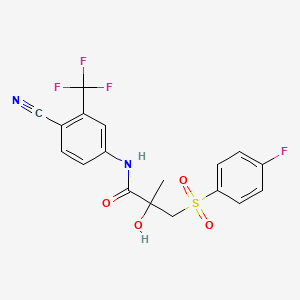
Bicalutamide
Overview
Description
Bicalutamide is a nonsteroidal antiandrogen medication primarily used in the treatment of prostate cancer. It is known for its ability to block the action of androgens, which are male hormones that stimulate the growth of prostate cancer cells . This compound is often used in combination with other treatments such as gonadotropin-releasing hormone analogues or surgical removal of the testicles to manage metastatic prostate cancer .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Bicalutamide involves several key stepsThis oxidation is typically carried out using potassium permanganate in the presence of water or a mixture of water and a water-miscible solvent . The resulting product is this compound.
Industrial Production Methods: Industrial production of this compound follows similar synthetic routes but on a larger scale. The process is designed to be simple, convenient, safe, and cost-effective. The key steps involve the preparation of the sulfide compound, followed by its oxidation to form this compound .
Chemical Reactions Analysis
Types of Reactions: Bicalutamide undergoes several types of chemical reactions, including:
Substitution: this compound can undergo substitution reactions, particularly involving the cyano and trifluoromethyl groups on the phenyl ring.
Common Reagents and Conditions:
Oxidizing Agents: Potassium permanganate, peroxy acids such as m-chloroperbenzoic acid.
Solvents: Water, methanol, methylene chloride.
Major Products Formed: The major product formed from the oxidation reaction is this compound itself, which is a sulfone compound .
Scientific Research Applications
Prostate Cancer Treatment
- Early and Advanced Prostate Cancer : Bicalutamide is approved for the treatment of stage D2 metastatic prostate cancer when used alongside surgical or medical castration. It is also administered as monotherapy for stage C or D1 locally advanced prostate cancer at a dosage of 150 mg/day .
- Survival Benefits : A study indicated that patients receiving this compound had a 76.3% overall survival rate at 12 years compared to 71.3% in the placebo group, highlighting its effectiveness in prolonging survival .
Hormonal Therapy
- Transgender Care : this compound is used in hormone therapy for transgender women to block testosterone effects and manage symptoms associated with high testosterone levels in women .
- Puberty Blocker : It is also employed to prevent premature puberty in transgender girls and treat early puberty in boys .
Other Indications
- This compound has shown potential in treating skin and hair conditions such as acne, seborrhea, hirsutism, and pattern hair loss in women .
- Research suggests its utility in managing paraphilias and hypersexuality in men .
Efficacy
- This compound has demonstrated comparable efficacy to castration therapies while preserving sexual interest better than traditional methods .
- Studies have shown that it can effectively inhibit androgen-mediated cell adhesion in prostate cancer cells, potentially reducing metastasis risk .
Side Effects
- Common side effects include breast pain and gynecomastia, which are typical for antiandrogens. However, this compound has a more favorable tolerability profile compared to other antiandrogens like flutamide .
- Long-term follow-up studies have indicated that while side effects exist, they are generally manageable and do not outweigh the benefits of treatment .
Case Studies
Mechanism of Action
Bicalutamide works by competitively inhibiting the binding of androgens such as dihydrotestosterone and testosterone to androgen receptors. This inhibition prevents the stimulation of cell growth in prostate cancer . This compound binds to the androgen receptor without activating gene expression, leading to the regression of prostatic tumors . The molecular targets involved include the androgen receptors in prostate cancer cells .
Comparison with Similar Compounds
- Flutamide
- Nilutamide
- Enzalutamide
- Apalutamide
- Cyproterone Acetate
- Spironolactone
Comparison:
- First-Generation Nonsteroidal Antiandrogens: Compared to flutamide and nilutamide, Bicalutamide has improved potency, efficacy, tolerability, and safety . It has largely replaced these medications in clinical practice.
- Second-Generation Nonsteroidal Antiandrogens: Compared to enzalutamide and apalutamide, this compound has inferior potency and efficacy but similar tolerability and safety .
- Steroidal Antiandrogens: Compared to cyproterone acetate and spironolactone, this compound has better selectivity in its action, superior efficacy as an antagonist of the androgen receptor, and better tolerability .
This compound’s unique properties, such as its selectivity and safety profile, make it a preferred choice in the treatment of prostate cancer .
Biological Activity
Bicalutamide is a nonsteroidal antiandrogen primarily used in the treatment of prostate cancer. Its biological activity is characterized by its ability to inhibit androgen receptors, thereby blocking the action of androgens like testosterone. This article provides a comprehensive overview of the biological activity of this compound, including its pharmacokinetics, mechanisms of action, clinical efficacy, and adverse effects.
Pharmacokinetics
This compound is administered orally, with a recommended dosage of 150 mg once daily for early nonmetastatic prostate cancer and 50 mg daily in combination with luteinizing hormone-releasing hormone analogues for advanced cases. Its pharmacokinetic profile reveals:
- Absorption : The (R)-enantiomer is absorbed slowly and saturably, while the (S)-enantiomer is more rapidly absorbed and cleared. Food intake does not significantly affect absorption.
- Elimination Half-life : Approximately one week, leading to a tenfold accumulation in plasma during daily administration.
- Metabolism : Primarily metabolized by cytochrome P450 enzymes, particularly CYP3A4. The (R)-enantiomer has potential inhibitory effects on CYP3A4 but does not show clinically relevant inhibition in vivo at standard doses .
This compound functions as a competitive antagonist at androgen receptors in target tissues such as the prostate. By binding to these receptors, it prevents androgens from exerting their biological effects, which is critical in managing androgen-dependent cancers.
Prostate Cancer Treatment
This compound has been extensively studied for its efficacy in treating prostate cancer. A notable study compared this compound monotherapy (50 mg/day) to castration. Results indicated that while this compound was associated with fewer adverse effects, it was inferior to castration regarding overall response rates and survival metrics:
- Time-to-Treatment Failure : Hazard ratio of 1.59 favoring castration.
- Prostate-Specific Antigen (PSA) Reduction : this compound showed a median fall of 86-88%, compared to 96-97% for castration .
Case Studies
- Phase II Trial in Breast Cancer : A multicenter trial assessed this compound's effectiveness in women with androgen receptor-positive metastatic breast cancer. The primary endpoint was the clinical benefit rate, which included complete responses and stable disease over six months. The results indicated potential benefits in this patient population, warranting further investigation .
- Weekly Administration Trial : A phase I-II trial evaluated the safety and activity of weekly this compound administration in men with elevated PSA levels. Results showed a significant reduction in PSA levels (50% reduction noted) and an improvement trend in high-grade prostatic intraepithelial neoplasia (HG-PIN) status among treated subjects .
Adverse Effects
While this compound is generally well-tolerated, it can lead to various side effects:
- Common Adverse Effects : Breast tenderness, gynecomastia, hot flushes, and liver enzyme alterations.
- Quality of Life Impact : Although some patients reported improved sexual functioning initially compared to those receiving castration, long-term studies indicate that higher doses may be needed to achieve comparable efficacy .
Comparative Efficacy Table
| Treatment Type | Objective Response Rate | Median Survival | PSA Reduction (%) |
|---|---|---|---|
| This compound (50 mg) | Lower than castration | Inferior | 86-88 |
| Castration | Higher | Superior | 96-97 |
Q & A
Basic Research Questions
Q. What experimental models are commonly used to investigate bicalutamide’s efficacy in androgen receptor (AR)-related pathologies, and how should researchers optimize dosing regimens?
- Methodological Guidance : Preclinical studies often employ knock-in (KI) mouse models, such as the AR113Q spinal and bulbar muscular atrophy (SBMA) model, to mimic human disease progression. This compound is administered subcutaneously (2 mg/kg twice weekly) in oil-based vehicles, with outcomes assessed via survival analysis (Kaplan-Meier curves) and motor behavior tests (rotarod, grip strength) . LC-MS is recommended for validating serum and tissue drug levels to ensure pharmacokinetic consistency .
Q. How can researchers design clinical trials to evaluate this compound’s impact on prostate cancer progression while minimizing bias?
- Methodological Guidance : Use the PICOT framework to structure trials:
- P opulation: Patients with locally advanced or metastatic prostate cancer (e.g., T1b-T4, M0).
- I ntervention: this compound 150 mg/day + standard care (radical prostatectomy, radiotherapy).
- C omparison: Placebo + standard care.
- O utcome: Progression-free survival (PFS), overall survival (OS).
- T ime: Median 5.4-year follow-up.
Stratify subgroups (e.g., localized vs. locally advanced disease) to address outcome discrepancies, as this compound benefits locally advanced patients but may harm those with localized disease .
Q. What analytical methods ensure reliable quantification of this compound in pharmaceutical formulations and biological samples?
- Methodological Guidance :
- Pharmaceutical purity : Use reverse-phase HPLC with UV detection (λ = 270 nm), validated per ICH guidelines. Ensure mobile phases include diluted phosphoric acid and acetonitrile for optimal resolution of related substances (e.g., isomers, degradation products) .
- Biological samples : LC-MS/MS for serum and tissue quantification, with calibration curves adjusted for matrix effects .
Advanced Research Questions
Q. How do AR variants (e.g., AR-V7) influence resistance to this compound, and what combinatorial strategies can overcome this?
- Methodological Guidance :
- Mechanistic studies : Use this compound-resistant cell lines (e.g., prostate cancer models) to quantify AR-V7 expression via qPCR/Western blot.
- Combinatorial therapy : Co-administer niclosamide (AR-V7 inhibitor) with this compound. Validate synergy using proliferation assays (Ki67 staining) and apoptosis markers (PARP cleavage) .
- Statistical analysis : Compare hazard ratios (HRs) for PFS between monotherapy and combination arms using Cox regression .
Q. What molecular pathways underlie this compound’s neuroprotective effects in SBMA, and how can autophagy modulation enhance therapeutic outcomes?
- Methodological Guidance :
- Autophagy analysis : Measure autophagic flux via LC3-II/p62 Western blot in skeletal muscle. This compound restores basal HSPB8 levels, resolving ARpolyQ aggregation. Combine with trehalose (autophagy inducer) to amplify efficacy .
- Mitochondrial markers : Quantify mtDNA copy number and OXPHOS enzyme accumulation to assess metabolic recovery .
Q. How should researchers resolve contradictions in survival outcomes from large-scale clinical trials (e.g., Early Prostate Cancer Program)?
- Methodological Guidance :
- Subgroup analysis : Stratify data by disease stage (localized vs. locally advanced). Use two-way ANOVA with Tukey’s post-hoc test to identify treatment effects within subgroups .
- Bias mitigation : Adjust for confounding variables (e.g., baseline PSA, Gleason score) using multivariate regression. Report HRs with 95% confidence intervals .
Q. What biomarkers predict this compound’s clinical benefit in AR-negative breast cancer, and how can they be validated?
- Methodological Guidance :
- Retrospective biomarker screening : Analyze archived tumor samples for AR splice variants or immune checkpoint markers (e.g., PD-L1) via immunohistochemistry.
- Validation cohort : Use a prospective trial design with pre-specified endpoints (e.g., 6-month clinical benefit rate). Apply log-rank tests to compare PFS between biomarker-positive and -negative cohorts .
Q. Methodological Best Practices
-
Experimental Design :
-
Data Analysis :
-
Analytical Validation :
Properties
IUPAC Name |
N-[4-cyano-3-(trifluoromethyl)phenyl]-3-(4-fluorophenyl)sulfonyl-2-hydroxy-2-methylpropanamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C18H14F4N2O4S/c1-17(26,10-29(27,28)14-6-3-12(19)4-7-14)16(25)24-13-5-2-11(9-23)15(8-13)18(20,21)22/h2-8,26H,10H2,1H3,(H,24,25) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
LKJPYSCBVHEWIU-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(CS(=O)(=O)C1=CC=C(C=C1)F)(C(=O)NC2=CC(=C(C=C2)C#N)C(F)(F)F)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C18H14F4N2O4S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID2022678 | |
| Record name | Bicalutamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022678 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
430.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Bicalutamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015260 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Slightly soluble in chloroform and absolute ethanol; sparingly soluble in methanol; soluble in acetone and tetrahydrofuan, Practically insoluble in water at 37 °C (5 mg/1000 mL), 9.28e-03 g/L | |
| Record name | Bicalutamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01128 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BICALUTAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7655 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bicalutamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015260 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Bicalutamide competes with androgen for the binding of androgen receptors, consequently blocking the action of androgens of adrenal and testicular origin which stimulate the growth of normal and malignant prostatic tissue., Bicalutamide is a nonsteroidal antiandrogen that is structurally and pharmacologically related to flutamide and nilutamide. Bicalutamide inhibits the action of androgens by competitively blocking nuclear androgen receptors in target tissues such as the prostate, seminal vesicles, and adrenal cortex; blockade of androgen receptors in the hormone-sensitive tumor cells may result in growth arrest or transient tumor regression through inhibition of androgen-dependent DNA and protein synthesis. Bicalutamide is a selective antiandrogen with no androgenic or progestational activity in various animal models. The relative binding affinity of bicalutamide at the androgen receptor is more than that of nilutamide and approximately 4 times that of hydroxyflutamide, the active metabolite of flutamide., Common pharmacologic therapies for prostate cancer (ie, gonadotropin-releasing hormone [GnRH] analogs, nonsteroidal antiandrogens) when used as monotherapy initially result in increased serum testosterone concentrations, which may limit the effects of the drugs. Androgen receptors in the hypothalamus are blocked by bicalutamide, which disrupts the inhibitory feedback of testosterone on luteinizing hormone (LH) release, resulting in a temporary increase in secretion of LH; the increase in LH stimulates an increase in the production of testosterone. As GnRH analogs have potent GnRH agonist properties, testicular steroidogenesis continues during the first few weeks after initiating therapy. However, the combination of orchiectomy or GnRH analog therapy to suppress testicular androgen production and an antiandrogen to block response of remaining adrenal androgens provides maximal androgen blockade. Concomitant administration of antiandrogens such as bicalutamide in patients initiating therapy with a GnRH analog can inhibit initial androgenic stimulation and potential exacerbation of symptoms (e.g., bone pain, urinary obstruction, liver pain, impending spinal cord compression) that may occur during the first month of GnRH analog therapy., Bicalutamide was developed from a series of nonsteroidal compounds related to flutamide that showed a range of pharmacologic activity from full androgen agonist to pure antiandrogen, including progestational and antiprogestational properties. Bicalutamide is a pure antiandrogen that binds to rat, dog, and human prostate; the affinity compared with the natural ligand 5 alpha-dihydrotestosterone is low, but bicalutamide has an affinity for the rat androgen receptor approximately four times higher than hydroxyflutamide, the active metabolite of flutamide. Bicalutamide also binds to androgen receptors found in the LNCaP human prostate tumor and the Shionogi S115 mouse mammary tumor cell line, as well as androgen receptors transfected into CV-1 and HeLa cells. In all cases, bicalutamide behaves as a pure antiandrogen and inhibits gene expression and cell growth stimulated by androgen. Studies with the LNCaP cell line are particularly interesting, as these cells contain a mutated androgen receptor (codon 868, Thr-->Ala), which behaves idiosyncratically with other antiandrogens (cyproterone acetate and flutamide): both these antiandrogens act as agonists in this cell line and stimulate proliferation. Studies in vivo show that bicalutamide is a potent antiandrogen in the rat. In immature, castrated male rats treated daily with testosterone propionate, bicalutamide produces a profound inhibition of accessory sex organ (ventral prostate and seminal vesicles) growth at oral doses as low as 0.25 mg/kg; it is more active in this test than flutamide or cyproterone acetate. In mature male rats, daily oral doses of bicalutamide produce a dose-related reduction in weights of the ventral prostate glands and seminal vesicles: in this test, bicalutamide is around five times as potent as flutamide. In contrast to flutamide, which produces dose-related, marked increases in serum luteinizing hormone (LH) and testosterone as a consequence of the central inhibition of the negative feedback effects of androgens on the hypothalamic-pituitary-tests axis, bicalutamide has little effect on serum LH and testosterone; i.e., it is peripherally selective. The peripheral selectivity of bicalutamide in the rat is not due to differences between the prostate versus hypothalamic or pituitary receptors, as bicalutamide reverses the suppressive effect of testosterone on luteinizing hormone-releasing hormone (LHRH) secretion from hypothalamic slices in vitro and is as effective as flutamide at sensitizing the pituitary gland to secrete LH in response to administered LHRH. The peripheral selectivity of bicalutamide has now been shown to be due to poor penetration across the blood-brain barrier: tissue distribution studies with [3H]bicalutamide show that although it is concentrated in the organs of metabolism and secretion as well as in the prostate, the pituitary glands, and the seminal vesicles, levels in the hypothalamus and the central nervous system (CNS) are much lower than in blood. Indeed, it is probable that levels found in the CNS reflect levels of blood contamination. In dogs, bicalutamide has exquisite potency and causes dose-related atrophy of the prostate gland and epididymides; with an oral ED50 of 0.1 mg/kg, it is around 50 times as potent as flutamide in this species and also more potent than the steroidal antiandrogen WIN49596 and the 5 alpha-reductase inhibitor MK-906. Even at substantial multiples of the active dose (up to 100 mg/kg orally), bicalutamide failed to increase serum testosterone, so it is also peripherally selective in the dog., Although widely accepted as an androgen receptor antagonist, the mechanism by which it induces apoptosis remains unclear. Defining exact pathways by which bicalutamide induces its apoptotic effects would help to advance its clinical applications. /Investigators/ aimed to examine the apoptotic effects of bicalutamide at 24 hr and comment on the role of the caspases and calpains in mediating bicalutamide-induced apoptosis in androgen-dependent and androgen-independent cells. PWR-1E, PC-3 and DU-145 cells were treated with bicalutamide and assessed for apoptosis by flow cytometry at 24 hr. DU-145 cells were used to compare differences between two different metastatic receptor-negative cells and to verify apoptotic induction at 48 hr. To delineate a specific pathway of action for bicalutamide, PC-3 and PWR-1E cells were pretreated with specific inhibitors of caspase-dependent (zVAD-FMK) and caspase-independent pathways (calpain 2 inhibitor). Bicalutamide induced apoptosis in androgen-dependent PWR-1E cells via a caspase-dependent and calpain-independent mechanism. In androgen-independent PC-3 cells, bicalutamide also induced apoptosis by mechanisms that were partially inhibited by pan-caspase inhibition but were partially calpain dependent. Understanding into how bicalutamide exerts its effects in androgen-independent cells will yield further insights into the treatment of hormone-refractory disease. | |
| Record name | Bicalutamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01128 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BICALUTAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7655 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals from 1:1 (v/v) mix of ethyl acetate and petroleum ether, Fine white to off-white powder | |
CAS No. |
90357-06-5 | |
| Record name | Bicalutamide | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=90357-06-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Bicalutamide [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0090357065 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Bicalutamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01128 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | bicalutamide | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759816 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Bicalutamide | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID2022678 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Propanamide, N-[4-cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfonyl]-2-hydroxy-2-methyl | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.126.100 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | BICALUTAMIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/A0Z3NAU9DP | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | BICALUTAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7655 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bicalutamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015260 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
191-193 °C, 191 - 193 °C | |
| Record name | Bicalutamide | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01128 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | BICALUTAMIDE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7655 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Bicalutamide | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015260 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods I
Procedure details








Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details







Synthesis routes and methods IV
Procedure details









Synthesis routes and methods V
Procedure details









Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













