avermectin B1a: 873.1 Da) .
- Use NMR spectroscopy (e.g., -NMR) to resolve structural differences at the C25 position (sec-butyl vs. iso-propyl substituents) .
Advanced Research Questions
Q. How should researchers design experiments to evaluate this compound’s efficacy in vivo versus in vitro models?
Methodological Answer:
- In vitro: Use cell-based assays (e.g., HEK293 cells expressing GluCl) to measure EC values. Include controls for nonspecific cytotoxicity (e.g., LDH release assays) .
- In vivo: Optimize dosage using pharmacokinetic modeling (e.g., compartmental analysis) to account for metabolic differences between species. For murine models, typical doses range from 0.2–2 mg/kg .
Q. How can contradictory data on this compound’s antiviral activity (e.g., against SARS-CoV-2) be resolved?
Methodological Answer:
- Conduct dose-response meta-analysis of existing studies, stratifying by experimental design (e.g., cell type, viral load, treatment duration). Use sensitivity analysis to identify confounding variables (e.g., purity of B1a vs. B1b mixtures) .
- Replicate key studies under standardized conditions, adhering to Good Laboratory Practice (GLP) guidelines for assay validation .
Q. What methodologies are appropriate for studying synergistic effects between this compound and other antiparasitics?
Methodological Answer:
- Use isobologram analysis to quantify synergy (e.g., with albendazole or praziquantel). Calculate combination indices (CI <1 indicates synergy) .
- Validate findings in co-infection models (e.g., Schistosoma mansoni and Plasmodium berghei in mice) to assess clinical relevance .
Q. How can degradation products of this compound be characterized under varying storage conditions?
Methodological Answer:
- Perform forced degradation studies (e.g., exposure to heat, light, or acidic/alkaline conditions). Identify degradation products via LC-MS/MS and compare toxicity profiles using in silico tools (e.g., Toxtree) .
- Establish stability-indicating methods with specificity for degradation markers (e.g., 8,9-Z isomer) .
Q. What pharmacokinetic modeling approaches are recommended for optimizing this compound dosing in clinical trials?
Methodological Answer:
- Use non-linear mixed-effects modeling (NONMEM) to analyze population PK data. Incorporate covariates such as body weight, hepatic function, and co-administered drugs .
- Validate models using bootstrap resampling and visual predictive checks (VPCs) .
Q. Methodological Considerations
- Reproducibility: Ensure raw data (e.g., chromatograms, electrophysiological traces) are archived in open-access repositories with metadata compliant with FAIR principles .
- Ethical Compliance: For human studies, adhere to protocols outlined in (e.g., informed consent, IRB approval) and ICH guidelines for clinical trial design .
Properties
Key on ui mechanism of action |
Ivermectin binds selectively and with high affinity to glutamate-gated chloride ion channels in invertebrate muscle and nerve cells of the microfilaria. This binding causes an increase in the permeability of the cell membrane to chloride ions and results in hyperpolarization of the cell, leading to paralysis and death of the parasite. Ivermectin also is believed to act as an agonist of the neurotransmitter gamma-aminobutyric acid (GABA), thereby disrupting GABA-mediated central nervous system (CNS) neurosynaptic transmission. Ivermectin may also impair normal intrauterine development of _O. volvulus_ microfilariae and may inhibit their release from the uteri of gravid female worms. |
|---|---|
CAS No. |
70288-86-7 |
Molecular Formula |
C48H74O14 |
Molecular Weight |
875.1 g/mol |
IUPAC Name |
(1S,4S,5'S,6R,6'R,8R,10E,12S,13S,14E,16E,20R,21R,24S)-6'-[(2S)-butan-2-yl]-21,24-dihydroxy-12-[(2R,4S,5S,6S)-5-[(2S,4S,5S,6S)-5-hydroxy-4-methoxy-6-methyloxan-2-yl]oxy-4-methoxy-6-methyloxan-2-yl]oxy-5',11,13,22-tetramethylspiro[3,7,19-trioxatetracyclo[15.6.1.14,8.020,24]pentacosa-10,14,16,22-tetraene-6,2'-oxane]-2-one |
InChI |
InChI=1S/C48H74O14/c1-11-25(2)43-28(5)17-18-47(62-43)23-34-20-33(61-47)16-15-27(4)42(26(3)13-12-14-32-24-55-45-40(49)29(6)19-35(46(51)58-34)48(32,45)52)59-39-22-37(54-10)44(31(8)57-39)60-38-21-36(53-9)41(50)30(7)56-38/h12-15,19,25-26,28,30-31,33-45,49-50,52H,11,16-18,20-24H2,1-10H3/b13-12+,27-15+,32-14+/t25-,26-,28-,30-,31-,33+,34-,35+,36-,37-,38-,39-,40+,41-,42-,43+,44-,45+,47+,48+/m0/s1 |
InChI Key |
AZSNMRSAGSSBNP-TYECJXEWSA-N |
SMILES |
CCC(C)C1C(CCC2(O1)CC3CC(O2)CC=C(C(C(C=CC=C4COC5C4(C(C=C(C5O)C)C(=O)O3)O)C)OC6CC(C(C(O6)C)OC7CC(C(C(O7)C)O)OC)OC)C)C |
Isomeric SMILES |
CC[C@H](C)[C@@H]1[C@H](CC[C@@]2(O1)C[C@@H]3C[C@H](O2)C/C=C(/[C@H]([C@H](/C=C/C=C/4\CO[C@H]5[C@@]4([C@H](C=C([C@H]5O)C)C(=O)O3)O)C)O[C@H]6C[C@@H]([C@H]([C@@H](O6)C)O[C@H]7C[C@@H]([C@H]([C@@H](O7)C)O)OC)OC)\C)C |
Canonical SMILES |
CCC(C)C1C(CCC2(O1)CC3CC(O2)CC=C(C(C(C=CC=C4COC5C4(C(C=C(C5O)C)C(=O)O3)O)C)OC6CC(C(C(O6)C)OC7CC(C(C(O7)C)O)OC)OC)C)C |
Appearance |
Solid powder |
Key on ui other cas no. |
71827-03-7 70288-86-7 |
Pictograms |
Acute Toxic; Health Hazard; Environmental Hazard |
Purity |
>90% (or refer to the Certificate of Analysis) |
shelf_life |
>2 years if stored properly |
solubility |
Insoluble |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
MK-933; Ivermectin; Ivomec; L 64047; 1 Mectizan; MK 933; MK-0933; Noromectin; Pandex. |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















