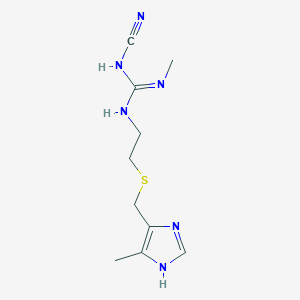
Cimetidine
Overview
Description
Cimetidine is a histamine H2 receptor antagonist that inhibits stomach acid production. It is primarily used to treat conditions such as heartburn, peptic ulcers, and gastroesophageal reflux disease (GERD) . This compound was developed in 1971 and came into commercial use in 1977 .
Preparation Methods
Synthetic Routes and Reaction Conditions: Cimetidine can be synthesized through various methods. One common method involves converting (5-methyl-1H-imidazole-4-yl) methanol into nitrate ester, which then reacts with N-cyano-N’-methyl-N’-mercaptoethylguanidine ether to produce this compound . Another method involves condensing 2-(4-methylimidazol-4-yl) methylthioethylamine hydrochloride with carbon disulfide in the presence of a base and a desulfurization reagent to form an intermediate, which is then reacted with monomethylamine and cyanamide to yield this compound .
Industrial Production Methods: Industrial production of this compound often focuses on optimizing yield and minimizing environmental impact. For example, some methods avoid generating volatile methyl mercaptan, which is harmful to the environment .
Chemical Reactions Analysis
Types of Reactions: Cimetidine undergoes various chemical reactions, including oxidation, reduction, and substitution. It is known to inhibit many isoenzymes of the hepatic cytochrome P450 enzyme system .
Common Reagents and Conditions: Common reagents used in the synthesis of this compound include nitrate esters, carbon disulfide, monomethylamine, and cyanamide . Reaction conditions typically involve mild temperatures and the presence of desulfurization reagents to ensure high yield and minimal byproducts .
Major Products Formed: The major products formed from the reactions involving this compound include this compound sulfoxide and hydroxythis compound .
Scientific Research Applications
Cimetidine has a wide range of scientific research applications. In medicine, it is used to manage conditions like GERD, peptic ulcer disease, and indigestion . It also has potential anticancer activity by interfering with histamine-mediated immunomodulation . In chemistry, this compound is used to study reaction kinetics and photochemistry . Additionally, it has been explored for its antiandrogenic properties in treating conditions like hirsutism and androgenetic alopecia .
Mechanism of Action
Cimetidine exerts its effects by competitively inhibiting the action of histamine at the histamine H2 receptors on the basolateral membrane of gastric parietal cells . This inhibition reduces gastric acid secretion, gastric volume, and acidity . This compound also blocks the activity of cytochrome P450 enzymes, which may explain its use in neoadjuvant therapy .
Comparison with Similar Compounds
Cimetidine is often compared with other H2 receptor antagonists like ranitidine and proton pump inhibitors like omeprazole and pantoprazole . While all these compounds reduce gastric acid secretion, this compound is unique in its ability to inhibit cytochrome P450 enzymes . This property makes it distinct from other similar compounds, which may have fewer drug interactions and adverse effects .
List of Similar Compounds:- Ranitidine
- Omeprazole
- Pantoprazole
- Famotidine
Biological Activity
Cimetidine, a histamine H2-receptor antagonist, has been widely used for the treatment of peptic ulcers and gastroesophageal reflux disease. However, recent research has uncovered its diverse biological activities beyond acid suppression, particularly in immunomodulation and potential anti-cancer effects. This article explores the biological activity of this compound, supported by data tables, case studies, and detailed research findings.
This compound primarily functions by inhibiting the action of histamine at H2 receptors located in the gastric mucosa. This inhibition reduces gastric acid secretion, leading to therapeutic effects in acid-related disorders . However, its biological activity extends into the modulation of immune responses and interactions with various cellular pathways.
Immunomodulatory Effects
Recent studies have highlighted this compound's role as an immunomodulatory agent. It has been shown to enhance T cell populations (CD3+, CD4+, and CD8+) and M1 macrophages in tumor microenvironments, which may augment immune responses against tumors . This is particularly significant in cancer therapy, where this compound has been observed to interact with immune checkpoint inhibitors like anti-PD-1 and anti-PD-L1.
Case Study: this compound in Colon Cancer
A study investigating this compound's effects on colon cancer demonstrated that it could reverse the decrease in circulating and tumor-associated neutrophils induced by anti-PD-L1 therapy. This suggests that this compound may reduce the sensitivity of certain tumors to immunotherapies . The findings indicate that while this compound can enhance immune cell populations, it may also attenuate the effectiveness of some cancer treatments.
Table 1: Summary of this compound's Biological Activities
Clinical Implications
This compound's ability to modulate immune responses has led to its exploration as an adjunct therapy in cancer treatment. Clinical studies suggest that it may improve outcomes when used alongside traditional chemotherapeutics. For example, this compound has been shown to increase the plasma concentration of drugs like epirubicin, potentially enhancing their efficacy .
Research Findings
In a comprehensive review of pre-clinical and clinical data, this compound was found to exhibit anti-cancer properties across various models. The mechanisms proposed include:
- Histamine Modulation : Altering histamine levels can influence tumor growth dynamics.
- Immune System Activation : Enhancing T cell activity and macrophage function can lead to improved anti-tumor responses.
- Synergistic Effects with Chemotherapy : this compound may potentiate the effects of certain chemotherapeutic agents .
Safety and Side Effects
While this compound is generally well-tolerated, its immunomodulatory effects necessitate caution in specific patient populations, particularly those undergoing immunotherapy. The potential for altered drug metabolism due to cytochrome P450 inhibition also raises concerns about drug interactions .
Table 2: Reported Side Effects of this compound
| Side Effect | Incidence |
|---|---|
| Gastrointestinal disturbances | Common |
| Central nervous system effects | Rare but possible |
| Drug interactions | Significant with CYP450 substrates |
Q & A
Q. What experimental designs are recommended for studying cimetidine’s antiandrogenic effects in preclinical models?
Answer: Preclinical studies on this compound’s antiandrogenic effects (e.g., gynecomastia, prolactin elevation) should employ multi-group designs with dose-dependent administration. For example, a study using pregnant albino rats divided into control and experimental groups (e.g., 5 g/day vs. lower doses) can assess offspring teratogenicity via behavioral tests like the T-maze . Hormonal assays (prolactin, estradiol) and histopathological analysis of mammary tissue are critical. Note that dose selection must account for species-specific metabolic differences and validate results with human cell lines (e.g., androgen receptor binding assays) .
Q. How can contradictory findings on this compound’s antitumor efficacy be reconciled in gastrointestinal cancer research?
Answer: Discrepancies arise from variations in tumor models and molecular targets. For instance, this compound inhibits E-selectin expression in TNF-α-stimulated endothelial cells (IC₅₀: ~42 nM) via RT-PCR and flow cytometry , but shows no effect on cisplatin cytotoxicity in OCT2-high ovarian cancer cells . Researchers should standardize models (e.g., syngeneic vs. xenograft), validate target engagement (e.g., H2 receptor vs. immune modulation), and use transcriptomic profiling to identify context-dependent pathways .
Q. What methodologies are optimal for analyzing this compound’s pharmacokinetic interactions with CYP450 substrates?
Answer: Use in vitro hepatic microsome assays to quantify CYP450 inhibition (e.g., CYP3A4, CYP2D6) and validate with clinical pharmacokinetic studies. For example, this compound prolongs diazepam half-life by 46% via competitive CYP3A4 inhibition . High-performance liquid chromatography (HPLC) with validated standard curves (e.g., linear range: 0.05–10 µg/mL) ensures precise measurement of drug concentrations in plasma . Physiologically based pharmacokinetic (PBPK) modeling further predicts interactions in populations with renal/hepatic impairment .
Q. How should researchers address this compound’s environmental persistence in ecotoxicology studies?
Answer: this compound’s high soil mobility (Koc = 39) and pH-dependent speciation (pKa = 6.8) necessitate studies using HPLC-UV or LC-MS to quantify photodegradation products . Laser flash photolysis identifies reactive moieties (e.g., imidazole ring oxidation by singlet oxygen, k = 3.3 × 10⁶ M⁻¹s⁻¹ at pH 4) . Field studies in river water should correlate degradation rates with dissolved organic carbon and sunlight intensity .
Q. What statistical approaches resolve conflicting data on this compound’s peri-operative benefits in cancer?
Answer: Meta-analyses of randomized trials (e.g., gastric cancer survival studies ) should apply fixed-effects models to account for heterogeneity in dosing (e.g., 800 mg/day vs. 400 mg/day) and outcome measures (e.g., recurrence vs. metastasis). Subgroup analyses stratified by tumor stage (e.g., TNM classification) and adjuvant therapies (e.g., 5-FU) improve specificity. Bayesian methods can weigh evidence from preclinical (e.g., 3LL cell migration assays ) and clinical datasets .
Q. How can researchers optimize solubility studies for this compound in formulation development?
Answer: Use the NRTL-SAC model in Aspen Properties to predict solubility in mixed solvents (e.g., ethanol-water) . Validate experimentally via UV-vis spectrophotometry (low solubility) or HPLC (high sensitivity) against USP reference standards . Report solubility as mean ± SD with one-way ANOVA and Bonferroni correction for multiple comparisons (α = 0.05) .
Q. What protocols mitigate bias in assessing this compound’s reproductive toxicity?
Answer: Follow OECD guidelines for teratogenicity studies, including blinded histopathology reviews and litter-based analysis to control for maternal effects . Measure fetal serum vitamin B12 (linked to long-term H2 blockade ) and use sham-operated controls to distinguish drug effects from surgical stress. Confounding factors like strain-specific susceptibility (e.g., albino rats vs. C57BL/6 mice) must be documented .
Q. How do OCT2 inhibition assays inform this compound’s role in drug-disease interactions?
Answer: Use HEK293 cells transfected with human OCT2 to quantify inhibition constants (e.g., this compound Ki = 15 µM ). In diabetic rat models, compare metformin pharmacokinetics with/without this compound co-administration, analyzing plasma via LC-MS/MS. PBPK models incorporating renal OCT2 expression levels predict clinical relevance .
Q. Basic vs. Advanced Research Focus
- Basic: Mechanisms (H2 receptor antagonism, CYP450 inhibition), standard PK/PD methods.
- Advanced: Tumor microenvironment modulation, environmental fate, systems pharmacology.
Properties
IUPAC Name |
1-cyano-2-methyl-3-[2-[(5-methyl-1H-imidazol-4-yl)methylsulfanyl]ethyl]guanidine | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C10H16N6S/c1-8-9(16-7-15-8)5-17-4-3-13-10(12-2)14-6-11/h7H,3-5H2,1-2H3,(H,15,16)(H2,12,13,14) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
AQIXAKUUQRKLND-UHFFFAOYSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1=C(N=CN1)CSCCNC(=NC)NC#N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C10H16N6S | |
| Record name | CIMETIDINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20034 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID4020329 | |
| Record name | Cimetidine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020329 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
252.34 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Physical Description |
Cimetidine appears as white crystals with a slight sulfur-mercaptan odor. (NTP, 1992), Solid | |
| Record name | CIMETIDINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20034 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Cimetidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014644 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Solubility |
5 mg/mL at 68 °F (NTP, 1992), IN WATER AT 37 °C: 1.14%; SOLUBILITY INCR BY DIL HYDROCHLORIC ACID, Soluble in alcohol, 8.16e-01 g/L | |
| Record name | CIMETIDINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20034 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Cimetidine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00501 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CIMETIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3917 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cimetidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014644 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Mechanism of Action |
Cimetidine binds to an H2-receptor located on the basolateral membrane of the gastric parietal cell, blocking histamine effects. This competitive inhibition results in reduced gastric acid secretion and a reduction in gastric volume and acidity., H2 antagonists inhibit gastric acid secretion elicited by histamine & other H2 agonists in a dose-dependent, competitive manner; the degree of inhibition parallels the concentration of the drug in plasma over a wide range. The H2 antagonists also inhibit acid secretion elicited by gastrin &, to a lesser extent, by muscarinic agonists. Importantly, these drugs inhibit basal (fasting) & nocturnal acid secretion & that stimulated by food, sham feeding, fundic distention, & various pharmacological agents; this property reflects the vital role of histamine in mediating the effects of diverse stimuli. The H2 antagonists reduce both the volume of gastric juice secreted & its H+ concentration. The output of pepsin, which is secreted by the chief cells of gastric glands (mainly under cholinergic control), generally falls in parallel with the reduction in volume of gastric juice. /H2 antagonists/, Cimetidine blocks H2-receptors, which in part are responsible for the inflammatory response, in the cutaneous blood vessels of humans., The effects of cimetidine, omeprazole and atropine sulfate on the healing of acetic acid-induced gastric ulcers in rats with limited food intake time (9:00-10:00 am and 5:00-6:00 pm) were evaluated 15 days after the acid injection. Oral repeated admin of cimetidine (25-100 mg/kg twice daily) or omeprazole (10-50 mg/kg once daily) dose dependently accelerated ulcer healing. ... A single oral admin of omeprazole (50 mg/kg) or cimetidine (100 mg/kg) resulted in potent and long-lasting anti-acid secretory and gastrin-releasing actions. The degree and duration of anti-acid secretion by atropine sulfate were equal to those of cimetidine, but the elevation of gastrin release by atropine sulfate was weak and temporary. These results indicate that the gastric ulcers of rats with a limited food intake time are useful for evaluating the healing effects of cimetidine and omeprazole on gastric ulcers. In addition, the effects of both drugs may be related to the incr gastrin release rather than to the reduced acid secretion., Both KB-5492, a new anti-ulcer agent, and cimetidine, admin po at 25-200 mg/kg, dose-dependently prevented cysteamine (400 mg/kg, sc)-induced duodenal ulcers in rats with ED50 values of 63 and 40 mg/kg, respectively. Anti-ulcer doses of cimetidine, but not KB-5492, inhibited gastric acid hypersecretion induced by cysteamine (400 mg/kg, sc). In contrast, anti-ulcer doses of KB-5492, but not cimetidine, incr duodenal HC03- secretion in normal anesthetized rats. These findings suggest that KB-5492 prevents cysteamine-induced duodenal ulcers by stimulating duodenal HC03- secretion, whereas cimetidine does so by inhibiting cysteamine-induced gastric acid hypersecretion. | |
| Record name | Cimetidine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00501 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CIMETIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3917 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Color/Form |
Crystals | |
CAS No. |
51481-61-9 | |
| Record name | CIMETIDINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20034 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Cimetidine | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=51481-61-9 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Cimetidine [USAN:USP:INN:BAN:JAN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0051481619 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Cimetidine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00501 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | cimetidine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=757428 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | cimetidine | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=335308 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Cimetidine | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID4020329 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | Cimetidine | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.052.012 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | CIMETIDINE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/80061L1WGD | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | CIMETIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3917 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cimetidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014644 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Melting Point |
284 to 290 °F (NTP, 1992), 141-143 °C, 142 °C | |
| Record name | CIMETIDINE | |
| Source | CAMEO Chemicals | |
| URL | https://cameochemicals.noaa.gov/chemical/20034 | |
| Description | CAMEO Chemicals is a chemical database designed for people who are involved in hazardous material incident response and planning. CAMEO Chemicals contains a library with thousands of datasheets containing response-related information and recommendations for hazardous materials that are commonly transported, used, or stored in the United States. CAMEO Chemicals was developed by the National Oceanic and Atmospheric Administration's Office of Response and Restoration in partnership with the Environmental Protection Agency's Office of Emergency Management. | |
| Explanation | CAMEO Chemicals and all other CAMEO products are available at no charge to those organizations and individuals (recipients) responsible for the safe handling of chemicals. However, some of the chemical data itself is subject to the copyright restrictions of the companies or organizations that provided the data. | |
| Record name | Cimetidine | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB00501 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | CIMETIDINE | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/3917 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
| Record name | Cimetidine | |
| Source | Human Metabolome Database (HMDB) | |
| URL | http://www.hmdb.ca/metabolites/HMDB0014644 | |
| Description | The Human Metabolome Database (HMDB) is a freely available electronic database containing detailed information about small molecule metabolites found in the human body. | |
| Explanation | HMDB is offered to the public as a freely available resource. Use and re-distribution of the data, in whole or in part, for commercial purposes requires explicit permission of the authors and explicit acknowledgment of the source material (HMDB) and the original publication (see the HMDB citing page). We ask that users who download significant portions of the database cite the HMDB paper in any resulting publications. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















