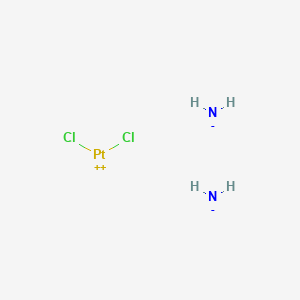
Azanide; dichloroplatinum(2+)
- Click on QUICK INQUIRY to receive a quote from our team of experts.
- With the quality product at a COMPETITIVE price, you can focus more on your research.
Overview
Description
Preparation Methods
Azanide; dichloroplatinum(2+) is synthesized through a series of chemical reactions involving platinum compounds. The most common synthetic route involves the reaction of potassium tetrachloroplatinate(II) with ammonia to form cis-diamminedichloroplatinum(II). The reaction conditions typically include an aqueous medium and controlled temperature to ensure the formation of the desired cis isomer . Industrial production methods often involve large-scale synthesis in controlled environments to maintain the purity and efficacy of the compound .
Chemical Reactions Analysis
Azanide; dichloroplatinum(2+) undergoes several types of chemical reactions, including hydrolysis, substitution, and reduction. One of the key reactions is hydrolysis, where the chloride ligands are replaced by water molecules, forming aquated species such as cis-[PtCl(NH3)2(H2O)]+ . These aquated species are highly reactive and can further react with nucleophiles, leading to the formation of DNA adducts, which are crucial for its anticancer activity . Common reagents used in these reactions include water, ammonia, and various nucleophiles . The major products formed from these reactions are DNA adducts and other platinum complexes .
Scientific Research Applications
Azanide; dichloroplatinum(2+) has a wide range of scientific research applications, particularly in the fields of chemistry, biology, medicine, and industry. In chemistry, it is used as a model compound to study the coordination chemistry of platinum complexes . In biology, cisplatin is used to investigate the mechanisms of DNA damage and repair . In medicine, it is a critical component of chemotherapy regimens for various cancers . Additionally, cisplatin is used in industrial applications, such as the development of new drug delivery systems and the study of drug resistance mechanisms .
Mechanism of Action
The mechanism of action of cisplatin involves several key steps: cellular uptake, activation by aquation, DNA binding, and the processing of DNA lesions . Once inside the cell, cisplatin undergoes aquation, where the chloride ligands are replaced by water molecules, forming reactive aquated species . These species then bind to DNA, forming intrastrand and interstrand cross-links that disrupt DNA replication and transcription . This leads to the activation of various cellular pathways, including apoptosis, ultimately resulting in cancer cell death .
Comparison with Similar Compounds
Azanide; dichloroplatinum(2+) is part of a class of platinum-based chemotherapy drugs, which also includes carboplatin and oxaliplatin . While all three compounds share a similar mechanism of action, they differ in their chemical structures and side effect profiles . Carboplatin, for example, has a bidentate dicarboxylate ligand instead of the chloride ligands found in cisplatin, which results in a different toxicity profile and reduced side effects . Oxaliplatin, on the other hand, has a diaminocyclohexane ligand, which makes it effective against some cisplatin-resistant cancers . Other similar compounds include nedaplatin, lobaplatin, and heptaplatin, each with unique properties and clinical applications .
Q & A
Basic Research Questions
Q. What are the key physicochemical properties of azanide; dichloroplatinum(2+) relevant to experimental design?
Methodological Answer: Azanide; dichloroplatinum(2+) (Cisplatin, CDDP) is characterized by:
- Molecular formula : Cl₂H₆N₂Pt (molecular weight: 300.05 g/mol) .
- Solubility : >12.5 mg/mL in DMF (dimethylformamide); insoluble in water and ethanol. Solutions are unstable and must be prepared fresh .
- Storage : Powder should be stored at room temperature (RT) in the dark. Solutions degrade rapidly and may precipitate at low temperatures .
Experimental Implications :
- Use DMF for dissolution; avoid DMSO, which deactivates the compound .
- Conduct kinetic studies within 24 hours of solution preparation to avoid decomposition artifacts.
Table 1: Key Physicochemical Properties
| Property | Value/Description | Reference |
|---|---|---|
| Molecular Weight | 300.05 g/mol | |
| Solubility (DMF) | >12.5 mg/mL | |
| Stability (solution) | Highly unstable; prepare fresh |
Q. How can researchers synthesize azanide; dichloroplatinum(2+) complexes with controlled isomer ratios?
Methodological Answer: The synthesis of dichloroplatinum(II) complexes often yields mixtures of cis and trans isomers. To control isomer ratios:
- Kinetic Control : React [PtCl₃(NCMe)]⁻ with acetonitrile. The cis isomer forms preferentially due to the stronger trans-labilizing effect of Cl⁻ compared to acetonitrile .
- Thermodynamic Control : Adjust reaction temperature and solvent polarity. Higher temperatures favor the trans isomer in equilibrium conditions .
Experimental Validation :
- Use infrared (IR) and ¹H NMR spectroscopy to confirm isomer purity .
- For antitumor studies, isolate the cis isomer via recrystallization in non-polar solvents.
Advanced Research Questions
Q. How do ligand substitutions in dichloroplatinum(II) complexes influence antitumor activity and water solubility?
Methodological Answer: Substitutions in the ligand framework directly impact:
- Antitumor Activity : Hydroxyl (-OH) groups in the 2-position of aromatic ligands (e.g., [1,2-bis(2-hydroxyphenyl)ethylenediamine]) enhance activity. In murine leukemia models, such complexes achieved 67% cure rates (4/6 animals) at 13.2 µmol/kg doses .
- Water Solubility : Polar substituents (e.g., -OH, -NH₂) increase solubility, improving bioavailability. For example, substituting hydrophobic phenyl groups with hydroxylated analogs increases solubility by >50% .
Table 2: Impact of Ligand Substitutions
| Ligand Modification | Antitumor Efficacy (P388 Leukemia) | Solubility Change | Reference |
|---|---|---|---|
| 2-hydroxyphenyl substitution | 67% cure rate | +40% | |
| 4-hydroxyphenyl substitution | Moderate activity | +20% |
Q. What analytical techniques are suitable for characterizing dichloroplatinum(II) complexes during solid-phase synthesis?
Methodological Answer:
- Gel-Phase ¹⁹⁵Pt NMR : Enables non-destructive analysis of resin-bound complexes. Chemical shifts correlate with solution-state spectra (e.g., δ = -2100 ppm for primary amine complexes) .
- DNA Binding Assays : Use gel electrophoresis to detect 1,2-intrastrand cross-links with oligonucleotides (e.g., d(GpG) sequences), mimicking cisplatin’s mechanism .
Experimental Workflow :
Synthesize complexes on solid support (e.g., octreotide analogs).
Perform on-resin ¹⁹⁵Pt NMR to confirm coordination geometry .
Cleave from resin and validate DNA binding via UV-vis spectroscopy or EMSA (Electrophoretic Mobility Shift Assay) .
Q. How can researchers address contradictions in cytotoxicity data between in vitro and in vivo models for dichloroplatinum(II) complexes?
Methodological Answer: Discrepancies often arise from:
- Activation Kinetics : Slow Cl⁻ → H₂O ligand exchange in vivo reduces bioavailability. Use AgNO₃ to accelerate activation in vitro, but validate with physiological chloride concentrations (e.g., 100 mM NaCl) .
- Endoplasmic Reticulum (ER) Stress : Co-treatment with O⁶-benzylguanine enhances cytotoxicity by prolonging ER stress, a mechanism less pronounced in vitro. Monitor CHOP (C/EBP homologous protein) expression as a stress marker .
Validation Strategy :
- Compare IC₅₀ values in cell lines (e.g., A549) with in vivo tumor regression rates in xenograft models.
- Use siRNA knockdown of ER stress pathways to isolate mechanistic contributions .
Q. What strategies optimize the Cl⁻/H₂O ligand exchange kinetics to improve therapeutic efficacy?
Methodological Answer:
- Ligand Design : Incorporate electron-withdrawing groups (e.g., -F, -NO₂) to labilize Cl⁻ ligands. For example, fluorophenyl-substituted complexes show 2× faster activation rates .
- Coordination Environment : Use bidentate ligands (e.g., ethylenediamine) to stabilize the active aquated species.
Kinetic Analysis :
- Conduct stopped-flow UV-vis spectroscopy to measure exchange rates (k = 10⁻³–10⁻² s⁻¹) .
- Compare with DFT calculations to predict labilization effects .
Q. How do researchers resolve discrepancies in isomer-dependent biological activity?
Methodological Answer:
- Isomer-Specific Assays : Test cis and trans isomers separately in cytotoxicity screens (e.g., NCI-60 panel). The cis isomer typically shows 10–100× higher activity due to DNA adduct formation .
- Structural Studies : Use X-ray crystallography to compare DNA adduct geometries. Cis isomers form stable 1,2-intrastrand cross-links, while trans isomers produce less stable monofunctional adducts .
Contradiction Mitigation :
Properties
Key on ui mechanism of action |
Alkylating agents work by three different mechanisms: 1) attachment of alkyl groups to DNA bases, resulting in the DNA being fragmented by repair enzymes in their attempts to replace the alkylated bases, preventing DNA synthesis and RNA transcription from the affected DNA, 2) DNA damage via the formation of cross-links (bonds between atoms in the DNA) which prevents DNA from being separated for synthesis or transcription, and 3) the induction of mispairing of the nucleotides leading to mutations. |
|---|---|
CAS No. |
15663-27-1 |
Molecular Formula |
Cl2H6N2Pt |
Molecular Weight |
300.05 g/mol |
IUPAC Name |
azane;dichloroplatinum |
InChI |
InChI=1S/2ClH.2H3N.Pt/h2*1H;2*1H3;/q;;;;+2/p-2 |
InChI Key |
LXZZYRPGZAFOLE-UHFFFAOYSA-L |
SMILES |
[NH2-].[NH2-].Cl[Pt+2]Cl |
Canonical SMILES |
N.N.Cl[Pt]Cl |
Appearance |
Yellow solid powder |
melting_point |
270 dec °C |
Key on ui other cas no. |
14283-03-5 |
physical_description |
Yellow crystalline solid; [MSDSonline] |
Pictograms |
Corrosive; Irritant; Health Hazard |
Purity |
>98% (or refer to the Certificate of Analysis) |
shelf_life |
>2 years if stored properly |
solubility |
H2O 1 (mg/mL) DMSO 10 (mg/mL) 10:1 PVP coprecipitate in 10% PVP 2.5 (mg/mL) Dimethylformamide (pure anhydrous) 24 (mg/mL) |
storage |
Dry, dark and at 0 - 4 C for short term (days to weeks) or -20 C for long term (months to years). |
Synonyms |
CACP; cisDDP; cisdiamminedichloro platinum (II); cisdiamminedichloroplatinum; Cisdichloroammine Platinum (II); Cismaplat; Cisplatina; cisplatinous diamine dichloride; cisplatinum; cisplatinum II; cisplatinum II diamine dichloride; CPDD; Cysplatyna; DDP; PDD; Peyrones Chloride; Peyrones Salt; Platinoxan; platinum diamminodichloride; US brand names: Platinol; PlatinolAQ; Foreign brand names: Abiplatin; Blastolem; Briplatin; Cisplatyl; Citoplatino; Citosin; Lederplatin; Metaplatin; Neoplatin; Placis; Platamine; Platiblastin; PlatiblastinS; Platinex; Platinol AQ; PlatinolAQ VHA Plus; Platiran; Platistin; Platosin; Abbreviations: CDDP; DDP. |
Origin of Product |
United States |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















