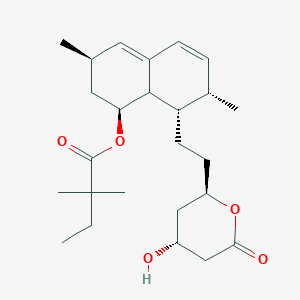
Simvastatin
Overview
Description
Simvastatin is a lipid-lowering medication belonging to the statin class. It is primarily used to reduce elevated lipid levels and decrease the risk of cardiovascular events such as heart attacks and strokes. This compound works by inhibiting the enzyme hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase, which plays a crucial role in cholesterol biosynthesis .
Preparation Methods
Simvastatin is synthesized from a fermentation product of the fungus Aspergillus terreus. The synthetic route involves several key steps:
Fermentation: The initial step involves the fermentation of Aspergillus terreus to produce the precursor compound.
Chemical Modification: The precursor undergoes chemical modifications, including esterification and lactonization, to form this compound.
Industrial production methods focus on optimizing these steps to ensure high yield and purity. The process involves stringent control of reaction conditions such as temperature, pH, and solvent use to achieve the desired product.
Chemical Reactions Analysis
Simvastatin undergoes several types of chemical reactions:
Hydrolysis: This compound can be hydrolyzed to its active β-hydroxyacid form by carboxyesterases in the liver and intestines.
Reduction and Substitution: These reactions are less common but can occur under specific conditions.
Common reagents used in these reactions include water for hydrolysis and cytochrome P450 enzymes for oxidation. The major products formed include the active β-hydroxyacid form and various oxidized metabolites.
Scientific Research Applications
Primary Use in Cardiovascular Health
Cholesterol Management:
Simvastatin is primarily indicated for the treatment of hyperlipidemia, specifically to lower levels of low-density lipoprotein cholesterol (LDL-C) and total cholesterol. It is effective in reducing the risk of cardiovascular events such as myocardial infarction and stroke. The drug functions as an HMG-CoA reductase inhibitor, blocking the enzyme responsible for cholesterol production in the liver .
Table 1: FDA-Approved Indications for this compound
| Indication | Description |
|---|---|
| Homozygous Familial Hypercholesterolemia | Genetic condition leading to very high cholesterol levels |
| Heterozygous Familial Hypercholesterolemia | Common genetic condition with elevated cholesterol levels |
| Hypertriglyceridemia | High levels of triglycerides in the blood |
| Dysbetalipoproteinemia | Abnormal lipid metabolism leading to high cholesterol |
| Cardiovascular Event Prevention | Reduces risk of heart attacks and strokes |
Antimicrobial Properties
Recent studies have explored this compound's potential as an antimicrobial agent. Research indicates that this compound exhibits activity against various pathogens, including Enterococcus faecalis , suggesting it may play a role in preventing infections .
Case Study: Antimicrobial Activity
In a study examining this compound's antimicrobial effects, it was found that treatment reduced bacterial load significantly in infected models, indicating its potential use as an adjunct therapy in infectious diseases .
Neuroprotective Effects
This compound has garnered attention for its possible neuroprotective properties, particularly in relation to Alzheimer's disease. Evidence suggests that this compound may reduce the incidence of Alzheimer's by:
- Anti-Inflammatory Effects: Reducing neuroinflammation associated with neurodegenerative diseases.
- Amyloid Beta Reduction: Lowering amyloid beta levels, which are implicated in Alzheimer's pathology.
- Mitochondrial Health Improvement: Enhancing mitochondrial function, which is crucial for neuronal health .
Table 2: Mechanisms of Neuroprotection by this compound
| Mechanism | Description |
|---|---|
| Anti-Inflammatory | Reduces inflammation in neural tissues |
| Amyloid Beta Reduction | Lowers levels of amyloid plaques |
| Mitochondrial Function | Improves energy production in neurons |
| Blood-Brain Barrier Penetration | Facilitates transport across the blood-brain barrier |
Vascular Health Enhancement
Research has revealed that this compound improves endothelial function and vascular health independent of its lipid-lowering effects. Studies conducted on endothelial cells demonstrated that this compound promotes angiogenesis (formation of new blood vessels) and prevents harmful transitions from endothelial to mesenchymal cells .
Case Study: Vascular Function Improvement
In diabetic mouse models, this compound treatment resulted in enhanced vascular function, indicating its potential application in managing diabetes-related vascular complications .
Off-Label Uses
This compound is also utilized off-label for several indications:
Mechanism of Action
Simvastatin exerts its effects by competitively inhibiting HMG-CoA reductase, the enzyme responsible for converting HMG-CoA to mevalonic acid, a precursor in cholesterol biosynthesis . This inhibition leads to a decrease in cholesterol synthesis in the liver, resulting in lower levels of low-density lipoprotein (LDL) cholesterol in the blood. Additionally, this compound has pleiotropic effects, including anti-inflammatory and antioxidant properties .
Comparison with Similar Compounds
Simvastatin is often compared with other statins such as atorvastatin, rosuvastatin, pravastatin, fluvastatin, and lovastatin. While all these compounds share a similar mechanism of action, they differ in their pharmacokinetic properties, potency, and side effect profiles . For example:
Atorvastatin: Known for its high potency and longer half-life compared to this compound.
Rosuvastatin: Has a higher efficacy in lowering LDL cholesterol but may cause more muscle-related side effects.
Pravastatin: Less lipophilic and has fewer drug interactions compared to this compound.
This compound’s unique ability to cross the blood-brain barrier and its extensive use in cardiovascular disease management highlight its distinct advantages .
Biological Activity
Simvastatin, a widely used statin for lowering cholesterol, has garnered attention for its diverse biological activities beyond lipid regulation. This article delves into the compound's mechanisms of action, antibacterial properties, and implications in various diseases, supported by research findings and case studies.
This compound primarily functions as an HMG-CoA reductase inhibitor, which is pivotal in cholesterol biosynthesis. By inhibiting this enzyme, this compound reduces hepatic cholesterol levels, leading to increased uptake of low-density lipoprotein (LDL) from the bloodstream. This mechanism is well-documented in clinical settings where this compound is prescribed to manage hyperlipidemia and reduce cardiovascular risks .
Key Metabolic Pathways Affected by this compound
- Cholesterol Biosynthesis : this compound inhibits the mevalonate pathway, crucial for cholesterol production.
- Protein Synthesis : Research indicates that this compound selectively inhibits bacterial protein synthesis without significantly affecting mammalian cells .
- Autophagy Modulation : this compound activates AMP-activated protein kinase (AMPK) and inhibits mTORC1, promoting autophagy, which has implications in cancer and infectious diseases .
Antibacterial Activity
Recent studies have highlighted this compound's antibacterial properties, particularly against Gram-positive bacteria such as Staphylococcus aureus.
In Vitro Findings
- Inhibition of Bacterial Growth : this compound demonstrated significant inhibition of macromolecular synthesis in S. aureus, affecting DNA, RNA, and protein synthesis pathways .
- Toxin Production Suppression : The compound effectively reduced the production of virulence factors like Panton-Valentine leukocidin (PVL) and α-hemolysin in methicillin-resistant S. aureus (MRSA) .
| Study | Bacteria | Effect | Concentration |
|---|---|---|---|
| Nature (2015) | S. aureus | Inhibition of protein synthesis | < 0.25 × MIC |
| PubMed (2020) | M. tuberculosis | Anti-tubercular activity | Clinically relevant doses |
Cardiovascular Disease
This compound is primarily used for cardiovascular disease prevention. Clinical trials have shown its effectiveness in reducing LDL levels and lowering the risk of heart attacks and strokes.
Diabetes Management
Emerging evidence suggests that this compound may improve glycemic control in diabetic patients when used alongside standard treatments. A systematic review indicated that periodontal disease treatment with subgingival this compound could lead to modest improvements in blood sugar levels among Type 2 diabetes patients .
Case Studies
- Antimicrobial Efficacy : A study explored this compound’s effect on various bacterial strains, revealing a synergistic effect when combined with traditional antibiotics like colistin against resistant strains .
- Cholesterol Reduction and Cancer Prevention : The HPS study assessed this compound’s role in colorectal cancer prevention but found no significant benefits when combined with antioxidants .
Q & A
Basic Research Questions
Q. What experimental designs are commonly used to optimize simvastatin formulations, and how do they ensure robustness?
Researchers employ statistical experimental designs such as D-optimal design and full-factorial design to evaluate critical variables in formulation development. For example, D-optimal design was applied to assess HPLC method robustness for this compound purity testing, analyzing variables like mobile phase composition, pH, and column temperature. This design minimizes experimental runs while maximizing predictive accuracy, with coefficients of determination (R² > 0.95) confirming model validity . Similarly, full-factorial designs optimize transdermal patches by testing factors like croscarmellose sodium concentration and compression force, enabling predictive polynomial equations for dissolution profiles .
Q. What are standard methodologies for quantifying this compound in pharmacokinetic studies?
High-performance liquid chromatography (HPLC) remains the gold standard. A validated protocol includes monitoring retention time, theoretical plates, and peak asymmetry. For instance, a study using Modde® software demonstrated HPLC robustness across variable ranges, with resolution of critical impurity pairs (e.g., A/I, E/F) ensuring accurate quantification . Dialysis techniques are also used to purify this compound in lipid nanoparticles, with ANOVA confirming no significant difference in free drug removal after 15 vs. 30 minutes (p > 0.05) .
Q. How are clinical trial endpoints selected for evaluating this compound’s efficacy in cardiovascular disease?
Primary endpoints often include carotid intima-media thickness (CIMT) and LDL cholesterol reduction . In a 24-month trial, this compound combined with ezetimibe reduced LDL by 16.5% (p < 0.01) but showed no significant CIMT improvement compared to monotherapy, highlighting the complexity of linking lipid metrics to clinical outcomes . Subgroup analyses in diabetic patients with coronary heart disease prioritized major CHD events (RR = 0.45, p = 0.002) and total mortality (RR = 0.57, p = 0.087) as key endpoints .
Advanced Research Questions
Q. How can researchers address contradictions between this compound’s biochemical efficacy and clinical outcomes?
Contradictions, such as LDL reduction without atherosclerosis improvement, require subgroup analysis and meta-regression . For example, in familial hypercholesterolemia trials, this compound + ezetimibe reduced LDL and CRP but did not alter CIMT progression, suggesting pleiotropic effects may not translate to structural benefits. Researchers must stratify data by risk factors (e.g., diabetes, baseline CRP) and apply multivariate models to isolate confounding variables . PRISMA-guided systematic reviews further contextualize contradictions by aggregating data across study designs .
Q. What advanced statistical models are used to optimize this compound delivery systems?
Response surface methodology (RSM) with Box-Behnken designs is widely used. A study optimizing transdermal patches employed a three-factor, three-level design to model this compound permeation, achieving a quadratic model with R² = 0.9948. Derringer’s desirability function predicted optimal permeation (78.77 µg/cm²) at specific poloxamer and D-limonene concentrations . Similarly, Design Expert® software analyzes dissolution data via ANOVA and multiple linear regression, identifying significant interactions between formulation variables .
Q. How can researchers investigate this compound’s pleiotropic effects in non-cardiovascular contexts, such as neuroprotection or bone metabolism?
Preclinical models use immunohistochemistry and longitudinal MRI to assess mechanisms. In experimental periodontitis, this compound reduced alveolar bone loss by downregulating RANKL (p < 0.05) and upregulating osteoprotegerin (OPG), confirmed via immunofluorescence . For neuroprotection, rodent ICH models combined MRI (susceptibility-weighted imaging) with neurobehavioral tests, showing this compound reduced tissue loss by 28% (p = 0.0003) and enhanced neurogenesis .
Q. Methodological Considerations
Q. How should researchers design studies to evaluate this compound’s antioxidant and anti-inflammatory properties?
- Oxidative stress markers : Measure gingival glutathione (GSH) and malonaldehyde (MDA) levels, as done in periodontitis models .
- Cytokine profiling : Use ELISA or multiplex assays for IL-1β, TNF-α, and IL-10 in tissue homogenates.
- Gene/protein expression : Apply qPCR and Western blotting for iNOS, MMPs, and BMP-2 .
- Statistical rigor : Two-way ANOVA with post-hoc tests (e.g., Tukey’s) to compare treatment groups.
Q. What are best practices for handling this compound’s low aqueous solubility in formulation studies?
- Surfactant optimization : Increase poloxamer concentration to enhance solubility, as demonstrated in solid lipid nanoparticles (SLNs) .
- Organic co-solvents : Use isopropyl alcohol (IPA) in nanodispersions, balancing solubility and toxicity .
- Dissolution testing : USP Apparatus II (paddle method) in phosphate buffer (pH 7.4) at 100 rpm, with sampling at 5–30 minutes .
Properties
IUPAC Name |
[(1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-hydroxy-6-oxooxan-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl] 2,2-dimethylbutanoate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C25H38O5/c1-6-25(4,5)24(28)30-21-12-15(2)11-17-8-7-16(3)20(23(17)21)10-9-19-13-18(26)14-22(27)29-19/h7-8,11,15-16,18-21,23,26H,6,9-10,12-14H2,1-5H3/t15-,16-,18+,19+,20-,21-,23-/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
RYMZZMVNJRMUDD-HGQWONQESA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCC(C)(C)C(=O)OC1CC(C=C2C1C(C(C=C2)C)CCC3CC(CC(=O)O3)O)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CCC(C)(C)C(=O)O[C@H]1C[C@H](C=C2[C@H]1[C@H]([C@H](C=C2)C)CC[C@@H]3C[C@H](CC(=O)O3)O)C | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C25H38O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID0023581 | |
| Record name | Simvastatin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0023581 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
418.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Simvastatin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005007 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Insoluble, In water, 3.0X10-2 mg/L, temp not specified, Solubility (mg/mL): chloroform 610; DMSO 540; methanol 200; ethanol 160; n-hexane 0.15; 0.1 M HCl 0.06; polyethylene glycol-400 70; propylene glycol 30; 0.1 M NaOH 70, 1.22e-02 g/L | |
| Record name | Simvastatin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00641 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Simvastatin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Simvastatin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005007 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Simvastatin is a prodrug in which the 6-membered lactone ring of simvastatin is hydrolyzed in vivo to generate the beta,delta-dihydroxy acid, an active metabolite structurally similar to HMG-CoA (hydroxymethylglutaryl CoA). Once hydrolyzed, simvastatin competes with HMG-CoA for HMG-CoA reductase, a hepatic microsomal enzyme, which catalyzes the conversion of HMG-CoA to mevalonate, an early rate-limiting step in cholesterol biosynthesis. Simvastatin acts primarily in the liver, where decreased hepatic cholesterol concentrations stimulate the upregulation of hepatic low density lipoprotein (LDL) receptors which increases hepatic uptake of LDL. Simvastatin also inhibits hepatic synthesis of very low density lipoprotein (VLDL). The overall effect is a decrease in plasma LDL and VLDL. At therapeutic doses, the HMG-CoA enzyme is not completely blocked by simvastatin activity, thereby allowing biologically necessary amounts of mevalonate to remain available. As mevalonate is an early step in the biosynthetic pathway for cholesterol, therapy with simvastatin would also not be expected to cause any accumulation of potentially toxic sterols. In addition, HMG-CoA is metabolized readily back to acetyl-CoA, which participates in many biosynthetic processes in the body. In vitro and in vivo animal studies also demonstrate that simvastatin exerts vasculoprotective effects independent of its lipid-lowering properties, also known as the pleiotropic effects of statins. This includes improvement in endothelial function, enhanced stability of atherosclerotic plaques, reduced oxidative stress and inflammation, and inhibition of the thrombogenic response. Statins have also been found to bind allosterically to β2 integrin function-associated antigen-1 (LFA-1), which plays an important role in leukocyte trafficking and in T cell activation., Simvastatin is a prodrug and is hydrolyzed to its active beta-hydroxyacid form, simvastatin acid, after administration. Simvastatin is a specific inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonate, an early and rate limiting step in the biosynthetic pathway for cholesterol. In addition, simvastatin reduces VLDL and TG and increases HDL-C., The HDL-associated enzyme paraoxonase protects LDLs from oxidative stress. 3-Hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statins) appear to favorably influence the atherosclerotic process by different mechanisms. The present study examined the influence of simvastatin on paraoxonase expression and serum paraoxonase levels. Simvastatin upregulated in a dose-dependent manner the activity of the promoter of the paraoxonase gene in expression cassettes transfected into HepG2 cells. Upregulation could be blocked by mevalonate and other intermediates of the cholesterol biosynthetic pathway. Simvastatin increased nuclear factors, notably sterol regulatory element-binding protein-2, capable of binding to the paraoxonase promoter; this was also blocked by mevalonate. Sterol regulatory element-binding protein-2 upregulated promoter activity in vitro. Patients treated with statin showed a significant increase in serum concentrations and activities of paraoxonase. The data indicate that simvastatin can modulate expression in vitro of the antioxidant enzyme paraoxonase and is associated with increased serum paraoxonase concentration and activity. It is consistent with effects of simvastatin treatment, which have the potential to influence beneficially antiatherogenic mechanisms at the HDL level. The study provides evidence for 1 molecular mechanism by which paraoxonase gene expression could be regulated., ... We report in this work that, unexpectedly, simvastatin enhances LPS-induced IL-12p40 production by murine macrophages, and that it does so by activating the IL-12p40 promoter. Mutational analysis and dominant-negative expression studies indicate that both C/EBP and AP-1 transcription factors have a crucial role in promoter activation. This occurs via a c-Fos- and c-Jun-based mechanism; we demonstrate that ectopic expression of c-Jun activates the IL-12p40 promoter, whereas expression of c-Fos inhibits IL-12p40 promoter activity. Simvastatin prevents LPS-induced c-Fos expression, thereby relieving the inhibitory effect of c-Fos on the IL-12p40 promoter. Concomitantly, simvastatin induces the phosphorylation of c-Jun by the c-Jun N-terminal kinase, resulting in c-Jun-dependent activation of the IL-12p40 promoter. This appears to be a general mechanism because simvastatin also augments LPS-dependent activation of the TNF-alpha promoter, perhaps because the TNF-alpha promoter has C/EBP and AP-1 binding sites in a similar configuration to the IL-12p40 promoter. The fact that simvastatin potently augments LPS-induced IL-12p40 and TNF-alpha production has implications for the treatment of bacterial infections in statin-treated patients., Statins are increasingly recognized as mediators of direct cellular effects independent of their lipid lowering capacity. Therefore, the time and concentration dependence of various statin-mediated cellular alterations was compared in renal mesangial cells. The effects of statins on cell proliferation, gene expression, cytoskeletal alterations, apoptosis, and cytotoxicity were analyzed in cultured mesangial cells using standard techniques. Results. Simvastatin and lovastatin decreased proliferation and cell number of rat mesangial cells concentration-dependently. Concurrently, the expression of the fibrogenic protein connective tissue growth factor (CTGF) was impaired and actin stress fibers, which are typical of mesangial cells in culture, became disassembled by simvastatin. A decrease of the posttranslational modification of RhoA by geranylgeranyl moieties was detected, supporting a role for RhoA as mediator of statin effects. Induction of apoptosis, determined by activation of caspase-3 and DNA fragmentation, and necrosis only occurred at later time points, when the morphology of the cells was strongly altered and the cells detached from the surface due to changes in the actin cytoskeleton. Basically, the same results were obtained with a human mesangial cell line. Furthermore, statin effects were mimicked by inhibition of the geranylgeranyltransferase. Most of the cellular effects of the lipophilic statins occurred within the same time and concentration range, suggesting a common molecular mechanism. Only apoptosis and necrosis were observed at later time points or with higher concentrations of simvastatin and thus seem to be secondary to the changes in gene expression and alterations of the actin cytoskeleton., For more Mechanism of Action (Complete) data for Simvastatin (6 total), please visit the HSDB record page. | |
| Record name | Simvastatin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00641 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Simvastatin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White to off-white crystalline powder from n-butyl chloride + hexane | |
CAS No. |
79902-63-9 | |
| Record name | Simvastatin | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=79902-63-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Simvastatin [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0079902639 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Simvastatin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00641 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | simvastatin | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758706 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Simvastatin | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0023581 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Butanoic acid, 2,2-dimethyl-, (1S,3R,7S,8S,8aR)-1,2,3,7,8,8a-hexahydro-3,7-dimethyl-8-[2-[(2R,4R)-tetrahydro-4-hydroxy-6-oxo-2H-pyran-2-yl]ethyl]-1-naphthalenyl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.115.749 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | (1S,3R,7S,8S,8aS)-1,2,3,7,8,8a-Hexahydro-3,7-dimethyl-8-{2-[(4R,6R)-tetrahydro-4-hydroxy-2-oxo-2H-pyran-6-yl]ethyl}-1-naphthyl-2,2-dimethylbutyrat | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | (1S,3R,7S,8S,8aR)-8-[2-[(2R,4R)-4-Hydroxy-6-oxotetrahydro-2H-pyran-2-yl]ethyl]-3,7-dimethyl-1,2,3,7,8,8a-hexahydronaphthalen-1-yl 2,2-dimethylbutanoate. | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | SIMVASTATIN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/AGG2FN16EV | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Simvastatin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Simvastatin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005007 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
135-138 °C, 135 - 138 °C | |
| Record name | Simvastatin | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00641 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Simvastatin | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7208 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Simvastatin | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0005007 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















