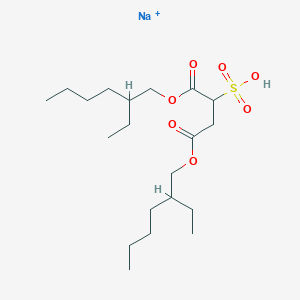
Docusate sodium
Overview
Description
Docusate sodium, also known as dioctyl sodium sulfosuccinate, is a widely used compound in the medical field, primarily as a stool softener. It is an anionic surfactant that helps to increase the amount of water the stool absorbs in the gut, making the stool softer and easier to pass. This compound is commonly used to treat constipation and is available in various forms, including oral and rectal administration .
Preparation Methods
Synthetic Routes and Reaction Conditions: Docusate sodium is synthesized through the esterification of sulfosuccinic acid with 2-ethylhexanol, followed by neutralization with sodium hydroxide. The reaction typically involves heating the reactants under reflux conditions to facilitate the esterification process. The resulting product is then purified through distillation or recrystallization to obtain this compound in its pure form .
Industrial Production Methods: In industrial settings, this compound is produced on a larger scale using similar synthetic routes. The process involves the continuous esterification of sulfosuccinic acid with 2-ethylhexanol in the presence of a catalyst, followed by neutralization with sodium hydroxide. The product is then subjected to various purification steps, including filtration, distillation, and drying, to achieve the desired purity and quality .
Chemical Reactions Analysis
Types of Reactions: Docusate sodium primarily undergoes hydrolysis and oxidation reactions. In the presence of strong acids or bases, it can hydrolyze to form sulfosuccinic acid and 2-ethylhexanol. Additionally, it can undergo oxidation reactions, especially when exposed to strong oxidizing agents .
Common Reagents and Conditions:
Hydrolysis: Strong acids (e.g., hydrochloric acid) or bases (e.g., sodium hydroxide) under reflux conditions.
Oxidation: Strong oxidizing agents (e.g., potassium permanganate) under controlled temperature and pH conditions.
Major Products Formed:
Hydrolysis: Sulfosuccinic acid and 2-ethylhexanol.
Oxidation: Various oxidized derivatives of this compound, depending on the specific conditions and reagents used.
Scientific Research Applications
Clinical Applications
Constipation Management
Docusate sodium is primarily utilized in the treatment of constipation, especially in patients with conditions that predispose them to hard stools. It acts as a stool softener by reducing the surface tension of stool, allowing water to penetrate and soften it.
Clinical Studies
- Historical Studies :
-
Recent Evaluations :
- A multi-site study compared this compound with psyllium in 170 adults and found that psyllium was more effective in increasing bowel movement frequency and stool output .
- A randomized trial involving hospice patients demonstrated no significant benefit of docusate when combined with sennosides compared to placebo .
| Study Year | Sample Size | Treatment | Outcome |
|---|---|---|---|
| 1956 | 74 | This compound vs. placebo | Improved stool consistency |
| 1968 | 34 | This compound vs. placebo | Increased bowel movements |
| 2020 | 170 | This compound vs. psyllium | Psyllium more effective |
| 2023 | 74 | Docusate + sennosides vs. placebo + sennosides | No significant difference |
Regulatory Status
This compound is recognized by various health authorities:
- U.S. FDA : Approved as an over-the-counter medication for constipation but lacks definitive evidence for its efficacy in all patient populations .
- EU Regulations : Registered under REACH since June 2012, indicating its compliance with safety standards for various industrial applications .
Industrial Applications
Beyond its medical use, this compound has several industrial applications:
Mechanism of Action
Docusate sodium works by lowering the surface tension of hard, dry stools, allowing water and fats to penetrate and soften the stool. This action reduces straining during bowel movements. The compound acts on the intestinal mucosa, promoting the incorporation of water and lipids into the stool, making it easier to pass . The molecular targets and pathways involved include the modulation of water and electrolyte transport across the intestinal epithelium .
Comparison with Similar Compounds
Polyethylene glycol 3350: Another commonly used laxative that works by retaining water in the stool, resulting in softer stools and easier bowel movements.
Lactulose: A synthetic sugar used to treat constipation by drawing water into the bowel, softening the stool.
Magnesium hydroxide: An osmotic laxative that increases water in the intestines, promoting bowel movements.
Uniqueness of Docusate Sodium: this compound is unique in its dual role as a stool softener and an anionic surfactant. Unlike other laxatives that primarily work by osmosis or increasing bowel motility, this compound specifically reduces the surface tension of stools, facilitating the incorporation of water and fats. This unique mechanism makes it particularly effective for patients who need to avoid straining during bowel movements .
Biological Activity
Docusate sodium, a widely used surfactant and stool softener, exhibits various biological activities that extend beyond its primary application in gastrointestinal health. This article reviews the biological activity of this compound, including its pharmacological effects, toxicity, and potential therapeutic applications based on diverse research findings.
This compound is the sodium salt of docusate, a compound derived from sulfosuccinic acid. Its chemical formula is . As an anionic surfactant, this compound reduces surface tension, facilitating the penetration of water into fecal matter, thus softening stools and promoting bowel movements .
Mechanism of Action:
- Surfactant Activity: this compound acts by increasing the wetting ability of stool, allowing water and lipids to mix with fecal matter.
- Emulsification: It emulsifies fats in the gastrointestinal tract, enhancing the absorption of nutrients and medications .
Laxative and Stool Softening Effects
This compound is primarily known for its use as a laxative. Clinical studies have demonstrated its effectiveness in:
- Reducing constipation in patients with limited mobility or those undergoing opioid therapy.
- Preventing straining during bowel movements, which can be crucial for post-operative patients .
Antimicrobial Activity
Recent studies have explored the antimicrobial properties of this compound:
- In Vitro Studies: this compound has been shown to enhance the efficacy of certain antibiotics against resistant strains of bacteria by disrupting biofilms and increasing drug permeability .
- Case Study: A study involving Candida neoformans showed that docusate-based ionic liquids improved the solubility and bioavailability of anthelmintic drugs, suggesting potential applications in antifungal therapies .
Acute Toxicity
This compound exhibits low acute toxicity. In animal studies, the median lethal dose (LD50) was reported to be greater than 2100 mg/kg body weight in rats . Other studies reported values ranging from 3080 to 4200 mg/kg, indicating a favorable safety profile for oral administration.
Genotoxicity and Reproductive Toxicity
Research on genotoxicity has yielded inconclusive results. In vitro studies indicated that while this compound did not induce mutations in Salmonella typhimurium, it did cause chromosomal aberrations in Chinese hamster ovary cells under specific conditions . Regarding reproductive toxicity, this compound has been classified as a Category 3 hazardous substance with possible risks to reproduction based on animal studies showing developmental toxicity at high doses .
Case Studies and Research Findings
- Veterinary Applications: this compound has been evaluated for its potential in veterinary medicine as a surfactant in drug delivery systems. Its ability to enhance drug permeability across biological membranes is being researched for treating infections in livestock .
- Clinical Trials: A clinical trial assessing the efficacy of this compound combined with other laxatives showed improved outcomes in patients with chronic constipation compared to monotherapy .
Summary Table of Biological Activities
Q & A
Basic Research Questions
Q. What standardized methodologies are recommended for characterizing the physicochemical properties of docusate sodium (e.g., solubility, stability) in preclinical studies?
To determine physicochemical properties, employ techniques such as high-performance liquid chromatography (HPLC) for purity assessment, differential scanning calorimetry (DSC) for thermal stability, and dynamic light scattering (DLS) for colloidal behavior in aqueous solutions. Ensure reproducibility by documenting environmental conditions (e.g., pH, temperature) and validating instruments against reference standards .
Q. How should researchers design in vitro experiments to evaluate this compound’s surfactant efficacy while minimizing confounding variables?
Use controlled cell culture models (e.g., Caco-2 monolayers) to assess permeability enhancement. Standardize variables such as buffer composition, incubation time, and surfactant concentration. Include positive controls (e.g., Tween 80) and negative controls (vehicle-only) to validate assay sensitivity. Replicate experiments across multiple batches to account for biological variability .
Q. What are the best practices for documenting the synthesis and characterization of this compound in compliance with FAIR data principles?
Provide detailed protocols for synthesis (e.g., reaction stoichiometry, purification steps) and characterization (e.g., NMR, FTIR spectra) in supplementary materials. Use structured metadata templates to describe experimental conditions, equipment calibration, and raw data repositories. Adhere to journal-specific guidelines for reporting novel compounds .
Advanced Research Questions
Q. How can researchers resolve contradictions between in vitro and in vivo toxicity data for this compound?
Conduct interspecies pharmacokinetic studies to identify metabolic differences. Use computational modeling (e.g., physiologically based pharmacokinetics, PBPK) to extrapolate in vitro toxicity thresholds to in vivo scenarios. Validate findings with histopathological analyses in animal models and correlate with clinical case reports .
Q. What statistical methods are appropriate for analyzing dose-response relationships in this compound studies with non-linear kinetics?
Apply non-parametric approaches (e.g., Hill equation modeling) or Bayesian hierarchical models to accommodate variability. Use bootstrapping to estimate confidence intervals for EC50 values. For longitudinal data, mixed-effects models can account for repeated measures and missing data .
Q. How can researchers optimize experimental designs to investigate this compound’s mechanism of action in complex biological systems (e.g., gut microbiome interactions)?
Integrate multi-omics approaches: metagenomics for microbiome profiling, transcriptomics for host-pathway analysis, and metabolomics for surfactant byproduct identification. Use gnotobiotic animal models to isolate microbial contributions. Employ machine learning to identify covarying factors in high-dimensional datasets .
Q. What strategies mitigate batch-to-batch variability in this compound formulations during preclinical testing?
Implement quality-by-design (QbD) principles: define critical material attributes (CMAs) and process parameters (CPPs) during formulation. Use design of experiments (DoE) to optimize excipient ratios. Characterize batches using orthogonal methods (e.g., X-ray diffraction for crystallinity, MALDI-TOF for polymer consistency) .
Q. How should researchers address discrepancies between published solubility data for this compound in polar vs. non-polar solvents?
Replicate experiments using standardized solvents (e.g., USP-grade) and controlled humidity/temperature. Compare results with computational solubility predictions (e.g., COSMO-RS). Publish raw data with metadata on solvent lot numbers and equilibration times to facilitate cross-study validation .
Q. Methodological Considerations
- Data Management : Archive raw datasets in FAIR-compliant repositories (e.g., Zenodo, Figshare) with DOI assignment. Include README files detailing experimental protocols and software versions .
- Ethical Reporting : Disclose conflicts of interest (e.g., funding sources) and adhere to ARRIVE guidelines for animal studies .
- Interdisciplinary Collaboration : Engage pharmacologists, computational biologists, and material scientists to address multifaceted research questions .
Properties
IUPAC Name |
sodium;1,4-bis(2-ethylhexoxy)-1,4-dioxobutane-2-sulfonate | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C20H38O7S.Na/c1-5-9-11-16(7-3)14-26-19(21)13-18(28(23,24)25)20(22)27-15-17(8-4)12-10-6-2;/h16-18H,5-15H2,1-4H3,(H,23,24,25);/q;+1/p-1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
APSBXTVYXVQYAB-UHFFFAOYSA-M | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CCCCC(CC)COC(=O)CC(C(=O)OCC(CC)CCCC)S(=O)(=O)[O-].[Na+] | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C20H37NaO7S | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
10041-19-7 (Parent) | |
| Record name | Docusate sodium [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000577117 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID8022959 | |
| Record name | Docusate sodium | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8022959 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
444.6 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Commercially available in rolls of very thin wax-like solid; as solution (50-75%) in various solvents; [Merck Index] White waxy solid; [MSDSonline] | |
| Record name | Docusate sodium | |
| Source | Haz-Map, Information on Hazardous Chemicals and Occupational Diseases | |
| URL | https://haz-map.com/Agents/3967 | |
| Description | Haz-Map® is an occupational health database designed for health and safety professionals and for consumers seeking information about the adverse effects of workplace exposures to chemical and biological agents. | |
| Explanation | Copyright (c) 2022 Haz-Map(R). All rights reserved. Unless otherwise indicated, all materials from Haz-Map are copyrighted by Haz-Map(R). No part of these materials, either text or image may be used for any purpose other than for personal use. Therefore, reproduction, modification, storage in a retrieval system or retransmission, in any form or by any means, electronic, mechanical or otherwise, for reasons other than personal use, is strictly prohibited without prior written permission. | |
Solubility |
SOL IN WATER (G/L): 15 (25 °C), 23 (40 °C), 30 (50 °C), 55 (70 °C); SOL IN CARBON TETRACHLORIDE, PETROLEUM ETHER, NAPHTHA, XYLENE, DIBUTYL PHTHALATE, LIQUID PETROLATUM, ACETONE, ALCOHOL, VEGETABLE OILS; VERY SOL IN WATER-MISCIBLE ORGANIC SOLVENTS, FREELY SOL IN GLYCERIN, Water Solubility = 71000 mg/L @ 25 °C | |
| Record name | BIS(2-ETHYLHEXYL) SODIUM SULFOSUCCINATE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3065 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Mechanism of Action |
In vitro studies suggest that these salts of dioctylsulfosuccinic acid lower the surface tension of the stool to permit water and lipids to enter more readily and thus soften the feces. ... More recent evidence indicates that they may stimulate the secretion of water and electrolytes on contact with the mucosa., Reduce surface film tension of interfacing liquid contents of the bowel, promoting permeation of additional liquid into the stool to form a softer mass. /Laxatives/ | |
| Record name | BIS(2-ETHYLHEXYL) SODIUM SULFOSUCCINATE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3065 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
WHITE, WAX-LIKE SOLID | |
CAS No. |
577-11-7 | |
| Record name | Docusate sodium [USAN:USP:INN:BAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0000577117 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Docusate sodium | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID8022959 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Docusate sodium | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.008.553 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DOCUSATE SODIUM | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/F05Q2T2JA0 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | BIS(2-ETHYLHEXYL) SODIUM SULFOSUCCINATE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3065 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















