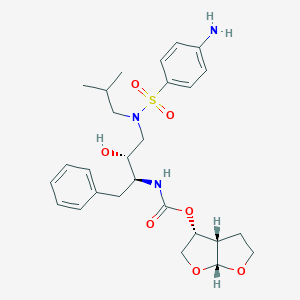
Darunavir
Overview
Description
Darunavir is a nonpeptidic inhibitor of protease used primarily in the treatment and prevention of human immunodeficiency virus (HIV) infection. It is often administered in combination with other antiretroviral agents to enhance its efficacy. This compound was approved by the United States Food and Drug Administration in 2006 and is included in the World Health Organization’s List of Essential Medicines .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of darunavir involves multiple steps, including the formation of key intermediates and their subsequent coupling. One common synthetic route includes the removal of the BOC group using trifluoroacetic acid in dichloromethane, followed by reaction with a carbonate in the presence of triethylamine to yield this compound .
Industrial Production Methods: Industrial production of this compound ethanolate involves a robust process with multiple isolations and drying steps. This process ensures high chemical yield and purity, with critical process impurities controlled below desired limits . Additionally, methods such as hot-melt extrusion and spray-drying are employed to improve the solubility and bioavailability of this compound .
Chemical Reactions Analysis
Types of Reactions: Darunavir undergoes various chemical reactions, including oxidation, reduction, and substitution. It is heavily oxidized and metabolized by hepatic cytochrome enzymes, primarily CYP3A4 .
Common Reagents and Conditions:
Oxidation: Involves hepatic cytochrome enzymes.
Reduction: Not commonly reported for this compound.
Substitution: Involves reactions with carbonates and amines.
Major Products: The major products formed from these reactions include hydroxylated and glucuronidated metabolites .
Scientific Research Applications
Efficacy in Treatment-Naïve Patients
A meta-analysis involving multiple studies has shown that darunavir/ritonavir-based regimens are effective for treatment-naïve patients. The virological response rate was comparable to other first-line therapies:
| Study | Population | Response Rate | |
|---|---|---|---|
| ARTEMIS Trial | Treatment-Naïve | 84% at 48 weeks | Effective compared to other ARTs |
| POWER Trials | Treatment-Experienced | 73% at 48 weeks | Superior virological response |
The ARTEMIS trial specifically highlighted that this compound/ritonavir was effective in achieving viral suppression in treatment-naïve patients over a 48-week period .
Efficacy in Treatment-Experienced Patients
This compound has shown significant efficacy in treatment-experienced patients, particularly those with prior exposure to other antiretroviral agents. A study indicated that this compound/ritonavir provided a higher virological response compared to alternative treatments:
| Study | Population | Response Rate | |
|---|---|---|---|
| This compound Outcomes Study | Three-Class Experienced | 63% at 48 weeks | Demonstrated superior efficacy over other regimens |
| GRACE Study | Sex-Based Outcomes | No significant differences between sexes | Effective across genders |
The this compound Outcomes Study confirmed that patients with multiple drug resistance had a sustained virological response when treated with this compound .
Safety Profile
This compound is generally well-tolerated, with adverse events reported at similar rates compared to other antiretrovirals. Common side effects include gastrointestinal symptoms and rash, but serious adverse events are rare:
| Adverse Event | Frequency (%) |
|---|---|
| Diarrhea | 10 |
| Rash | 7 |
| Nausea | 5 |
In clinical trials, this compound's safety profile was comparable to other antiretroviral therapies, reinforcing its use as a first-line agent .
Innovative Research and Future Directions
Recent studies have focused on developing this compound analogs using advanced computational methods such as fragment molecular orbital techniques. These efforts aim to create compounds with enhanced efficacy against resistant HIV strains:
- FMO-guided Design : Novel this compound analogs are being developed to overcome resistance issues associated with HIV-1 protease mutations.
- Molecular Docking Studies : These studies assess the binding affinity of new analogs against wild-type and mutant strains of HIV-1 protease.
The ongoing research aims to improve the therapeutic options available for patients who exhibit resistance to current treatments .
Case Studies
-
Case Study: Treatment-Naïve Patient
- Background : A 35-year-old male diagnosed with HIV-1.
- Treatment : Initiated on this compound/ritonavir plus two nucleoside reverse transcriptase inhibitors.
- Outcome : Achieved viral load suppression (<50 copies/mL) by week 48.
-
Case Study: Treatment-Experienced Patient
- Background : A 50-year-old female with extensive treatment history and resistance mutations.
- Treatment : Switched to this compound/ritonavir.
- Outcome : Achieved sustained viral suppression after 24 weeks despite previous failures on other regimens.
Mechanism of Action
Darunavir exerts its effects by inhibiting the HIV-1 protease enzyme, which is essential for viral precursor protein processing and viral maturation. By binding to the enzyme’s active site, this compound prevents the cleavage of the HIV Gag-Pol polyprotein, thereby blocking the formation of infectious virions . This mechanism is crucial for its antiretroviral activity.
Comparison with Similar Compounds
- Amprenavir
- Indinavir
- Saquinavir
- Nelfinavir
- Ritonavir
Comparison: Darunavir is unique among protease inhibitors due to its high genetic barrier to resistance and its ability to inhibit a wide range of protease inhibitor-resistant HIV-1 strains . Unlike some other protease inhibitors, this compound is often used in combination with ritonavir or cobicistat to enhance its pharmacokinetic profile .
Biological Activity
Darunavir (DRV) is a second-generation protease inhibitor (PI) used in the treatment of HIV-1 infection. It exhibits potent antiviral activity and is particularly effective against both wild-type and drug-resistant strains of the virus. This article delves into the biological activity of this compound, including its mechanisms of action, efficacy in clinical settings, and pharmacological properties, supported by relevant case studies and research findings.
This compound works by inhibiting the HIV-1 protease enzyme, which is crucial for the viral replication process. By binding to the active site of the protease, this compound prevents the cleavage of viral polyproteins into functional proteins, thereby halting viral maturation and replication.
Structural Insights
Recent studies have provided insights into this compound's binding interactions with HIV-1 protease. Modifications in its structure have been shown to enhance its binding affinity through improved hydrogen bonding interactions within the enzyme's active site. For instance, modifications to the P2’ 4-amino group have led to derivatives with enhanced enzyme inhibitory activity .
Efficacy in Clinical Settings
This compound has been extensively studied for its efficacy in both treatment-naïve and treatment-experienced patients. A meta-analysis encompassing multiple randomized controlled trials revealed significant findings:
- For Treatment-Naïve Patients : this compound/ritonavir (DRV/r) demonstrated comparable virological response rates to other treatments at both 48 and 96 weeks, with no significant differences noted in efficacy .
- For Treatment-Experienced Patients : DRV/r showed a significantly higher virological response rate compared to other regimens (Risk Ratio: 1.45), indicating its effectiveness in patients who had previously failed therapy .
Pharmacokinetics and Safety Profile
This compound is primarily metabolized by CYP3A4 enzymes in the liver. Its pharmacokinetic profile allows it to be used effectively even in patients with varying degrees of liver function. Notably, a case study reported an unintentional overdose of this compound in an adolescent patient which resulted in minimal toxicity yet improved virological suppression, suggesting that higher doses may be beneficial under certain circumstances .
Table 1: Summary of Pharmacokinetic Properties
| Property | Value |
|---|---|
| Half-life | 15 hours |
| Bioavailability | ~37% |
| Peak plasma concentration | 2-4 hours post-dose |
| Volume of distribution | ~100 L |
Case Studies
- High-Dose this compound Use : An adolescent with reduced susceptibility to this compound underwent a regimen involving a high dose (1200 mg) which resulted in sustained viral load suppression (<400 copies/mL) and normalization of CD4 counts over a 24-month period .
- Meta-Analysis Findings : In a comprehensive review involving over 6,000 patients, DRV/r was found to be effective and well-tolerated across diverse populations, reinforcing its role as a frontline therapy for HIV .
Resistance and Future Directions
Emerging multidrug-resistant strains of HIV pose challenges to treatment efficacy. Research into this compound analogs aims to overcome these limitations by enhancing binding affinity and efficacy against resistant strains . Continuous monitoring of cardiovascular risks associated with this compound therapy is also essential, as some studies have indicated potential adverse effects on cardiovascular health .
Q & A
Basic Research Questions
Q. How does darunavir’s molecular design confer a high genetic barrier to resistance compared to earlier protease inhibitors (PIs)?
this compound was designed using structural biology principles to preemptively address common resistance mutations. Its bis-tetrahydrofuranylurethane (bis-THF) group and sulfonamide moiety form extensive hydrogen bonds with conserved regions of the HIV-1 protease, reducing susceptibility to mutations. Unlike first-generation PIs, this compound’s scaffold minimizes steric clashes with mutated protease variants, maintaining binding affinity even when 18–21 resistance-associated mutations (RAMs) accumulate . Methodological Insight: Use X-ray crystallography and enzyme kinetics to compare this compound-protease binding patterns against mutant vs. wild-type proteases .
Q. What pharmacokinetic (PK) models are used to optimize this compound dosing in special populations (e.g., pregnant women)?
Semi-mechanistic population PK models integrating total and unbound this compound concentrations are critical. For pregnant women, nonlinear protein binding (due to increased α-1-acid glycoprotein) necessitates adjusted dosing. A study analyzing 2,601 plasma samples from 85 women used simulations to predict AUC0-τ and Ctrough during the third trimester, recommending therapeutic drug monitoring (TDM) to maintain unbound this compound levels above the protein-adjusted IC50 (PA-IC50) of 55 ng/mL .
Q. Which mutations are most strongly associated with reduced this compound susceptibility in treatment-experienced patients?
Primary RAMs include V32I, L33F, I47V, I54L/M, and L76V. Pooled data from POWER and DUET trials show that ≥3 RAMs reduce virologic response by 41% (vs. 78% in RAM-free strains). Genotypic resistance scores (e.g., ≥10 mutations from the IAS-USA list) correlate with phenotypic fold-changes in EC50 . Methodological Insight: Use phenotypic susceptibility assays (e.g., Antivirogram®) alongside genotypic sequencing to quantify resistance thresholds .
Advanced Research Questions
Q. How do structural dynamics in this compound-resistant protease variants impair inhibitor binding despite retained catalytic activity?
Highly mutated proteases (e.g., with 18–21 substitutions) exhibit distorted hydrogen-bond networks in the P2′ pocket, reducing this compound’s binding affinity by 20-fold. However, catalytic efficiency remains ~5% of wild-type levels, sufficient for viral replication. Crystallographic studies reveal that mutations like I84V shift the this compound aminophenyl group, disrupting key van der Waals interactions . Experimental Design: Combine cryo-EM for conformational analysis with molecular dynamics simulations to map residue flexibility .
Q. Why do some clinical trials show no correlation between this compound PK exposure and virologic suppression in susceptible strains?
In PI-naive patients, this compound/ritonavir 800/100 mg once-daily achieved similar AUCs to 600/100 mg twice-daily, but virologic responses were comparable (74–78% at 24 weeks). This suggests that exceeding the PA-IC50 threshold—not AUC maximization—is critical. Subanalyses indicate that adherence and baseline viral load are stronger predictors of outcome than PK variability . Data Contradiction Resolution: Apply multivariate regression to isolate PK parameters from confounding variables (e.g., adherence logs, baseline resistance) .
Q. What analytical methods validate this compound-ritonavir coformulations in bioequivalence studies?
- HPLC: A validated method with retention times of 2.358 min (this compound) and 3.099 min (ritonavir), resolution >2, and tailing factor <2% ensures specificity. System suitability testing requires ≤15% variability in peak area ratios .
- Spectrophotometry: A QBD-optimized fluorometric method using 0.1 N HCl achieves 98.15% recovery with ≤2% RSD in intraday/interday precision. Calibration curves are linear (5–30 μg/mL) with LOD/LOQ of 0.12/0.37 μg/mL .
Q. How can this compound resistance pathways inform the design of next-generation PIs?
Resistance to this compound requires accumulation of “clusters” of mutations (e.g., V32I + L33F + I47V + I54L). Structural modeling of these clusters reveals compensatory mechanisms (e.g., L76V restores protease stability). Novel PIs targeting the flap or hinge regions (e.g., GRL-0202) are designed to avoid these adaptive networks . Methodological Insight: Use deep mutational scanning to identify epistatic interactions between mutations .
Properties
IUPAC Name |
[(3aS,4R,6aR)-2,3,3a,4,5,6a-hexahydrofuro[2,3-b]furan-4-yl] N-[(2S,3R)-4-[(4-aminophenyl)sulfonyl-(2-methylpropyl)amino]-3-hydroxy-1-phenylbutan-2-yl]carbamate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C27H37N3O7S/c1-18(2)15-30(38(33,34)21-10-8-20(28)9-11-21)16-24(31)23(14-19-6-4-3-5-7-19)29-27(32)37-25-17-36-26-22(25)12-13-35-26/h3-11,18,22-26,31H,12-17,28H2,1-2H3,(H,29,32)/t22-,23-,24+,25-,26+/m0/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
CJBJHOAVZSMMDJ-HEXNFIEUSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC(C)CN(CC(C(CC1=CC=CC=C1)NC(=O)OC2COC3C2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CC(C)CN(C[C@H]([C@H](CC1=CC=CC=C1)NC(=O)O[C@H]2CO[C@@H]3[C@H]2CCO3)O)S(=O)(=O)C4=CC=C(C=C4)N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C27H37N3O7S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID0046779 | |
| Record name | Darunavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0046779 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
547.7 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Solid | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
Approximately 0.15 mg/mL at, 6.68e-02 g/L | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
The HIV-1 protease enzyme is necessary for viral precursor protein processing and viral maturation in preparation for infection, and is therefore a target for antiretroviral therapy for HIV. Protease inhibitors are used as a part of highly active antiretroviral therapy (HAART) in patients diagnosed with HIV infection. It has been shown to effectively suppress the virus, leading to significantly decreased morbidity and mortality rates. Darunavir, a HIV protease inhibitor, prevents HIV replication through binding to the enzyme, stopping the dimerization and the catalytic activity of HIV-1 protease. In particular, it inhibits the cleavage of HIV encoded Gag-Pol proteins in cells that have been infected with the virus, halting the formation of mature virus particles, which spread the infection. The close contact that darunavir makes with the primary chains of the active site amino acids (Asp-29 and Asp-30) on the protease likely contributes to its potency and efficacy against resistant variants of HIV-1. Darunavir is known to bind to different sites on the enzyme: the active site cavity and the surface of one of the flexible flaps in the protease dimer. Darunavir can adapt to changes in the shape of a protease enzyme due to its molecular flexibility., Darunavir as a protease inhibitor inhibits the cleavage of HIV encoded gag-pol polyproteins in virus infected cells, thereby preventing the formation of mature and infectious new virions. It was selected for its potency against wild type HIV-1 and HIV strains resistant to currently approved protease inhibitors., Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles. | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
White, amorphous solid | |
CAS No. |
206361-99-1 | |
| Record name | Darunavir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=206361-99-1 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Darunavir [USAN:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0206361991 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID0046779 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Carbamic acid, N-[(1S,2R)-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-, (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ester | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DARUNAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/YO603Y8113 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Darunavir | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0015393 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
74-76, 74 °C (decomposes) | |
| Record name | Darunavir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB01264 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Darunavir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/7788 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















