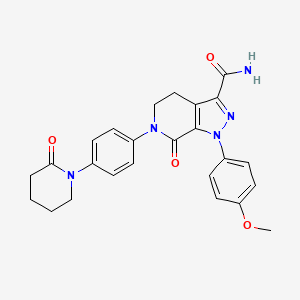
Apixaban
Overview
Description
Apixaban, marketed under the brand name Eliquis, is an anticoagulant medication used to treat and prevent blood clots and to prevent stroke in people with nonvalvular atrial fibrillation. It works by directly inhibiting factor Xa, an enzyme crucial for blood clotting . This compound is also used to prevent blood clots following hip or knee replacement surgery and in patients with a history of prior clots .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of apixaban involves multiple steps, starting from inexpensive 4-chloronitrobenzene and piperidine. An eight-step procedure is typically employed, which includes the use of sodium chlorite to oxidize the piperidine cycle to the corresponding lactam under a carbon dioxide atmosphere . This process results in the construction of two six-ring lactams, which are essential for the final structure of this compound .
Industrial Production Methods: In industrial settings, the synthesis of this compound is optimized for efficiency and scalability. The process involves the use of diisopropylethylamine and methane sulfonyl chloride in dichloromethane at low temperatures, followed by the addition of sodium ethylate . The reaction mixture is then subjected to extraction and recrystallization to obtain high-purity this compound .
Chemical Reactions Analysis
Types of Reactions: Apixaban undergoes various chemical reactions, including oxidation, reduction, and substitution reactions.
Common Reagents and Conditions:
Oxidation: Sodium chlorite is used to oxidize the piperidine cycle to lactams under a carbon dioxide atmosphere.
Substitution: Methane sulfonyl chloride is used in the presence of diisopropylethylamine to introduce sulfonyl groups.
Major Products: The major products formed from these reactions are the intermediate lactams and the final this compound compound .
Scientific Research Applications
Clinical Indications
1. Atrial Fibrillation
Apixaban is indicated for the prevention of stroke and systemic embolism in patients with nonvalvular atrial fibrillation. The ARISTOTLE trial demonstrated that this compound significantly reduced the risk of stroke or systemic embolism compared to warfarin, with a hazard ratio of 0.79 (95% confidence interval 0.66-0.95) . Additionally, it showed a 31% reduction in major bleeding events compared to warfarin, making it a preferred option for many clinicians .
2. Venous Thromboembolism (VTE)
this compound is also used for thromboprophylaxis following hip or knee replacement surgery and for the treatment of deep vein thrombosis and pulmonary embolism. In clinical trials, this compound has demonstrated efficacy in reducing recurrent VTE events . The compound's pharmacokinetics support its use; it has an oral bioavailability of approximately 50%, with peak plasma concentrations occurring 3-4 hours post-administration .
3. Secondary Stroke Prevention
Recent studies, such as the ARCADIA trial, have explored this compound's role in preventing recurrence after cryptogenic stroke. This trial compares this compound with aspirin in patients with recent cryptogenic strokes and underlying atrial cardiopathy . The ongoing research aims to establish this compound's effectiveness in this specific population.
Pharmacological Profile
This compound's mechanism involves inhibiting both free and clot-bound factor Xa, which is crucial for thrombin generation and subsequent platelet activation. Its pharmacokinetic properties include rapid absorption and a half-life of approximately 12 hours, allowing for twice-daily dosing without the need for routine monitoring .
Comparative Efficacy
| Study | Population | Outcome | Result |
|---|---|---|---|
| ARISTOTLE | Atrial fibrillation patients | Stroke/systemic embolism reduction | 21% reduction vs. warfarin |
| AVERROES | High-risk atrial fibrillation | Efficacy vs. aspirin | 45% reduction in stroke/systemic embolism risk |
| VTE Trials | Post-surgery patients | Recurrence of VTE | Significant reduction compared to placebo |
Case Studies
Case Study 1: Long-term Use in Atrial Fibrillation
The AVERROES trial included patients unsuitable for vitamin K antagonists who were treated with this compound during an open-label extension phase. Results indicated that long-term use maintained efficacy in reducing stroke risk without significantly increasing major bleeding rates .
Case Study 2: Postoperative Thromboprophylaxis
In a cohort study involving patients undergoing hip or knee replacement surgeries, this compound was shown to effectively reduce the incidence of postoperative deep vein thrombosis without increasing bleeding complications compared to traditional anticoagulants .
Mechanism of Action
Apixaban exerts its effects by directly inhibiting factor Xa, an enzyme that plays a crucial role in the conversion of prothrombin to thrombin, a key step in the blood clotting process . By inhibiting factor Xa, this compound prevents the formation of thrombin and, consequently, the formation of blood clots . This mechanism is independent of antithrombin III, making this compound a direct and selective inhibitor of factor Xa .
Comparison with Similar Compounds
Rivaroxaban: Another oral factor Xa inhibitor with similar bioavailability and selectivity.
Edoxaban: Also a direct factor Xa inhibitor used for similar indications.
Dabigatran: A direct thrombin inhibitor, which works by a different mechanism compared to apixaban.
Uniqueness of this compound: this compound is unique due to its predictable pharmacokinetic and pharmacodynamic properties, which allow for fixed dosages without the need for routine monitoring . It has a rapid onset and offset of action, low potential for food or drug interactions, and is effective in a wide range of patients, including those with moderate renal impairment .
Biological Activity
Apixaban is a direct oral anticoagulant (DOAC) that selectively inhibits Factor Xa (FXa), a crucial enzyme in the coagulation cascade. Its biological activity is characterized by its ability to prevent thrombin generation and thrombus formation, making it effective in various clinical settings, including atrial fibrillation, venous thromboembolism, and in patients undergoing certain surgical procedures.
This compound functions as a competitive inhibitor of FXa. It binds to the active site of the enzyme, preventing the conversion of prothrombin to thrombin. This inhibition is dose-dependent and reversible, with a rapid onset of action observed in both in vitro and in vivo studies. The dissociation rate constant for this compound's inhibition of FXa is approximately at 25°C and at 37°C .
Inhibition Profiles
- Free FXa : IC50 of 1.3 nM.
- Thrombus-associated FXa : Effective inhibition demonstrated.
- Prothrombinase complex : Exhibits non-competitive inhibition, indicating that prothrombin binding is influenced by interactions at exosites of FXa .
Pharmacodynamics
This compound’s pharmacodynamic properties have been extensively studied. In vitro assays show that this compound significantly inhibits thrombin generation in human plasma:
- Tissue factor-initiated thrombin generation : IC50 values range from 37 nM to 100 nM depending on the assay conditions .
- Platelet aggregation : While this compound does not directly affect platelet aggregation, it indirectly reduces aggregation induced by thrombin due to its inhibition of thrombin generation .
Clinical Efficacy
Several clinical trials have assessed the efficacy of this compound across different patient populations:
- Atrial Fibrillation : The AVERROES trial demonstrated that this compound is superior to aspirin in preventing stroke and systemic embolism in patients with atrial fibrillation .
- Venous Thromboembolism Prevention : In cancer patients receiving chemotherapy, this compound was well tolerated with a low incidence of major bleeding (2.2%) while effectively preventing venous thromboembolism (VTE) .
- Comparative Studies : A study comparing this compound with warfarin indicated that while both drugs are effective, this compound had a higher adherence rate and fewer complications related to anticoagulation monitoring .
Case Study 1: Efficacy in Cancer Patients
In a randomized pilot study involving cancer patients undergoing chemotherapy, patients were assigned to receive varying doses of this compound (5 mg, 10 mg, or 20 mg) or placebo. The study found that this compound was well tolerated with minimal major bleeding incidents reported .
Case Study 2: Stroke Prevention
The ARCADIA trial investigated the effectiveness of this compound versus aspirin for secondary stroke prevention in patients with recent cryptogenic strokes. Preliminary results suggest that this compound may provide better outcomes compared to traditional antiplatelet therapy .
Pharmacokinetics
This compound exhibits favorable pharmacokinetic properties:
- Bioavailability : Approximately 50% for doses up to 10 mg.
- Absorption : Rapidly absorbed with peak plasma concentrations occurring within 3 to 4 hours post-administration .
- Half-life : Approximately 12 hours, allowing for twice-daily dosing.
Comparative Efficacy Table
| Study/Trial | Population | This compound Dosage | Major Outcomes |
|---|---|---|---|
| AVERROES | Atrial Fibrillation | 5 mg twice daily | Reduced stroke/systemic embolism |
| Cancer Study | Chemotherapy Patients | 5 mg, 10 mg, 20 mg | Low major bleeding (2.2%) |
| ARCADIA | Cryptogenic Stroke | TBD | Improved outcomes vs aspirin |
Q & A
Basic Research Questions
Q. What are the primary pharmacological mechanisms of apixaban in thrombosis prevention, and how are they validated experimentally?
this compound selectively inhibits factor Xa (FXa), disrupting the coagulation cascade by preventing thrombin generation. Its mechanism is validated through in vitro assays measuring FXa inhibition (Ki = 0.08 nM for human FXa) and in vivo models like arteriovenous shunt thrombosis (AVST) in rabbits. These studies confirm dose-dependent antithrombotic efficacy without significant interference with platelet aggregation, supporting its clinical safety profile .
Q. How are clinical trials for this compound designed to assess efficacy and safety endpoints?
Phase III trials (e.g., ARISTOTLE, AMPLIFY) use randomized, double-blind designs with non-inferiority/superiority objectives. Primary endpoints include stroke/systemic embolism rates, while safety endpoints focus on major bleeding (ISTH criteria). For example, in ARISTOTLE, this compound (5 mg BID) reduced stroke risk by 21% (HR 0.79; 95% CI 0.66–0.95) and major bleeding by 31% (HR 0.69; 95% CI 0.60–0.80) compared to warfarin, with mortality as a secondary endpoint .
Q. What standard dosing regimens are applied for this compound in different clinical scenarios, and how are they derived?
Dosing is based on pharmacokinetic (PK) studies showing a half-life of ~12 hours and renal clearance (~25%). The 5 mg BID dose for atrial fibrillation (AF) was optimized in Phase II trials balancing efficacy/bleeding risks. Reduced dosing (2.5 mg BID) applies to patients with ≥2 of: age ≥80 years, body weight ≤60 kg, or serum creatinine ≥1.5 mg/dL. These thresholds were validated in subgroup analyses of the ARISTOTLE trial .
Advanced Research Questions
Q. What experimental models are used to evaluate this compound’s antithrombotic effects, and how do they inform translational research?
In vitro flow chambers under arterial shear rates (1,500 s⁻¹) assess thrombus formation dynamics, revealing this compound’s inhibition of fibrin-rich clot stabilization. In vivo models like electrically mediated carotid arterial thrombosis (ECAT) in rabbits demonstrate dose-dependent efficacy (ED₅₀ = 0.07 mg/kg/h IV). These models correlate with clinical outcomes, such as reduced D-dimer levels in venous thromboembolism (VTE) patients .
Q. How do researchers address discrepancies in bleeding risk outcomes across this compound studies?
Contradictions (e.g., lower major bleeding in RCTs vs. real-world data) are resolved via meta-regression adjusting for comorbidities (e.g., renal impairment) and adherence rates. Sensitivity analyses in cost-effectiveness studies isolate variables like drug discontinuation rates. For instance, RWD-based models show this compound’s bleeding risk aligns with RCTs when adherence exceeds 80% .
Q. What methodologies are employed in meta-analyses comparing this compound with other direct oral anticoagulants (DOACs)?
Network meta-analyses (NMAs) integrate RCTs (e.g., RE-LY, ROCKET-AF) using Bayesian frameworks. Efficacy/safety hierarchies are derived via surface under the cumulative ranking curve (SUCRA) scores. For example, this compound ranks highest in safety (SUCRA 94%) due to 32% lower major bleeding risk vs. rivaroxaban (RR 0.68; 95% CI 0.61–0.76) in pooled observational data .
Q. How do pharmacokinetic studies inform this compound dosing in special populations, such as renal-impaired patients?
Population PK models quantify exposure-response relationships using covariates like creatinine clearance (CrCl). Simulations show a 56% AUC increase in severe renal impairment (CrCl 15–29 mL/min), justifying dose reduction to 2.5 mg BID. These models are validated against Phase I data showing linear PK and minimal drug-drug interactions with CYP3A4 inhibitors .
Properties
IUPAC Name |
1-(4-methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-yl)phenyl]-4,5-dihydropyrazolo[3,4-c]pyridine-3-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C25H25N5O4/c1-34-19-11-9-18(10-12-19)30-23-20(22(27-30)24(26)32)13-15-29(25(23)33)17-7-5-16(6-8-17)28-14-3-2-4-21(28)31/h5-12H,2-4,13-15H2,1H3,(H2,26,32) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
QNZCBYKSOIHPEH-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
COC1=CC=C(C=C1)N2C3=C(CCN(C3=O)C4=CC=C(C=C4)N5CCCCC5=O)C(=N2)C(=O)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C25H25N5O4 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80436500 | |
| Record name | Apixaban | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80436500 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
459.5 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
0.11mg/mL, Aqueous solubility across the physiological pH range is approximately 0.04 mg/L | |
| Record name | Apixaban | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06605 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Apixaban | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8223 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
Apixaban selectively inhibits factor Xa in its free and bound forms, independant of antithrombin III. Apixaban also inhibits prothrominase. These effects prevent the formation of a thrombus. | |
| Record name | Apixaban | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06605 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Color/Form |
White to pale-yellow powder | |
CAS No. |
503612-47-3 | |
| Record name | Apixaban | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=503612-47-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Apixaban | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB06605 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Apixaban | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80436500 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (1-(4-methoxyphenyl)-7-oxo-6-[4-(2-oxopiperidin-1-yl)phenyl]-4,5-dihydropyrazolo[3,4-c]pyridine-3-carboxamide) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | APIXABAN | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/3Z9Y7UWC1J | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Apixaban | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8223 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Synthesis routes and methods I
Procedure details







Synthesis routes and methods II
Procedure details








Synthesis routes and methods III
Procedure details








Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.














