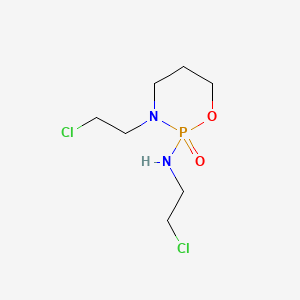
Ifosfamide
Overview
Description
Ifosfamide is a chemotherapy medication used to treat various types of cancer, including testicular cancer, soft tissue sarcoma, osteosarcoma, bladder cancer, small cell lung cancer, cervical cancer, and ovarian cancer . It belongs to the alkylating agent and nitrogen mustard family of medications . This compound works by disrupting the duplication of DNA and the creation of RNA, thereby inhibiting cancer cell growth .
Preparation Methods
Ifosfamide can be synthesized through several routes. One method involves reacting an this compound intermediate with a chlorinating agent, followed by cyclization under the action of an organic base . Another method uses aziridine as the starting material, which undergoes cyclization and subsequent reactions to form this compound . Industrial production methods often involve optimizing these synthetic routes to ensure high yield and purity while minimizing the use of toxic and explosive chemicals .
Chemical Reactions Analysis
Ifosfamide undergoes various chemical reactions, including oxidation, reduction, and substitution. It is metabolized in the liver by cytochrome P450 enzymes, leading to the formation of active and inactive metabolites . Common reagents used in these reactions include mixed-function oxidases and organic bases . Major products formed from these reactions include chloroacetaldehyde and other metabolites that contribute to the drug’s therapeutic and toxic effects .
Scientific Research Applications
FDA-Approved Indications
Ifosfamide is utilized in several FDA-approved settings:
- Germ Cell Tumors : Administered at a dose of 1.2 g/m²/day for 5 days in combination with mesna, it has shown complete remission rates of 21% to 26% when paired with cisplatin or etoposide .
- Soft Tissue Sarcomas : Combination therapy with this compound and mesna yields objective response rates around 40% as induction therapy .
- Lymphomas : It serves as salvage therapy in non-Hodgkin lymphoma and is part of the RICE regimen for relapsed Hodgkin lymphoma .
- Ovarian Cancer : Particularly in platinum-resistant cases, this compound has demonstrated response rates exceeding 40% when combined with other agents .
Off-Label Uses
This compound's versatility extends to several off-label applications:
- Ewing Sarcoma : High-dose this compound has been shown to extend survival compared to other regimens like topotecan .
- Cervical Cancer : Administered alongside mesna at 1500 mg/m²/day, response rates range from 18% to 45% when combined with paclitaxel and cisplatin .
- Bladder Carcinoma : Utilized for advanced bladder cancer cases .
- Lung Cancer : More beneficial than standard regimens as maintenance or consolidation therapy for both small-cell and non-small-cell lung cancer .
Delayed Encephalopathy
A notable case involved a 19-year-old female patient with Hodgkin lymphoma who developed neurotoxicity 16 days post-ifosfamide infusion. She recovered rapidly after treatment with methylene blue, highlighting the importance of recognizing delayed effects of this compound .
Acute Kidney Injury
Another case study documented a 44-year-old woman with leiomyosarcoma who experienced acute kidney injury following this compound treatment. This case emphasizes the need for monitoring renal function during therapy .
Comparative Efficacy Data
| Cancer Type | Treatment Regimen | Response Rate |
|---|---|---|
| Germ Cell Tumors | This compound + Mesna + Cisplatin | 21% - 26% |
| Soft Tissue Sarcomas | This compound + Mesna | ~40% |
| Ewing Sarcoma | High-dose this compound | Improved survival |
| Cervical Cancer | This compound + Mesna + Paclitaxel + Cisplatin | 18% - 45% |
| Bladder Carcinoma | This compound (advanced cases) | Variable |
Mechanism of Action
Ifosfamide requires biotransformation in the liver by the cytochrome P450 system before it becomes active . The active metabolites of this compound form DNA crosslinks at the guanine N-7 position, leading to cellular damage and death . This disruption of DNA replication and RNA production inhibits cancer cell growth and proliferation . The exact molecular targets and pathways involved in this compound’s cytotoxicity are still being studied, but it is known to interfere with DNA and RNA synthesis .
Comparison with Similar Compounds
Ifosfamide is chemically related to other nitrogen mustards, such as cyclophosphamide, trofosfamide, mafosfamide, sufosfamide, and glufosfamide . Compared to these compounds, this compound has unique pharmacokinetic properties, including a longer half-life and different metabolic pathways . Its ability to form DNA crosslinks and its effectiveness in treating a wide range of cancers make it a valuable chemotherapeutic agent .
Biological Activity
Ifosfamide is a nitrogen mustard alkylating agent used primarily in the treatment of various cancers, including sarcomas and lymphomas. As a prodrug, it requires metabolic activation to exert its therapeutic effects. This article explores the biological activity of this compound, focusing on its pharmacokinetics, mechanisms of action, gene expression alterations, and clinical efficacy based on diverse research findings.
This compound acts primarily through DNA alkylation , leading to the formation of cross-links that inhibit DNA replication and transcription. The active metabolite, isophosphoramide mustard, predominantly interacts with the N-7 position of guanine residues in DNA, causing both intra- and inter-strand cross-links. This mechanism ultimately leads to cell death, particularly in rapidly dividing cancer cells .
Metabolism and Pharmacokinetics
The metabolism of this compound involves several cytochrome P450 (CYP) enzymes, including CYP2B6, CYP2C8, CYP2C9, and CYP3A4. In vitro studies using human hepatocyte cultures have demonstrated that these enzymes catalyze the 4-hydroxylation of this compound, which is crucial for its activation . Notably, rifampin has been identified as a potent inducer of this compound metabolism, enhancing its pharmacokinetic profile in patients .
Gene Expression Changes Induced by this compound
Research has shown that this compound administration significantly alters gene expression in liver and kidney tissues. A study identified 2,672 differentially expressed genes (DEGs) in the liver after treatment with 100 mg/kg body weight per day. Of these, 1,283 genes were upregulated while 1,389 were downregulated. In the kidneys, 401 DEGs were observed with a similar trend of downregulation predominating . These changes indicate potential toxicity and organ-specific responses to this compound.
Table 1: Differential Gene Expression Induced by this compound
| Organ | Dose (mg/kg) | Upregulated Genes | Downregulated Genes | Total DEGs |
|---|---|---|---|---|
| Liver | 100 | 1283 | 1389 | 2672 |
| Kidney | 100 | 149 | 252 | 401 |
Clinical Efficacy: Case Studies and Trials
This compound has been evaluated in numerous clinical settings. A notable phase II trial assessed the efficacy of this compound combined with lenvatinib and etoposide in children with high-grade osteosarcoma. Results indicated improved outcomes compared to this compound alone . Similarly, another study reported a progression-free rate (PFR) of 66% at three months for patients treated with sorafenib plus this compound for advanced soft tissue sarcoma .
Case Study: Efficacy in Small Cell Lung Cancer (SCLC)
This compound monotherapy has shown a response rate of approximately 42% in SCLC patients. However, a recent phase II study indicated that its efficacy diminishes in heavily pre-treated populations, leading to an early termination due to futility . This underscores the importance of considering prior treatment history when evaluating therapeutic options.
Toxicity Profile
While effective, this compound is associated with various toxicities. Common adverse effects include myelosuppression, neurotoxicity (notably encephalopathy), and renal toxicity. The alteration of gene expression related to immune cell localization and organ development suggests that careful monitoring is required during treatment to mitigate these risks .
Q & A
Basic Research Questions
Q. What experimental techniques are critical for studying the alkylation kinetics of ifosfamide metabolites?
- Answer: ³¹P NMR spectroscopy is a key method for tracking the kinetics of this compound's active metabolites, such as isophosphoramide mustard, to quantify bisalkylation rates and compare them to cyclophosphamide analogs. This technique helps elucidate structural differences impacting DNA cross-linking efficiency .
Q. How does this compound-induced glutathione depletion influence therapeutic efficacy, and how is this monitored in clinical studies?
- Answer: Intracellular glutathione (GSH) levels in peripheral blood lymphocytes are measured via HPLC or enzymatic assays to assess this compound's oxidative stress effects. Depletion correlates with reduced detoxification capacity and may predict treatment resistance. Mesna co-administration is critical to mitigate bladder toxicity but does not fully restore systemic GSH .
Q. What are the standard protocols for mitigating nephrotoxicity in pediatric this compound regimens?
- Answer: Hydration (≥2 L/day) and mesna are mandatory. Tubular dysfunction (e.g., Fanconi syndrome) is monitored via urinary β2-microglobulin and serum bicarbonate. Dose adjustments are required for patients with pre-existing renal impairment or retroperitoneal tumors .
Advanced Research Questions
Q. How can preclinical models elucidate the synergy between this compound and immunomodulators like interferon?
- Answer: Murine Ewing sarcoma models demonstrate that interferon-α/β enhances this compound's DNA damage response by upregulating pro-apoptotic pathways. Tumor volume regression and survival metrics are compared between monotherapy and combination arms. RNA-seq analysis identifies IFN-regulated genes (e.g., STAT1) as biomarkers for synergy .
Q. What methodological considerations are critical for designing trials testing high-dose this compound (HDI) in refractory soft tissue sarcomas?
- Answer: HDI (4 g/m²/day ×3 days) requires granulocyte colony-stimulating factor (G-CSF) support due to grade 3–4 neutropenia. Response evaluation uses RECIST criteria, with emphasis on distinguishing SDI-resistant (progression on ≤4 cycles) vs. SDI-refractory (no response) subgroups. Pharmacokinetic monitoring of chloroacetaldehyde (CAA) levels informs renal toxicity risk .
Q. How do metabolic differences between this compound and cyclophosphamide explain their distinct toxicity profiles?
- Answer: this compound generates higher CAA, a neurotoxic and nephrotoxic metabolite, due to slower 4-hydroxylation. Comparative LC-MS/MS studies of hepatic CYP3A4/5 activity and urinary CAA excretion rates clarify interpatient variability. Glutathione S-transferase (GST) polymorphisms further modulate detoxification efficiency .
Q. What statistical approaches resolve contradictions in survival outcomes for this compound combinations across sarcoma subtypes?
- Answer: Meta-analyses (e.g., Cochrane Review) stratify by histology (leiomyosarcoma vs. synovial sarcoma) and prior therapy. For uterine carcinosarcoma, weighted log-rank tests show a 1.82 odds ratio favoring this compound-cisplatin over monotherapy, but toxicity-adjusted benefit-risk ratios must account for neurotoxicity (RR = 1.59) and febrile neutropenia .
Q. How can microencapsulation technologies improve this compound's therapeutic index in pancreatic cancer?
- Answer: Cell-in-a-Box® microencapsulation allows localized this compound activation via cytochrome P450 enzymes. Phase II trials use a 33% dose reduction (1.6 g/m² vs. 2.4 g/m²) with CT-based tumor response criteria. RNA-seq of tumor biopsies post-treatment identifies hypoxia-related genes (e.g., HIF1A) as predictors of encapsulation efficacy .
Q. Methodological Guidelines
Q. What pharmacokinetic parameters should be prioritized in phase I this compound trials?
- Answer: Area under the curve (AUC) for 4-hydroxy-ifosfamide and CAA, renal clearance rates, and half-life (t½ = 6–7 hours). Population PK models incorporate creatinine clearance and albumin levels to predict neurotoxicity risk (e.g., serum bicarbonate <20 mmol/L) .
Q. How are zebrafish models used to study this compound's environmental impact and genomic toxicity?
Properties
IUPAC Name |
N,3-bis(2-chloroethyl)-2-oxo-1,3,2λ5-oxazaphosphinan-2-amine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C7H15Cl2N2O2P/c8-2-4-10-14(12)11(6-3-9)5-1-7-13-14/h1-7H2,(H,10,12) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
HOMGKSMUEGBAAB-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1CN(P(=O)(OC1)NCCCl)CCCl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C7H15Cl2N2O2P | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID7020760 | |
| Record name | Ifosfamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7020760 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
261.08 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Ifosfamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015312 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Soluble in water, 1.50e+01 g/L | |
| Record name | Ifosfamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01181 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | IFOSFAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7023 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ifosfamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015312 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The exact mechanism of ifosfamide has not been determined, but appears to be similar to other alkylating agents. Ifosfamide requires biotransformation in the liver by mixed-function oxidases (cytochrome P450 system) before it becomes active. After metabolic activation, active metabolites of ifosfamide alkylate or bind with many intracellular molecular structures, including nucleic acids. The cytotoxic action is primarily through the alkylation of DNA, done by attaching the N-7 position of guanine to its reactive electrophilic groups. The formation of inter and intra strand cross-links in the DNA results in cell death., Mechanism of action: metabolites cause alkylation of DNA. /from table/, Ifosfamide, a structural analog of cyclophosphamide, belongs to the oxazaphosphorine class of antitumor alkylating agents which must be activated by the mixed function oxidase system of the liver. The 4-hydroxy oxazaphosphorines are a reactive species capable of interacting with nucleic acids & cellular materials to cause cell damage & death. The 4-hydroxy metabolite spontaneously liberates acrolein in many sites throughout the body & it is this substance that is responsible for oxazaphosphorine urotoxicity. Both ifosfamide & cyclophosphamide produce cystitis characterized by tissue edema & ulceration followed by sloughing of mucosal epithelial cells, necrosis of smooth muscle fibers & arteries, & culminating in focal hemorrhage. The selective urotoxicity of oxazaphosphorine occurs because the bladder contains a very low concn of thiol cmpds (glutathione, cysteine) which, by virtue of their nucleophilic sulfhydryl groups, are able to react & neutralize many reactive chemicals. Because the metabolic activation of ifosfamide proceeds more slowly than that of cyclophosphamide, doses of ifosfamide are 3-4 times higher than those of cyclophosphamide. This explains the higher incidence of urotoxicity associated with ifosfamide. | |
| Record name | Ifosfamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01181 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | IFOSFAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7023 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from anhyd ether, White crystalline powder | |
CAS No. |
3778-73-2 | |
| Record name | Ifosfamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=3778-73-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Ifosfamide [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0003778732 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Ifosfamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01181 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | ifosfamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759154 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | ifosfamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=109724 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Ifosfamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7020760 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Ifosfamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.021.126 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | IFOSFAMIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/UM20QQM95Y | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | IFOSFAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7023 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ifosfamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015312 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
39-41 °C, 39 - 41 °C | |
| Record name | Ifosfamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01181 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | IFOSFAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7023 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Ifosfamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015312 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















