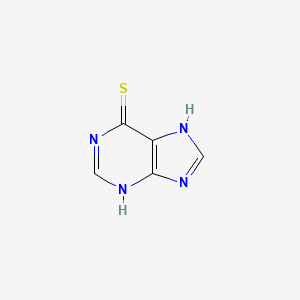
6-Mercaptopurine
Overview
Description
Preparation Methods
Synthetic Routes and Reaction Conditions: Mercaptopurine can be synthesized through several methods. One common method involves the reaction of 4-amino-5-imidazolecarboxamide with thiourea under acidic conditions to yield 6-mercaptopurine . Another method involves the cyclization of 5-amino-1H-imidazole-4-carboxamide with carbon disulfide and subsequent reduction .
Industrial Production Methods: In industrial settings, mercaptopurine is produced through a series of chemical reactions involving the condensation of 4-amino-5-imidazolecarboxamide with thiourea, followed by cyclization and purification steps . The process is optimized to ensure high yield and purity of the final product.
Chemical Reactions Analysis
Types of Reactions: Mercaptopurine undergoes various chemical reactions, including oxidation, reduction, and substitution reactions .
Common Reagents and Conditions:
Reduction: Reduction of mercaptopurine can be achieved using reducing agents like sodium borohydride.
Substitution: Mercaptopurine can undergo nucleophilic substitution reactions with halogenated compounds to form various derivatives.
Major Products:
Oxidation: 6-Thioxanthine
Reduction: Reduced mercaptopurine derivatives
Substitution: Various substituted mercaptopurine derivatives
Scientific Research Applications
6-Mercaptopurine (6-MP) is a medication primarily used as a first-line treatment for leukemia, demonstrating significant effects against the disease . It is also used in the treatment of inflammatory bowel disease . However, 6-MP has limitations, including poor water solubility, a tendency to bind with serum proteins, a short circulation time, and considerable toxic side effects, which restrict its application .
Scientific Research Applications
To counter these limitations, researchers have developed 6-MP nanomedicines that aim to improve the drug's water solubility, extend its circulation time, increase bioavailability, and reduce toxicity . Approaches include polymer prodrugs and drug-loaded vesicles designed for targeted delivery .
One strategy involves a hyaluronic acid (HA)-based, glutathione-responsive 6-MP polymer prodrug (HA-GS-MP) for targeted therapy of acute myeloid leukemia. HA-GS-MP is designed for efficient targeted delivery and treatment of 6-MP, using hyaluronic acid to target malignant tumor cells that overexpress the CD44 receptor . The 6-MP is connected to the HA chain through a vinyl sulfide bond, which remains stable under physiological conditions, preventing drug release until it reaches the intracellular reducing environment where the bond breaks and 6-MP is released .
Pharmacokinetics and Pharmacogenetics
Population pharmacokinetic models have been developed to understand the behavior of 6-MP active metabolites in pediatric patients with acute lymphoblastic leukemia (ALL) . These models help assess demographic and genetically controlled factors that lead to variability in how individuals respond to the drug .
Key findings include:
- Significant interindividual variability in the clearance of 6-MP active metabolites, such as 6-thioguanine nucleotides (6-TGNs) and 6-methylmercaptopurine nucleotides (6-mMPNs) .
- Body surface area and thiopurine methyltransferase (TPMT) genotype are influential covariates affecting the fractional metabolic transformation of 6-MP into 6-TGNs .
- The developed pharmacokinetic model offers a more rational dosing approach for 6-MP than the traditional empirical method by combining body surface area with pharmacogenetically guided dosing based on TPMT genotype .
The cytotoxic effects of 6-MP are mainly achieved through the incorporation of 6-TGNs into the DNA of leukocytes because of their structural similarity to guanine. Additionally, 6-mMPNs are strong inhibitors of purine de novo synthesis, a well-established protocol to achieve immunosuppression .
Essential Medicines
Mechanism of Action
Mercaptopurine exerts its effects by competing with hypoxanthine and guanine for the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRTase). It is converted to thioinosinic acid, which inhibits several reactions involving inosinic acid, such as the conversion to xanthylic acid and adenylic acid . This inhibition disrupts DNA and RNA synthesis, leading to cell death .
Comparison with Similar Compounds
Thioguanine: Another purine analogue used in the treatment of leukemia.
Azathioprine: A prodrug that is metabolized to mercaptopurine and used as an immunosuppressant.
Allopurinol: A xanthine oxidase inhibitor that can interact with mercaptopurine.
Uniqueness: Mercaptopurine is unique in its ability to inhibit purine metabolism and its dual role as an antineoplastic and immunosuppressant agent . Unlike thioguanine, which is primarily used for its antineoplastic properties, mercaptopurine has broader applications in treating autoimmune diseases .
Biological Activity
6-Mercaptopurine (6-MP) is a purine analog and antimetabolite widely used in the treatment of various hematological malignancies, particularly acute lymphoblastic leukemia (ALL), as well as autoimmune diseases like inflammatory bowel disease (IBD). This article explores the biological activity of 6-MP, focusing on its mechanisms of action, pharmacokinetics, clinical efficacy, and associated risks.
6-MP exerts its therapeutic effects primarily through the following mechanisms:
- Incorporation into Nucleic Acids : 6-MP is metabolized into active thiopurine nucleotides, which can be incorporated into DNA and RNA, leading to the inhibition of purine synthesis and ultimately affecting cell proliferation in rapidly dividing cells .
- Inhibition of Enzymes : It inhibits enzymes involved in the de novo purine synthesis pathway, particularly through the formation of 6-thioguanine nucleotides (6-TGNs), which are responsible for its cytotoxic effects .
- Activation of Nuclear Receptors : Recent studies indicate that 6-MP activates the orphan nuclear receptor NR4A3, enhancing glucose transport activity in skeletal muscle cells and potentially improving insulin sensitivity .
Pharmacokinetics
The pharmacokinetics of 6-MP are characterized by significant interindividual variability influenced by genetic factors such as thiopurine methyltransferase (TPMT) activity. The metabolism involves several key enzymes:
- Thiopurine Methyltransferase (TPMT) : This enzyme methylates 6-MP to inactive metabolites. Individuals with low TPMT activity are at increased risk for toxicity due to higher levels of active metabolites .
- Xanthine Oxidase (XO) : This enzyme converts 6-MP into 6-thiouric acid, another inactive metabolite. Co-administration with allopurinol, an XO inhibitor, can alter the metabolism of 6-MP, leading to increased levels of active thiopurine metabolites .
In Hematological Malignancies
In pediatric patients with ALL, population pharmacokinetic studies have shown that TPMT genotype significantly impacts the clearance of 6-TGNs. Patients with low TPMT activity exhibit higher risks for developing secondary malignancies when treated with 6-MP alongside other cytotoxic agents .
In Inflammatory Bowel Disease
Clinical studies indicate that 6-MP is effective in treating patients with IBD who have previously experienced intolerance to azathioprine. In one study, 73.3% of patients tolerated 6-MP and achieved therapeutic goals after switching from azathioprine . However, adverse effects were noted in approximately 52.6% of patients treated with mercaptopurine .
Case Studies
-
Case Study on Allopurinol Co-Treatment :
A retrospective review involving pediatric ALL patients demonstrated that combining allopurinol with 6-MP effectively reduced gastrointestinal toxicity while maintaining therapeutic efficacy. In this cohort, 18 out of 19 patients achieved desired therapeutic responses after initiating allopurinol therapy . -
Adverse Effects in IBD Patients :
A cohort study reported that adverse effects led to withdrawal in 49 out of 152 patients treated with mercaptopurine for IBD. Despite these challenges, the drug was effective in achieving remission in approximately 39% of cases .
Summary Table: Key Findings on this compound
| Aspect | Findings |
|---|---|
| Mechanism | Incorporation into nucleic acids; inhibition of purine synthesis |
| Key Enzymes | TPMT (methylation), XO (degradation) |
| Efficacy in ALL | High variability; low TPMT linked to increased risk for secondary malignancies |
| Efficacy in IBD | Effective alternative post-azathioprine intolerance; ~39% remission rate |
| Adverse Effects | Common; withdrawal rates ~52.6% in IBD patients |
Q & A
Basic Research Questions
Q. What are the primary biochemical mechanisms of 6-mercaptopurine, and how can researchers experimentally validate its antimetabolite activity?
- Answer : 6-MP inhibits de novo purine synthesis by competing with endogenous purines, disrupting DNA/RNA metabolism. To validate this, researchers can:
- Use radiolabeled purine analogs (e.g., [³H]-hypoxanthine) to measure competitive inhibition in cell cultures .
- Quantify intracellular thioguanine nucleotides (TGNs), the active metabolites of 6-MP, via HPLC or mass spectrometry .
- Conduct proliferation assays (e.g., MTT) in leukemic cell lines (e.g., CCRF-CEM) to assess dose-dependent cytotoxicity .
Q. What experimental models are most appropriate for studying 6-MP's immunosuppressive effects?
- Answer :
- In vitro : Human T-cell activation assays (e.g., anti-CD3/CD28-stimulated PBMCs) with cytokine profiling (IL-2, IFN-γ) .
- In vivo : Murine models of graft-versus-host disease (GVHD) or collagen-induced arthritis, monitoring lymphocyte subsets via flow cytometry .
- Key controls : Include azathioprine (6-MP prodrug) and validate metabolite levels in plasma/tissue .
Q. How should researchers design dose-response studies for 6-MP in preclinical models?
- Answer :
- Use a logarithmic dose range (e.g., 0.1–100 µM in vitro; 1–50 mg/kg in vivo) to capture EC₅₀ values .
- Account for species-specific metabolism: Mice require higher doses (10–40 mg/kg) due to rapid TPMT (thiopurine methyltransferase) activity .
- Monitor toxicity via liver/kidney function markers (ALT, creatinine) and hematological parameters .
Advanced Research Questions
Q. How can meta-analytic methods resolve contradictions in clinical trial data on 6-MP efficacy for inflammatory bowel disease (IBD)?
- Answer :
- Protocol standardization : Pool data from RCTs with consistent endpoints (e.g., Crohn’s Disease Activity Index ≤150 for remission) .
- Statistical approaches : Use fixed-effect models if heterogeneity (I²) <50%; subgroup analyses for steroid-dependent vs. steroid-naïve cohorts .
- Example : A 2016 meta-analysis (n=1,211 patients) found no significant remission benefit (RR 1.23, 95% CI 0.97–1.55) but confirmed steroid-sparing effects (RR 1.34, 95% CI 1.02–1.77) .
Q. What strategies optimize 6-MP’s physicochemical properties for enhanced bioavailability?
- Answer :
- Co-crystallization : Improve solubility using coformers like nicotinamide (e.g., 6-MP:nicotinamide cocrystal, 12-fold solubility increase) .
- Nanoformulations : Liposomal encapsulation to reduce hepatic first-pass metabolism .
- Prodrug design : Develop pH-sensitive derivatives for targeted colonic delivery in IBD .
Q. How can researchers address discrepancies in 6-MP’s carcinogenicity risk assessments?
- Answer :
- Preclinical models : Conduct long-term carcinogenicity studies in TPMT-deficient mice to mimic metabolic variability in humans .
- Genotoxicity assays : Use Comet assays (DNA damage) and in vitro micronucleus tests with metabolic activation (S9 fraction) .
- Epidemiological data : Retrospective cohort studies of IBD patients on 6-MP, stratified by duration (>2 years) and cumulative dose .
Q. What pharmacokinetic-pharmacodynamic (PK-PD) modeling approaches are suitable for 6-MP?
- Answer :
- Population PK models : Incorporate TPMT and NUDT15 polymorphisms to predict TGN variability .
- PD endpoints : Relate erythrocyte TGN levels to clinical response (e.g., >235 pmol/8×10⁸ RBCs correlates with remission in IBD) .
- Software tools : NONMEM or Monolix for nonlinear mixed-effects modeling .
Q. Methodological Guidance
Q. How to ensure reproducibility in 6-MP cytotoxicity assays?
- Key steps :
- Standardize cell passage number and culture conditions (e.g., RPMI-1640 + 10% FBS) .
- Include positive controls (e.g., methotrexate) and normalize viability to untreated cells.
- Pre-treat cells with allopurinol (xanthine oxidase inhibitor) to mimic in vivo metabolism .
Q. What are best practices for synthesizing and characterizing 6-MP derivatives?
- Synthesis : Use Suzuki-Miyaura coupling for purine ring modifications; protect thiol group with trityl chloride .
- Characterization :
- Purity : HPLC with UV detection (λ=322 nm) .
- Structure : ¹H/¹³C NMR (DMSO-d6) and X-ray crystallography (monoclinic P2₁/c space group for 6-MP monohydrate) .
Q. Data Presentation
Table 1 : Key Clinical Trial Outcomes for 6-MP in IBD
Properties
IUPAC Name |
3,7-dihydropurine-6-thione | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C5H4N4S/c10-5-3-4(7-1-6-3)8-2-9-5/h1-2H,(H2,6,7,8,9,10) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
GLVAUDGFNGKCSF-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=NC2=C(N1)C(=S)N=CN2 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C5H4N4S | |
| Record name | mercaptopurine | |
| Source | Wikipedia | |
| URL | https://en.wikipedia.org/wiki/Mercaptopurine | |
| Description | Chemical information link to Wikipedia. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
6112-76-1 (monohydrate) | |
| Record name | Mercaptopurine [USAN:USP:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000050442 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID0020810 | |
| Record name | 6-Mercaptopurine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0020810 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
152.18 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Mercaptopurine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015167 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
22.5 [ug/mL] (The mean of the results at pH 7.4), In water, 6848 mg/L at 30 °C, Insoluble in water, Soluble in boiling water (1 in 100), Soluble in hot alcohol and dilute alkali solutions; slightly soluble in dilute sulfuric acid, Soluble in alkaline solutions (with decomposition), hot ethanol and ethanol (1 in 950); slightly soluble in dilute sulphuric acid; almost insoluble in water, acetone, chloroform and diethyl ether., 7.35e-01 g/L | |
| Record name | SID49675007 | |
| Source | Burnham Center for Chemical Genomics | |
| URL | https://pubchem.ncbi.nlm.nih.gov/bioassay/1996#section=Data-Table | |
| Description | Aqueous solubility in buffer at pH 7.4 | |
| Record name | Mercaptopurine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01033 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Mercaptopurine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3235 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Mercaptopurine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015167 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Mercaptopurine competes with hypoxanthine and guanine for the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRTase) and is itself converted to thioinosinic acid (TIMP). TIMP inhibits several reactions that involve inosinic acid (IMP), such as the conversion of IMP to xanthylic acid (XMP) and the conversion of IMP to adenylic acid (AMP) via adenylosuccinate (SAMP). Upon methylation, TIMP forms 6-methylthioinosinate (MTIMP) which inhibits glutamine-5-phosphoribosylpyrophosphate amidotransferase in addition to TIMP. Glutamine-5-phosphoribosylpyrophosphate amidotransferase is the first enzyme unique to the _de novo_ pathway for purine ribonucleotide synthesis. According to experimental findings using radiolabeled mercaptopurine, mercaptopurine may be recovered from the DNA in the form of deoxythioguanosine. In comparison, some mercaptopurine may be converted to nucleotide derivatives of 6-thioguanine (6-TG) via actions of inosinate (IMP) dehydrogenase and xanthylate (XMP) aminase that convert TIMP to thioguanylic acid (TGMP)., The pathogenesis of several neurodegenerative diseases often involves the microglial activation and associated inflammatory processes. Activated microglia release pro-inflammatory factors that may be neurotoxic. 6-Mercaptopurine (6-MP) is a well-established immunosuppressive drug. Common understanding of their immunosuppressive properties is largely limited to peripheral immune cells. However, the effect of 6-MP in the central nervous system, especially in microglia in the context of neuroinflammation is, as yet, unclear. Tumor necrosis factor-alpha (TNF-a) is a key cytokine of the immune system that initiates and promotes neuroinflammation. The present study aimed to investigate the effect of 6-MP on TNF-a production by microglia to discern the molecular mechanisms of this modulation. Lipopolysaccharide (LPS) was used to induce an inflammatory response in cultured primary microglia or murine BV-2 microglial cells. Released TNF-a was measured by enzyme-linked immunosorbent assay (ELISA). Gene expression was determined by real-time reverse transcription polymerase chain reaction (RT-PCR). Signaling molecules were analyzed by western blotting, and activation of NF-kB was measured by ELISA-based DNA binding analysis and luciferase reporter assay. Chromatin immunoprecipitation (ChIP) analysis was performed to examine NF-kB p65 and coactivator p300 enrichments and histone modifications at the endogenous TNF-a promoter. Treatment of LPS-activated microglia with 6-MP significantly attenuated TNF-a production. In 6-MP pretreated microglia, LPS-induced MAPK signaling, I?B-a degradation, NF-kB p65 nuclear translocation, and in vitro p65 DNA binding activity were not impaired. However, 6-MP suppressed transactivation activity of NF-?B and TNF-a promoter by inhibiting phosphorylation and acetylation of p65 on Ser276 and Lys310, respectively. ChIP analyses revealed that 6-MP dampened LPS-induced histone H3 acetylation of chromatin surrounding the TNF-a promoter, ultimately leading to a decrease in p65/coactivator-mediated transcription of TNF-a gene. Furthermore, 6-MP enhanced orphan nuclear receptor Nur77 expression. Using RNA interference approach, we further demonstrated that Nur77 upregulation contribute to 6-MP-mediated inhibitory effect on TNF-a production. Additionally, 6-MP also impeded TNF-a mRNA translation through prevention of LPS-activated PI3K/Akt/mTOR signaling cascades. These results suggest that 6-MP might have a therapeutic potential in neuroinflammation-related neurodegenerative disorders through downregulation of microglia-mediated inflammatory processes., Mercaptopurine (6-MP) competes with hypoxanthine and guanine for the enzyme hyphoxanthine-guanine phosphoribosyltransferase (HGPRTase) and is itself converted to thioinosinic acid (TIMP). This intracellular nucleotide inhibits several reactions involving inosinic acid (IMP), including the conversion of IMP to xanthylic acid (XMP) and the conversion of IMP to adenylic acid (AMP) via adenylosuccinate (SAMP). In addition, 6-methylthioinosinate (MTIMP) is formed by the methylation of TIMP. Both TIMP and MTIMP have been reported to inhibit glutamine-5-phosphoribosylpyrophosphate amidotransferase, the first enzyme unique to the de novo pathway for purine ribonucleotide synthesis. Experiments indicate that radiolabeled mercaptopurine may be recovered from the DNA in the form of deoxythioguanosine. Some mercaptopurine is converted to nucleotide derivatives of 6-thioguanine (6-TG) by the sequential actions of inosinate (IMP) dehydrogenase and xanthylate (XMP) aminase, converting TIMP to thioguanylic acid (TGMP). Animal tumors that are resistant to mercaptopurine often have lost the ability to convert mercaptopurine to TIMP. However, it is clear that resistance to mercaptopurine may be acquired by other means as well, particularly in human leukemias. It is not known exactly which of any one or more of the biochemical effects of mercaptopurine and its metabolites are directly or predominantly responsible for cell death., Inflammatory bowel disease is characterized by chronic intestinal inflammation. Azathioprine and its metabolite 6-mercaptopurine (6-MP) are effective immunosuppressive drugs that are widely used in patients with inflammatory bowel disease. ... Azathioprine and 6-MP have been shown to affect small GTPase Rac1 in T cells and endothelial cells, whereas the effect on macrophages and gut epithelial cells is unknown. Macrophages (RAW cells) and gut epithelial cells (Caco-2 cells) were activated by cytokines and the effect on Rac1 signaling was assessed in the presence or absence of 6-MP. Rac1 is activated in macrophages and epithelial cells, and treatment with 6-MP resulted in Rac1 inhibition. In macrophages, interferon-gamma induced downstream signaling through c-Jun-N-terminal Kinase (JNK) resulting in inducible nitric oxide synthase (iNOS) expression. iNOS expression was reduced by 6-MP in a Rac1-dependent manner. In epithelial cells, 6-MP efficiently inhibited tumor necrosis factor-a-induced expression of the chemokines CCL2 and interleukin-8, although only interleukin-8 expression was inhibited in a Rac1-dependent manner. In addition, activation of the transcription factor STAT3 was suppressed in a Rac1-dependent fashion by 6-MP, resulting in reduced proliferation of the epithelial cells due to diminished cyclin D1 expression. These data demonstrate that 6-MP affects macrophages and gut epithelial cells beneficially, in addition to T cells and endothelial cells. Furthermore, mechanistic insight is provided to support development of Rac1-specific inhibitors for clinical use in inflammatory bowel disease. | |
| Record name | Mercaptopurine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01033 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Mercaptopurine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3235 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Yellow crystalline powder, Yellow prisms from water (+1 water), Dark yellow /Mercaptopurine hydrate/ | |
CAS No. |
50-44-2, 6112-76-1 | |
| Record name | 6-Mercaptopurine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=50-44-2 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Mercaptopurine [USAN:USP:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000050442 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Mercaptopurine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01033 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | mercaptopurine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=759614 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Mercaptopurine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=755 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | 6-Mercaptopurine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0020810 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 1,7-dihydro-6H-purine-6-thione hydrate (1:1) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.126.167 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | Mercaptopurine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.000.035 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | MERCAPTOPURINE ANHYDROUS | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/PKK6MUZ20G | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Mercaptopurine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3235 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Mercaptopurine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015167 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
308 °C (decomposes), 313 °C | |
| Record name | Mercaptopurine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01033 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Mercaptopurine | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3235 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Mercaptopurine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015167 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Synthesis routes and methods
Procedure details





Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













