Lamivudine
Overview
Description
Lamivudine, also known as 2’,3’-dideoxy-3’-thiacytidine, is a synthetic nucleoside analogue with potent antiviral properties. It is primarily used to treat infections caused by the Human Immunodeficiency Virus (HIV) and the Hepatitis B Virus (HBV). This compound is a nucleoside reverse transcriptase inhibitor that works by blocking the reverse transcriptase enzyme, which is essential for viral replication .
Preparation Methods
Synthetic Routes and Reaction Conditions: Lamivudine can be synthesized through various methods, including enantioselective synthesis and dynamic kinetic resolution. One common method involves the use of L-menthyl glyoxylate as a chiral auxiliary. The synthesis typically includes esterification of menthol with carboxyl derivatives such as glyoxyl acid, followed by ozonolysis and hydration .
Industrial Production Methods: In industrial settings, this compound is produced using large-scale chemical synthesis processes that ensure high yield and purity. The process involves multiple steps, including the preparation of intermediates, purification, and crystallization to obtain the final product .
Chemical Reactions Analysis
Types of Reactions: Lamivudine undergoes various chemical reactions, including:
Oxidation: this compound can be oxidized to form its corresponding sulfoxide and sulfone derivatives.
Reduction: Reduction reactions can convert this compound into its dihydro derivatives.
Substitution: Nucleophilic substitution reactions can modify the thiol group in this compound to form different analogues.
Common Reagents and Conditions:
Oxidation: Common oxidizing agents include hydrogen peroxide and potassium permanganate.
Reduction: Reducing agents such as sodium borohydride and lithium aluminum hydride are used.
Substitution: Nucleophiles like alkyl halides and thiolates are employed in substitution reactions.
Major Products: The major products formed from these reactions include sulfoxides, sulfones, dihydro derivatives, and various substituted analogues .
Scientific Research Applications
Treatment of HIV-1
Mechanism of Action
Lamivudine exerts its antiviral effects by inhibiting the reverse transcriptase enzyme, which is crucial for the replication of HIV. This inhibition leads to a reduction in viral load and an increase in CD4+ cell counts, ultimately improving patient quality of life and longevity .
Clinical Studies
Recent studies have demonstrated that this compound remains a cornerstone in HIV treatment regimens. For instance, the DYAD study showed that switching to a combination therapy of dolutegravir and this compound was noninferior to traditional multi-drug regimens in maintaining virologic suppression among adults with HIV . Additionally, this compound is often combined with other antiretrovirals to prevent drug resistance, particularly the M184V mutation associated with rapid viral replication .
Treatment of Hepatitis B
Efficacy in Chronic Hepatitis B
this compound has been shown to be effective in treating chronic hepatitis B. A placebo-controlled study indicated that 63% of patients receiving this compound achieved a complete response at week 24, compared to only 6% in the placebo group . Furthermore, this compound is recognized for its safety profile and efficacy in improving liver histology among patients with HBV .
Infantile Hepatitis B Case Study
A notable case report documented the successful treatment of an infant with hepatitis B using this compound. The early initiation of therapy resulted in significant clinical improvement and may inform future clinical guidelines for managing hepatitis B in infants .
Prophylactic Use
Preventing HBV Reactivation
this compound is also employed prophylactically to prevent HBV reactivation in patients undergoing immunosuppressive therapy or chemotherapy. A meta-analysis revealed that this compound significantly reduces the incidence of HBV reactivation but is associated with a higher risk of developing drug resistance compared to other antiviral agents like entecavir .
Combination Therapies
Innovative Regimens
this compound's role extends into combination therapies, where it is paired with other drugs to enhance efficacy and minimize side effects. For example, studies have shown that combining this compound with dolutegravir maintains high efficacy while reducing the complexity of treatment regimens . This approach is particularly beneficial for treatment-naive patients.
Data Table: Summary of Clinical Applications
Mechanism of Action
Lamivudine exerts its antiviral effects by inhibiting the reverse transcriptase enzyme. Once inside the cell, this compound is phosphorylated to its active triphosphate form, which competes with natural nucleotides for incorporation into the viral DNA. This incorporation results in chain termination, effectively halting viral DNA synthesis and replication .
Comparison with Similar Compounds
Emtricitabine: Another nucleoside reverse transcriptase inhibitor with a similar mechanism of action.
Zidovudine: An older nucleoside analogue used in the treatment of HIV.
Abacavir: A nucleoside reverse transcriptase inhibitor often used in combination with lamivudine
Uniqueness: this compound is unique due to its high efficacy against both HIV-1 and HIV-2, as well as its ability to treat chronic hepatitis B. Its favorable safety profile and low incidence of side effects make it a preferred choice in many antiretroviral therapy regimens .
Biological Activity
Lamivudine, also known as 3TC, is a nucleoside reverse transcriptase inhibitor (NRTI) used primarily in the treatment of Human Immunodeficiency Virus Type 1 (HIV-1) and hepatitis B virus (HBV). Its biological activity is characterized by its mechanism of action, pharmacokinetics, clinical efficacy, and safety profile.
This compound exerts its antiviral effects by being phosphorylated intracellularly to its active form, this compound triphosphate (L-TP). This metabolite competes with naturally occurring nucleotides for incorporation into viral DNA during replication. Due to its structure, L-TP acts as a chain terminator, preventing further elongation of the DNA strand by inhibiting the reverse transcriptase enzyme essential for viral replication. This mechanism is crucial for both HIV and HBV, leading to a reduction in viral load and improvement in patient outcomes .
Pharmacokinetics
The pharmacokinetic profile of this compound includes:
- Absorption : Rapidly absorbed after oral administration with an absolute bioavailability of approximately 86% .
- Distribution : The apparent volume of distribution is about 1.3 L/kg, independent of dose and body weight .
- Protein Binding : Less than 36% binds to plasma proteins .
- Metabolism : this compound undergoes minimal metabolism (about 5.2%) and is primarily excreted unchanged via renal pathways .
- Half-Life : The elimination half-life ranges from 5 to 7 hours .
HIV Treatment
This compound has been shown to be effective in reducing HIV viral load and increasing CD4+ cell counts. In combination therapies, it has demonstrated non-inferiority compared to other NRTIs such as emtricitabine. For instance, studies indicate that this compound combined with dolutegravir shows comparable efficacy to three-drug regimens in ART-naïve individuals .
Hepatitis B Treatment
In chronic hepatitis B patients, this compound has shown significant histological improvement. A one-year trial involving 358 patients indicated that those receiving 100 mg daily exhibited a substantial reduction in necroinflammatory activity and fibrosis progression compared to placebo groups. The study reported a 98% reduction in HBV DNA levels at week 52 and a seroconversion rate of 16% for HBeAg .
Case Studies
- HIV Cohort Study : A cohort analysis from the ATHENA study highlighted that patients receiving this compound had a higher risk of virological failure compared to those on efavirenz-based regimens. This indicates the need for careful monitoring and possible adjustments in therapy for patients on this compound .
- Chronic Hepatitis B Trial : In a randomized trial comparing different dosages of this compound, the 100 mg group showed the highest rates of sustained normalization of alanine aminotransferase levels (72%) and effective suppression of HBV DNA .
Safety Profile
This compound is generally well-tolerated with a low incidence of severe adverse effects. Common side effects include headache, fatigue, and gastrointestinal disturbances. Long-term use can lead to the development of resistance mutations such as M184V in HIV patients, underscoring the importance of combination therapy to mitigate this risk .
Q & A
Basic Research Questions
Q. What are the standard methodological approaches for quantifying lamivudine in pharmaceutical formulations, and how are these methods validated?
- Answer : Reverse-phase HPLC with UV detection is widely used for simultaneous quantification of this compound and co-administered drugs (e.g., zidovudine). Validation includes robustness testing under variations in mobile phase composition (e.g., ±2% acetonitrile), pH (±0.2 units), and flow rate (±0.1 mL/min). Acceptable precision requires %RSD <2% for replicate analyses. For example, a validated method achieved 100.02% recovery for this compound in marketed tablets, ensuring reliability for pharmacokinetic or stability studies .
Q. What safety protocols should researchers follow when handling this compound in laboratory settings?
- Answer : this compound is classified under GHS as a Category 2 specific target organ toxicant (repeated exposure). Mandatory precautions include:
- Use of PPE (gloves, lab coats, and eye protection) to avoid dermal contact .
- Conducting experiments in well-ventilated areas or fume hoods to prevent inhalation of aerosols .
- Implementing rigorous waste disposal protocols for contaminated materials. Safety data sheets (SDS) must be reviewed prior to experimental design .
Advanced Research Questions
Q. How can researchers design experiments to evaluate this compound’s pharmacokinetic alterations in post-bariatric surgery populations?
- Answer : Longitudinal pharmacokinetic (PK) studies should compare pre- and post-surgery plasma concentrations using high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS). Key considerations include:
- Sampling at multiple time points (e.g., 0, 1, 2, 4, 8, 12 hours post-dose) to capture absorption changes due to altered gastrointestinal anatomy.
- Inclusion of control groups (non-surgical patients) to isolate surgery-specific effects. For example, post-sleeve gastrectomy (SG) cases showed a 2.5-fold increase in this compound exposure, necessitating dose adjustments .
Q. What computational strategies are effective in predicting this compound’s spectral properties, and how do these compare to experimental data?
- Answer : Density functional theory (DFT) simulations can predict UV-Vis and IR spectra by modeling molecular orbitals and vibrational modes. Validation involves:
- Calculating % error between predicted and experimental absorption frequencies (e.g., −24.26% to +18.89% error observed for this compound’s IR spectra).
- Regression analysis (e.g., R² = 0.970 for UV-Vis correlations) to assess model reliability. Discrepancies may arise from solvent effects or approximations in theoretical frameworks .
Q. How can adaptive Bayesian optimization (AdBO) improve this compound crystallization process development?
- Answer : AdBO reduces experimental iterations by predicting optimal conditions (e.g., supersaturation, temperature) for nucleation and crystal growth. Key steps include:
- Defining parameter search spaces based on prior kinetic data (e.g., induction times for slow kinetics in this compound).
- Iteratively updating Bayesian models with in-situ PAT (process analytical technology) data. A case study achieved a 5-fold reduction in material usage compared to traditional DoE approaches, enhancing sustainability .
Q. How should researchers address contradictions between computational predictions and experimental results in this compound studies?
- Answer : Discrepancies (e.g., in UV-Vis spectral predictions) require systematic error analysis:
- Identify sources of variance (e.g., implicit solvation models in DFT vs. experimental solvent interactions).
- Refine computational parameters (e.g., basis sets, exchange-correlation functionals) to better approximate experimental conditions.
- Validate with orthogonal methods (e.g., NMR crystallography for structural verification) .
Q. What experimental designs are optimal for studying this compound resistance mechanisms in in vitro models?
- Answer : Use site-directed mutagenesis to introduce known resistance-associated mutations (e.g., M184V in HIV reverse transcriptase). Key steps:
- Conduct time-kill assays to quantify viral replication efficiency under this compound pressure.
- Pair with molecular dynamics simulations to map drug-binding affinity changes.
- Cross-validate with clinical isolates to ensure translational relevance .
Q. Methodological Guidelines
- Data Interpretation : For spectral or pharmacokinetic data, always report confidence intervals (e.g., 95% CI for R² values) and effect sizes to contextualize findings .
- Ethical Compliance : When using human-derived samples (e.g., post-surgery PK studies), ensure institutional review board (IRB) approval and explicit informed consent protocols .
Properties
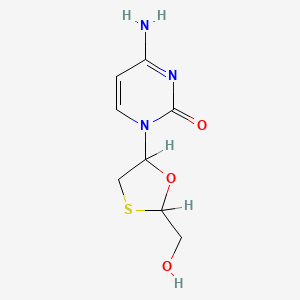
IUPAC Name |
4-amino-1-[(2R,5S)-2-(hydroxymethyl)-1,3-oxathiolan-5-yl]pyrimidin-2-one | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C8H11N3O3S/c9-5-1-2-11(8(13)10-5)6-4-15-7(3-12)14-6/h1-2,6-7,12H,3-4H2,(H2,9,10,13)/t6-,7+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
JTEGQNOMFQHVDC-NKWVEPMBSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1C(OC(S1)CO)N2C=CC(=NC2=O)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C1[C@H](O[C@H](S1)CO)N2C=CC(=NC2=O)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C8H11N3O3S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID7023194 | |
| Record name | Lamivudine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7023194 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
229.26 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Lamivudine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014847 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
In water, 70,000 mg/L @ 20 °C, 2.76e+00 g/L | |
| Record name | Lamivudine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00709 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LAMIVUDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7155 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Lamivudine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014847 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Vapor Pressure |
8.3X10-16 mm Hg @ 25 °C /Estimated/ | |
| Record name | LAMIVUDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7155 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Lamivudine is a synthetic nucleoside analogue and is phosphorylated intracellularly to its active 5'-triphosphate metabolite, lamivudine triphosphate (L-TP). This nucleoside analogue is incorporated into viral DNA by HIV reverse transcriptase and HBV polymerase, resulting in DNA chain termination., Lamivudine enters cells by passive diffusion and is phosphorylated to its active metabolite, lamivudine triphosphate. Lamivudine triphosphate competes with deoxycytidine triphosphate for binding to reverse transcriptase, and incorporation into DNA results in chain termination. Lamivudine has very low affinity for human alpha and omega DNA polymerases, moderate affinity for beta DNA polymerase, and higher affinity for gamma DNA polymerase. | |
| Record name | Lamivudine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00709 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LAMIVUDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7155 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from boiling ethanol | |
CAS No. |
134678-17-4 | |
| Record name | Lamivudine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=134678-17-4 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Lamivudine [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0134678174 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Lamivudine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00709 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Lamivudine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID7023194 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | LAMIVUDINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/2T8Q726O95 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | LAMIVUDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7155 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Lamivudine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014847 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
160-162 °C, 160 - 162 °C | |
| Record name | Lamivudine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00709 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LAMIVUDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7155 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Lamivudine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014847 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.
















