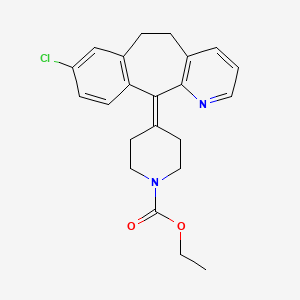
Loratadine
Overview
Description
Loratadine is a second-generation antihistamine widely used to manage symptoms of allergic rhinitis and urticaria (hives). It is known for its effectiveness in treating allergy symptoms such as sneezing, itching, and watery eyes without causing significant sedation, making it preferable over first-generation antihistamines .
Preparation Methods
Synthetic Routes and Reaction Conditions: Loratadine can be synthesized through various methods. One common method involves the reaction of ethyl 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)-1-piperidinecarboxylate with appropriate reagents under controlled conditions. The process typically includes steps such as dissolving crude this compound in an organic solvent, adding active carbon for adsorption, heating, stirring, and filtering to obtain purified this compound .
Industrial Production Methods: In industrial settings, this compound is produced using high-speed shear-high pressure homogenization followed by freeze-drying to create this compound nanocrystals. This method enhances the solubility and bioavailability of this compound, making it more effective for oral administration .
Chemical Reactions Analysis
Types of Reactions: Loratadine undergoes various chemical reactions, including oxidation, reduction, and substitution. For instance, oxidation is likely to occur in the piperidine and cycloheptane rings .
Common Reagents and Conditions: Common reagents used in these reactions include hydrogen peroxide for oxidation and sodium borohydride for reduction. The conditions typically involve controlled temperatures and pH levels to ensure the desired reaction outcomes.
Major Products Formed: The major products formed from these reactions include desthis compound, which is an active metabolite of this compound and retains antihistaminic properties .
Scientific Research Applications
Clinical Applications
Loratadine is primarily indicated for:
- Allergic Rhinitis : It alleviates symptoms such as sneezing, runny nose, and itchy eyes.
- Urticaria : It reduces wheal formation and itching associated with hives.
- Other Allergic Conditions : It may be used for various dermatological allergies.
Allergic Rhinitis
A double-blind placebo-controlled study demonstrated that this compound significantly relieved nasal symptoms compared to placebo within three days of treatment. In this study involving 69 patients allergic to grass pollen, this compound showed a rapid onset of action and was well-tolerated without significant side effects .
Combination Therapy
Recent meta-analysis results indicate that combining this compound with montelukast can significantly enhance the reduction of total nasal symptom scores compared to either agent alone or placebo. This combination therapy is particularly beneficial for patients with moderate to severe allergic rhinitis .
Safety Profile
This compound has a favorable safety profile, especially in pediatric and geriatric populations. It is generally well-tolerated, with minimal side effects compared to first-generation antihistamines. However, caution is advised in elderly patients or those on higher doses due to potential cardiotoxicity risks .
Data Table: Summary of Clinical Studies on this compound
Mechanism of Action
Loratadine acts as a selective inverse agonist for peripheral histamine H1-receptors. When histamine is released during an allergic reaction, it binds to these receptors, causing symptoms such as itching and sneezing. This compound blocks this binding, effectively halting the allergic response . It has minimal effects on the central nervous system, reducing the risk of sedation .
Comparison with Similar Compounds
Loratadine is often compared with other second-generation antihistamines such as cetirizine and fexofenadine. Unlike first-generation antihistamines like diphenhydramine, this compound does not cross the blood-brain barrier significantly, resulting in fewer sedative effects . Similar compounds include:
Cetirizine: Known for its effectiveness in treating allergic reactions but may cause mild drowsiness.
Fexofenadine: Another non-sedating antihistamine with a similar mechanism of action.
Chlorpheniramine: A first-generation antihistamine that is effective but causes significant sedation.
This compound’s unique advantage lies in its ability to provide effective allergy relief without causing drowsiness, making it a preferred choice for many patients.
Biological Activity
Loratadine is a widely used second-generation antihistamine primarily prescribed for the treatment of allergic rhinitis and chronic urticaria. Recent studies have expanded its profile, revealing additional biological activities beyond its antihistaminic effects. This article explores the multifaceted biological activities of this compound, including its anti-inflammatory properties, effects on bacterial virulence, and implications in various clinical settings.
This compound functions by selectively antagonizing peripheral H1 receptors, thereby inhibiting the physiological effects of histamine, which include vasodilation and increased vascular permeability. Unlike first-generation antihistamines, this compound does not readily cross the blood-brain barrier, which minimizes central nervous system side effects such as sedation.
Anti-Inflammatory Effects
Recent research has highlighted this compound's potential as an anti-inflammatory agent. A study demonstrated that this compound effectively suppresses inflammation by targeting the TAK1 signaling pathway, which subsequently inhibits the activation of the AP-1 transcription factor. This inhibition leads to a significant reduction in pro-inflammatory gene expression, including matrix metalloproteinases (MMP1, MMP3, MMP9) and cytokines (IL-6, TNF-α) in macrophage cell lines and in vivo models .
Table 1: Summary of Anti-Inflammatory Effects
Inhibition of Bacterial Virulence
This compound has also been shown to inhibit the virulence of Staphylococcus aureus, a significant pathogen responsible for various infections. In vitro studies indicated that concentrations as low as 25 μM could effectively reduce biofilm formation and virulence factor production without impacting bacterial growth. Notably, this compound reduced the expression of several virulence-related genes in clinical isolates .
Table 2: Effects on Staphylococcus aureus
| Concentration | Effect on Biofilm Formation | Impact on Virulence Factors |
|---|---|---|
| 25 μM | Inhibition observed | Reduced hemolysin and pigmentation; downregulation of agrA, splB genes |
| 50 μM | Significant inhibition | Further reduction in mRNA levels of virulence genes |
Clinical Applications
This compound's biological activity extends to various clinical applications beyond allergy treatment. A randomized controlled trial showed that this compound significantly alleviated pegfilgrastim-induced bone pain in cancer patients, suggesting its utility in managing pain associated with chemotherapy . Additionally, a meta-analysis indicated that combining this compound with montelukast significantly improved nasal symptom scores in allergic rhinitis patients compared to either drug alone .
Case Study: this compound for Bone Pain Management
In a study involving 213 cancer patients undergoing pegfilgrastim treatment:
Q & A
Basic Research Questions
Q. How can researchers design a validated HPLC method for simultaneous quantification of Loratadine and pseudoephedrine in combination formulations?
- Methodological Answer : Utilize a cation-exchange column and apply experimental design optimization (e.g., factorial design) to balance resolution, retention time, and sensitivity. Validate the method by assessing accuracy (bias < 2.0%), repeatability, intermediate precision (%RSD < 2.0%), and system suitability parameters (e.g., tailing factor, theoretical plates) . Include robustness testing under varying mobile phase compositions and flow rates.
Q. What statistical approaches are essential for analyzing pharmacokinetic data from this compound bioavailability studies?
- Methodological Answer : Apply a two-way ANOVA to evaluate carryover effects and inter-product variability. Use natural log (Ln) transformation for plasma concentration data to stabilize variance. Calculate 90% confidence intervals (80–125%) for AUC0-t, AUC0-inf, and Cmax to establish bioequivalence. Ensure compliance with regulatory guidelines for parametric analysis of Ln-transformed metrics .
Q. How can researchers ensure reproducibility in quantifying this compound and its metabolite desthis compound in plasma?
- Methodological Answer : Employ LC-MS/MS with MRM transitions (e.g., 383.0 → 337.0 for this compound). Validate the method using matrix-matched calibration curves and quality controls. Address matrix effects via post-column infusion studies and internal standardization. Include stability tests for freeze-thaw cycles and long-term storage .
Advanced Research Questions
Q. What experimental strategies resolve contradictions in this compound’s metabolic pathway contributions across CYP enzymes?
- Methodological Answer : Conduct in vitro inhibition and correlation studies using human liver microsomes to quantify CYP3A4, CYP2D6, and CYP2C19 contributions. Apply siRNA knockdown of specific CYP isoforms (e.g., NRF2 in chondrocytes) to isolate metabolic pathways. Pair clinical pharmacokinetic data with genotyping (e.g., CYP2D6 polymorphisms) to explain inter-subject variability .
Q. How can researchers validate this compound’s anticancer mechanisms observed in retrospective cohort studies?
- Methodological Answer : Use RNA-seq to identify apoptosis- and pyroptosis-related genes modulated by this compound in lung cancer cell lines (e.g., A549). Validate findings with in vivo xenograft models, measuring tumor volume and caspase-3 activation. Cross-reference clinical survival data with histamine receptor expression levels in tumor biopsies .
Q. What methodologies address the environmental impact of this compound in freshwater ecosystems?
- Methodological Answer : Perform ecotoxicological assays (e.g., Daphnia magna acute toxicity tests) to determine EC50 values. Model environmental fate using hydrophobicity (log Kow = 5.20) and photolysis susceptibility. Prioritize analysis of WWTP sludge samples via SPE-LC-MS/MS to quantify sorption behavior .
Q. How can response surface methodology optimize this compound’s buccoadhesive wafer formulation?
- Methodological Answer : Apply a 3<sup>2</sup> factorial design to evaluate sodium alginate and lactose monohydrate effects on bioadhesive force and drug release. Use Design-Expert software to generate contour plots and desirability functions. Validate optimized formulations via DSC, FTIR, and in vivo retention studies .
Q. Data Analysis & Contradiction Resolution
Q. How should researchers reconcile discrepancies in this compound’s anti-inflammatory effects across different cell models?
- Methodological Answer : Standardize experimental conditions (e.g., AGEs concentration in SW1353 chondrocytes vs. macrophage models). Use multiplex ELISA to quantify NLRP3 inflammasome components (e.g., IL-1β, caspase-1) and Western blotting for NOX4 expression. Perform meta-analysis of RNA-seq datasets to identify context-dependent signaling pathways .
Q. What statistical frameworks address variability in this compound’s steady-state plasma concentrations?
- Methodological Answer : Implement population pharmacokinetic (PopPK) modeling to account for covariates like CYP genotype, age, and renal function. Use NONMEM or Monolix for parameter estimation. Validate models with bootstrapping and visual predictive checks to ensure robustness .
Q. Experimental Design & Validation
Q. How to design a study evaluating this compound’s drug-drug interactions with CYP3A4 inhibitors (e.g., omeprazole)?
- Methodological Answer : Conduct a crossover pharmacokinetic study in beagles, comparing this compound exposure with/without omeprazole co-administration. Use LC-MS/MS for plasma quantification and non-compartmental analysis for AUC comparisons. Include in vitro CYP3A4 inhibition assays to confirm mechanistic overlap .
Properties
IUPAC Name |
ethyl 4-(13-chloro-4-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,12,14-hexaen-2-ylidene)piperidine-1-carboxylate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C22H23ClN2O2/c1-2-27-22(26)25-12-9-15(10-13-25)20-19-8-7-18(23)14-17(19)6-5-16-4-3-11-24-21(16)20/h3-4,7-8,11,14H,2,5-6,9-10,12-13H2,1H3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
JCCNYMKQOSZNPW-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCOC(=O)N1CCC(=C2C3=C(CCC4=C2N=CC=C4)C=C(C=C3)Cl)CC1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C22H23ClN2O2 | |
| Record name | loratadine | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Loratadine | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID2023224 | |
| Record name | Loratadine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2023224 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
382.9 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
<1 mg/ml at 25°C | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Histamine release is a key mediator in allergic rhinitis and urticaria. As a result, loratadine exerts it's effect by targeting H1 histamine receptors. Loratadine binds to H1 histamine receptors found on the surface of epithelial cells, endothelial cells, eosinophils, neutrophils, airway cells, and vascular smooth muscle cells among others. H1 histamine receptors fall under the wider umbrella of G-protein coupled receptors, and exist in a state of equilibrium between the active and inactive forms. Histamine binding to the H1-receptor facilitates cross linking between transmembrane domains III and V, stabilizing the active form of the receptor. On the other hand, antihistamines bind to a different site on the H1 receptor favouring the inactive form. Hence, loratadine can more accurately be classified as an "inverse agonist" as opposed to a "histamine antagonist", and can prevent or reduce the severity of histamine mediated symptoms., All of the available H1 receptor antagonists are reversible, competitive inhibitors of the interaction of histamine with H1 receptors. /H1 Receptor Antagonists/, H1 antagonists inhibit most responses of smooth muscle to histamine. /H1 Antagonists Receptors/, Within the vascular tree, the H1 antagonists inhibit both the vasoconstrictor effects of histamine and, to a degree, the more rapid vasodilator effects that are mediated by H1 receptors on endothelial cells. /H1 Receptor Antagonists/, H1 antagonists strongly block the action of histamine that results in increased capillary permeability and formation of edema and wheal. /H1 Receptor Antagonists/, For more Mechanism of Action (Complete) data for LORATADINE (6 total), please visit the HSDB record page. | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from acetonitrile | |
CAS No. |
79794-75-5 | |
| Record name | Loratadine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=79794-75-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Loratadine [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0079794755 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | loratadine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758628 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | loratadine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=721075 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Loratadine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2023224 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | ethyl 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)piperidine-1-carboxylate | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.216.235 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | 1-Piperidinecarboxylic acid, 4-(8-chloro-5,6-dihydro-11H-benzo[5,6]cyclohepta[1,2-b]pyridin-11-ylidene)-, ethyl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.120.122 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | LORATADINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/7AJO3BO7QN | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
134-136 °C, 134 - 136 °C | |
| Record name | Loratadine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00455 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | LORATADINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3578 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Loratadine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005000 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details









Synthesis routes and methods II
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














