Selumetinib
Overview
Description
Selumetinib, sold under the brand name Koselugo, is a medication primarily used for the treatment of neurofibromatosis type 1 (NF-1) in children aged two years and older. NF-1 is a genetic disorder of the nervous system that causes tumors to grow on nerves . This compound is a selective inhibitor of mitogen-activated protein kinase 1 and 2 (MEK1/2), which are key components of the Raf-MEK-ERK signaling pathway .
Preparation Methods
Selumetinib is synthesized through a multi-step chemical processThe final step involves the formation of the carboxamide group . Industrial production methods typically involve optimizing reaction conditions to maximize yield and purity while minimizing the formation of impurities.
Chemical Reactions Analysis
Selumetinib undergoes several types of chemical reactions, including:
Oxidation: This compound is sensitive to oxidation, leading to the formation of an amide derivative.
Photooxidation: Exposure to light can cause photooxidation, resulting in the formation of an ester derivative.
Hydrolysis: This compound can undergo hydrolysis under acidic or basic conditions, leading to the degradation of the compound.
Common reagents and conditions used in these reactions include oxidizing agents, light exposure, and acidic or basic solutions. The major products formed from these reactions are amide and ester derivatives.
Scientific Research Applications
Selumetinib has a wide range of scientific research applications, including:
Chemistry: Used as a tool to study the Raf-MEK-ERK signaling pathway and its role in various cellular processes.
Biology: Investigated for its effects on cell growth, differentiation, and apoptosis in various cell lines.
Industry: Utilized in the development of targeted therapies for cancer treatment.
Mechanism of Action
Selumetinib exerts its effects by selectively inhibiting MEK1 and MEK2, which are key components of the Raf-MEK-ERK signaling pathway . By inhibiting these proteins, this compound blocks the downstream signaling that promotes cell proliferation and survival. This inhibition leads to the arrest of the cell cycle in the G1-S phase and induces apoptosis in cancer cells .
Comparison with Similar Compounds
Selumetinib is compared with other MEK inhibitors such as trametinib and cobimetinib. While all three compounds target the MEK1/2 proteins, this compound is unique in its chemical structure and pharmacokinetic properties . For example, trametinib and cobimetinib have different molecular structures and may exhibit different efficacy and safety profiles in clinical settings .
Similar Compounds
- Trametinib
- Cobimetinib
- Binimetinib
This compound’s uniqueness lies in its specific binding affinity and selectivity for MEK1/2, which contributes to its effectiveness in treating neurofibromatosis type 1 and other tumors .
Biological Activity
Selumetinib, an oral selective inhibitor of mitogen-activated protein kinase 1 and 2 (MEK1/2), has emerged as a promising therapeutic agent in oncology. It has been particularly noted for its efficacy in treating various solid tumors, including neurofibromatosis type 1 (NF1) associated plexiform neurofibromas, low-grade gliomas, and certain types of lung and colorectal cancers. This article provides a detailed examination of the biological activity of this compound, including its pharmacokinetics, mechanisms of action, clinical efficacy, and case studies.
This compound exerts its effects by specifically inhibiting MEK1/2, which are key components of the MAPK signaling pathway. This pathway is crucial for regulating cell proliferation, survival, and differentiation. By binding to an allosteric site on MEK1/2, this compound prevents the phosphorylation of ERK1/2, thereby disrupting downstream signaling that promotes tumor growth.
Key Mechanisms:
- Inhibition of ERK Phosphorylation: this compound has demonstrated an IC50 value of approximately 14.1 nM for inhibiting MEK1/2 activity .
- Cell Cycle Arrest: It induces G1-S phase cell cycle arrest and promotes apoptosis in cancer cells .
- Mutational Dependency: The efficacy of this compound is enhanced in tumors with activating mutations in B-Raf or Ras genes .
Pharmacokinetics
This compound displays favorable pharmacokinetic properties:
- Absorption: Rapid absorption with peak plasma concentrations reached within 4 hours post-administration.
- Half-life: The mean terminal elimination half-life is approximately 6.2 to 7.5 hours .
- Steady State: Achieved within 1-2 days with minimal accumulation noted .
Table 1: Pharmacokinetic Parameters of this compound
| Parameter | Value |
|---|---|
| Peak Plasma Concentration | ~4 hours post-dose |
| Half-life | 6.2 - 7.5 hours |
| Steady State | 1 - 2 days |
| Bioavailability | High (exact percentage not specified) |
Clinical Efficacy
This compound has shown promising results across various clinical trials:
Case Studies
- Neurofibromatosis Type 1 : In pediatric patients with NF1-associated plexiform neurofibromas, this compound has been approved due to its significant tumor response rates.
- Pancreatic Cancer : A Phase II study indicated that patients with KRAS G12R mutations showed a better response to this compound compared to those with other KRAS mutations .
- Low-Grade Gliomas : this compound demonstrated efficacy as a monotherapy or in combination with chemotherapy in treating pediatric low-grade gliomas .
Table 2: Summary of Clinical Trials Involving this compound
| Study Type | Population | Outcome |
|---|---|---|
| Phase II (NF1) | Pediatric patients | Significant reduction in tumor size |
| Phase II (Pancreatic Cancer) | KRAS G12R mutation patients | Improved overall response rates |
| Combination Therapy (Gliomas) | Pediatric low-grade gliomas | Enhanced efficacy compared to standard therapy |
Safety Profile
The safety profile of this compound is generally favorable, though it can lead to adverse effects such as gastrointestinal disturbances, rash, and fatigue. Close monitoring is recommended during treatment.
Summary of Adverse Effects
- Common : Rash, diarrhea, nausea.
- Serious : Liver enzyme elevations, cardiac issues in specific populations.
Q & A
Basic Research Questions
Q. What are the primary mechanisms of action of selumetinib, and how do they influence experimental design in preclinical studies?
this compound is a selective, non-ATP competitive inhibitor of MEK1/2, targeting the MAPK/ERK pathway, which is frequently dysregulated in cancers with RAS/RAF mutations. Preclinical studies should incorporate cell lines or animal models with confirmed mutations in KRAS, NRAS, or BRAF to validate pathway inhibition. Methodologically, dose-response assays (e.g., IC50 determination) and downstream phosphorylation analysis (e.g., ERK1/2 via Western blot) are critical for establishing mechanistic proof-of-concept .
Q. What standardized pharmacokinetic (PK) protocols should be followed when administering this compound in clinical trials?
this compound exhibits dose-dependent exposure with a recommended dose of 75 mg twice daily (BID) in adults. PK studies must account for food effects: while low-fat meals reduce AUC by 22.5% and high-fat meals by 20.8%, these changes are not clinically significant due to the flat exposure-response relationship at 20–30 mg/m². Trials should standardize fasting conditions or document meal composition to ensure reproducibility .
Q. How should researchers address ethical considerations when designing this compound trials involving human subjects?
Protocols must include informed consent processes, documentation of Institutional Review Board (IRB) approval, and adherence to Declaration of Helsinki principles. For trials in vulnerable populations (e.g., pediatric neurofibromatosis), additional safeguards like assent forms and long-term toxicity monitoring are required. Adverse event reporting should follow CONSORT guidelines, emphasizing grade ≥3 toxicities (e.g., rash, diarrhea) .
Advanced Research Questions
Q. How can contradictory efficacy outcomes in this compound trials (e.g., NSCLC vs. uveal melanoma) be analyzed methodologically?
The SELECT-1 trial (NSCLC) showed no progression-free survival (PFS) benefit for this compound + docetaxel vs. docetaxel alone (HR 0.93, P = 0.44), whereas the phase II uveal melanoma trial demonstrated improved PFS (HR 0.46, P < 0.001). Researchers should conduct meta-analyses adjusting for confounding variables:
- Tumor mutational heterogeneity (e.g., GNAQ mutations in uveal melanoma vs. KRAS in NSCLC) .
- Differential MEK dependency across cancer types . Subgroup analyses using stratified randomization or Bayesian adaptive designs can refine patient selection criteria .
Q. What statistical approaches are optimal for analyzing survival endpoints in this compound trials with high censoring rates?
For trials with >50% censored data (e.g., overall survival in SELECT-1), Cox proportional hazards models with time-dependent covariates are preferred. Sensitivity analyses (e.g., inverse probability weighting) should address informative censoring. For PFS, RECIST 1.1 criteria must be applied consistently, with independent radiological review to reduce bias .
Q. How can pharmacogenomic data be integrated into this compound trial design to optimize predictive biomarker discovery?
Retrospective genomic profiling (e.g., BRAF V600E status in thyroid cancer) should be paired with prospective validation. In the phase II papillary thyroid carcinoma trial, BRAF-mutant tumors had longer median PFS (33 vs. 11 weeks, P = 0.3), suggesting enrichment strategies. Adaptive trial designs (e.g., basket trials) can evaluate this compound’s efficacy across multiple biomarker-defined cohorts .
Q. What methodologies resolve discrepancies between preclinical and clinical efficacy data for this compound?
Preclinical models often overestimate efficacy due to lack of tumor microenvironment complexity. To bridge this gap:
- Use patient-derived xenografts (PDXs) with intact stromal components.
- Apply dynamic biomarkers (e.g., circulating tumor DNA) to monitor clonal evolution during treatment .
- Validate findings in ex vivo organoid cultures .
Q. How should researchers design combination therapy trials to overcome this compound resistance mechanisms?
Resistance often arises from PI3K/AKT pathway activation or feedback loops. Rational combinations include:
- PI3K inhibitors : Preclinical synergy shown in KRAS-mutant models.
- CDK4/6 inhibitors : To counteract cell-cycle re-entry post-MEK inhibition. Phase I trials should use dose-escalation designs (e.g., 3+3) with pharmacodynamic endpoints (e.g., phospho-RB suppression) .
Q. Methodological Resources
- Data Synthesis : Follow PRISMA guidelines for systematic reviews of this compound’s efficacy across indications .
- PK/PD Modeling : Use nonlinear mixed-effects modeling (e.g., NONMEM) to characterize exposure-response relationships .
- Ethical Compliance : Reference NIH guidelines for adverse event reporting and data safety monitoring boards (DSMBs) .
Properties
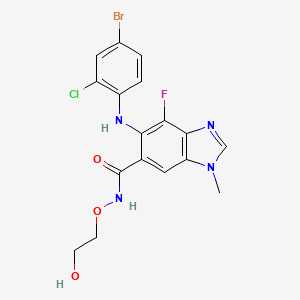
IUPAC Name |
6-(4-bromo-2-chloroanilino)-7-fluoro-N-(2-hydroxyethoxy)-3-methylbenzimidazole-5-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C17H15BrClFN4O3/c1-24-8-21-16-13(24)7-10(17(26)23-27-5-4-25)15(14(16)20)22-12-3-2-9(18)6-11(12)19/h2-3,6-8,22,25H,4-5H2,1H3,(H,23,26) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CYOHGALHFOKKQC-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CN1C=NC2=C1C=C(C(=C2F)NC3=C(C=C(C=C3)Br)Cl)C(=O)NOCCO | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C17H15BrClFN4O3 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID3048944 | |
| Record name | Selumetinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3048944 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
457.7 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Mechanism of Action |
The Ras-Raf-MEK-ERK signaling cascade is known to be activated in several types of cancer, and regulates the transcription of proteins involved in apoptosis. In addition, studies have shown that mutations of the Raf component of the pathway can contribute to chemotherapy drug resistance. Ras as well as several kinases and phosphatases are responsible for regulating the Raf-MEK-ERK pathway. Often in cancers, Ras (a G-protein coupled receptor) is deregulated, allowing downstream signalling to proceed unchecked. Through several complex steps, Raf phosphorylates and activates MEK, which then phosphorylates and activates ERK. ERK is then able to exert its effects on several downstream targets. As such, therapies inhibiting upstream components of this pathway have become attractive targets for cancer treatment. Selumetinib exerts its effects by selectively inhibiting MEK1 and MEK2 which can effectively blunt the pleiotropic effects of the Ras-Raf-MEK-ERK cascade. By inhibiting this oncogenic pathway, selumetinib reduces cell proliferation, and promotes pro-apoptotic signal transduction. | |
| Record name | Selumetinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11689 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
606143-52-6 | |
| Record name | Selumetinib | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=606143-52-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Selumetinib [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0606143526 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Selumetinib | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB11689 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Selumetinib | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID3048944 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 5-[(4-bromo-2-chlorophenyl)amino]-4-fluoro-N-(2-hydroxyethoxy)-1-methyl-1H-1,3-benzodiazole-6-carboxamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | SELUMETINIB | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/6UH91I579U | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













