Perampanel
Overview
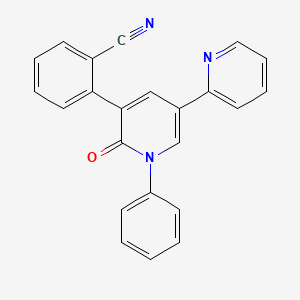
Description
Perampanel is a novel antiepileptic drug that functions as a selective, non-competitive antagonist of the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor. It is primarily used as an adjunctive therapy for the treatment of partial-onset seizures and primary generalized tonic-clonic seizures in patients with epilepsy . This compound is marketed under the brand name Fycompa and has been approved in over 35 countries .
Preparation Methods
The synthesis of perampanel involves several steps, starting with the protection of the hydroxyl group using an alkali-resistant protecting group such as dihydropyranyl or trityl . The intermediate is then subjected to various reaction conditions, including the use of p-toluenesulfonic acid monohydrate and dihydropyran, to obtain the final product . Industrial production methods typically involve high-performance liquid chromatography for the quantification and validation of this compound in plasma samples .
Chemical Reactions Analysis
Primary Metabolic Pathways
Perampanel is metabolized through three principal routes (Figure 3 in ):
-
Ring hydroxylation : Produces hydroxylated metabolites (M1, M3, M4, M5) and their glucuronide conjugates.
-
Pyridine ring rearrangement : Forms the carboxylic acid metabolite M2.
-
Dihydrodiol formation : Generates metabolite M7 via epoxide intermediates.
Key Enzymes :
Metabolite Profiles and Activity
| Metabolite | Structural Modification | Relative Potency (vs. This compound) | Contribution to Efficacy |
|---|---|---|---|
| M1 | Hydroxylation at pyridine ring | 44-fold weaker | Negligible |
| M2 | Pyridine ring rearrangement | No activity up to 10 µM | None |
| M3 | Hydroxylation at biphenyl moiety | 3.0-fold weaker | Negligible |
| M4 | Hydroxylation at benzonitrile group | 3.8-fold weaker | Negligible |
| M5 | Hydroxylation at phenyl group | 7.7-fold weaker | Negligible |
| M7 | Dihydrodiol formation | 27-fold weaker | Negligible |
Data from rat, monkey, and human studies .
Reaction Kinetics and Species Variability
-
Humans : Metabolites account for <10% of circulating drug plasma concentrations .
-
Rats/Monkeys : Higher metabolite levels observed, but parent compound remains dominant .
-
Half-Life : Terminal half-life () in humans is 105 hours but shortens to 25–70 hours with CYP3A4 inducers (e.g., carbamazepine) .
Excretion Pathways
In Vitro Reactivity
-
Solubility : Poor in neutral/alkaline aqueous solutions () but highly soluble in acidic conditions () due to protonation () .
-
Stability : No significant degradation under physiological conditions; photostability confirmed in formulation studies .
Drug-Drug Interactions
-
CYP3A4 Inducers : Reduce this compound plasma levels (e.g., carbamazepine, phenytoin) .
-
CYP3A4 Inhibitors : Increase exposure (e.g., ketoconazole) .
Toxicological Byproducts
Scientific Research Applications
Clinical Applications
1. Focal-Onset Seizures
- Perampanel is approved as an adjunctive treatment for adults and children aged 12 years and older with focal-onset seizures (FOS). Clinical studies show that patients receiving this compound as an add-on therapy experience a higher responder rate compared to those on placebo. For instance, a study reported a 50% responder rate of 35.3% for patients on 8 mg/day of this compound versus 19.3% for placebo .
2. Generalized Tonic-Clonic Seizures
- In addition to FOS, this compound is also indicated for generalized tonic-clonic seizures (GTCS). The efficacy of this compound in this context has been validated through extensive clinical trials, indicating its broad-spectrum anti-seizure activity .
3. Pediatric Use
- Recent findings suggest that this compound is effective and safe for pediatric patients aged 4 to <12 years. A long-term study highlighted that while younger children may discontinue treatment due to inadequate effectiveness, older children often do so due to adverse effects . The median percent reduction in seizure frequency was significant, indicating its potential as a reliable treatment option in younger populations .
Efficacy Data
A summary of key efficacy outcomes from various studies is presented below:
Safety Profile
This compound's safety profile has been extensively documented across various studies. The most common adverse effects reported include dizziness, somnolence, and irritability. The incidence of these effects varies based on age and dosage but remains consistent with the known safety profile established in clinical trials .
Case Studies
Case Study 1: Efficacy in Adults
A cohort study involving adults with refractory epilepsy demonstrated that adjunctive treatment with this compound resulted in a significant reduction in seizure frequency over a treatment period of six months, achieving a responder rate of approximately 70% .
Case Study 2: Pediatric Efficacy
In pediatric patients diagnosed with Dravet syndrome, this compound showed promising results, particularly among those aged eight years or younger, where higher efficacy rates were observed compared to older patients . This highlights the importance of tailored treatment approaches based on age and specific epilepsy syndromes.
Mechanism of Action
Perampanel exerts its effects by selectively inhibiting the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor, which is involved in excitatory synaptic transmission in the brain . By blocking the activity of this receptor, this compound reduces neuronal excitation and helps control seizures . The exact molecular targets and pathways involved in this mechanism are still being studied, but it is known that this compound has a prolonged terminal half-life and is highly bound to plasma proteins .
Comparison with Similar Compounds
Perampanel is unique among antiepileptic drugs due to its selective inhibition of the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor . Similar compounds include other antiepileptic drugs that target different receptors, such as levetiracetam and lamotrigine . Unlike these drugs, this compound offers the convenience of once-daily administration and has been shown to provide sustained seizure control in patients with drug-resistant epilepsy .
Biological Activity
Perampanel is a non-competitive antagonist of the AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) glutamate receptor, primarily utilized in the management of epilepsy, particularly for partial-onset seizures. This article explores the biological activity of this compound, including its pharmacodynamics, clinical efficacy, safety profile, and real-world performance based on diverse studies.
This compound works by inhibiting the AMPA receptors in the central nervous system, which are crucial mediators of excitatory neurotransmission. This inhibition leads to a decrease in neuronal excitability, thereby reducing seizure activity. The exact mechanism of action in the context of seizure reduction remains partially understood but is linked to its ability to modulate glutamatergic transmission .
Pharmacokinetics
Absorption and Distribution:
- This compound is rapidly and completely absorbed after oral administration.
- It exhibits a high volume of distribution, although specific values are not quantified.
- Approximately 95-96% of this compound is bound to plasma proteins, primarily α1-acid glycoprotein and albumin .
Metabolism:
- The drug is extensively metabolized by cytochrome P450 enzymes (CYP3A4 and CYP3A5), followed by glucuronidation.
- The elimination half-life ranges from 52 to 129 hours, allowing for once-daily dosing .
Elimination:
Randomized Controlled Trials
In pivotal phase III trials, this compound demonstrated significant efficacy in reducing seizure frequency among patients with refractory partial-onset seizures:
- Study 305 Findings:
Real-World Evidence
A retrospective analysis involving over 1700 patients indicated favorable retention rates and sustained efficacy:
- Retention Rates:
- At 24 months, approximately 48.1% of patients continued treatment with this compound.
- Seizure Frequency Reduction:
Safety Profile
This compound is generally well-tolerated; however, treatment-emergent adverse events (TEAEs) have been documented:
- Common TEAEs include dizziness, somnolence, fatigue, and headache.
- Discontinuation due to adverse events occurred in approximately 22.9% of patients .
Case Study Analysis
-
Patient Cohort Characteristics:
- A study involving 1121 patients revealed a median exposure duration to this compound of 16.6 months with an overall mean daily dose of 5.7 mg.
- Seizure Freedom Rates:
Summary Table of Key Findings
| Parameter | Value/Description |
|---|---|
| Mechanism | Non-competitive AMPA receptor antagonist |
| Absorption | Rapid and complete after oral administration |
| Protein Binding | 95-96% bound to plasma proteins |
| Metabolism | CYP3A4/CYP3A5 mediated |
| Half-Life | 52 to 129 hours |
| Retention Rate (24 months) | ~48.1% |
| Median Seizure Frequency Reduction (12 months) | 75% |
| Common TEAEs | Dizziness, somnolence, fatigue |
Q & A
Basic Research Questions
Q. What methodological considerations are critical when designing randomized controlled trials (RCTs) for perampanel in epilepsy?
- RCTs should account for CYP3A4-inducing antiepileptic drugs (AEDs) , which reduce this compound plasma concentrations by up to 66%, necessitating dose adjustments .
- Use stratified randomization to balance enzyme-inducing AED usage across study arms.
- Include long-term extension phases (≥52 weeks) to assess sustained efficacy and delayed adverse events (AEs), as 21% of discontinuations occur during extensions due to AEs like dizziness and aggression .
- Address funding bias: Only 5 industry-sponsored RCTs exist, limiting generalizability; consider integrating real-world data (RWD) for validation .
Q. How should researchers evaluate this compound’s hepatotoxicity risk compared to older AEDs?
- Use in vitro models (e.g., HepG2 cells) to compare this compound with carbamazepine, which increases ALT/AST levels. This compound shows no significant hepatotoxicity in preclinical or clinical studies .
- Prioritize post-marketing surveillance for rare hepatotoxic events, as clinical trials lack elderly/special populations and long-term data .
Q. What pharmacokinetic (PK) parameters must be modeled for this compound in diverse populations?
- Apparent clearance (CL/F) varies: 0.668 L/hr (adults without enzyme inducers) vs. 0.682 L/hr (adolescents). Incorporate covariates like age, CYP3A4 inducer use, and albumin levels .
- Use a one-compartment model with linear elimination for PK/PD analyses, validated in pooled Phase III data .
Advanced Research Questions
Q. How can conflicting efficacy data between RCTs and real-world studies (e.g., responder rates) be reconciled?
- RCTs : 50% responder rates range from 22–38% for adjunctive therapy (8–12 mg/day) .
- Real-world studies : Higher variability (13–75%) due to unselected populations and flexible dosing .
- Apply meta-regression to adjust for confounders (e.g., prior AED failures, titration speed). Use RWD to identify subgroups with optimal response (e.g., patients on non-enzyme-inducing AEDs) .
Q. What statistical approaches are optimal for analyzing dose-dependent cognitive effects of this compound?
- Use longitudinal mixed-effects models to assess changes in global cognition T-scores (e.g., −1.0 ± 9.9 from baseline, p = 0.96 over 52 weeks) .
- Stratify by dose (4–12 mg/day) and monitor aggression/irritability via validated scales (e.g., Neuropsychiatric Inventory) .
Q. How should researchers design studies to evaluate this compound’s broad-spectrum potential beyond focal seizures?
- Include preclinical models of generalized tonic-clonic seizures (GTCS), where this compound reduces seizure frequency by 76.5% vs. placebo (p < 0.0001) .
- Conduct adaptive trials with Bayesian designs to assess efficacy in mixed seizure types (e.g., FOS + GTCS) .
Q. What methodologies address underrepresentation of elderly patients in this compound safety studies?
- Pooled analyses of elderly subgroups (≥65 years) show higher AE rates (dizziness: 45% vs. 29% in adults). Use propensity score matching to compare safety outcomes with younger cohorts .
- Implement fall-risk assessments (e.g., Tinetti Scale) during titration phases .
Q. Methodological Guidelines for Data Analysis
Q. How to conduct bioequivalence studies for generic this compound formulations?
- Use a crossover design with ≥28 subjects, 42-day washout, and 90% CI thresholds for AUC₀–₇₂ (92–104%) and Cₘₐₓ (90–108%) .
- Exclude outliers with pre-dose concentrations >5% of Cₘₐₓ to avoid carryover effects .
Q. What strategies mitigate bias in post-hoc analyses of this compound extension studies?
- Apply inverse probability weighting to address attrition bias (24% discontinuation in extensions) .
- Report median percent seizure reduction (e.g., −39.6% vs. +2.1% for placebo) with IQRs to capture skewed distributions .
Q. How to optimize PK/PD modeling for this compound in pediatric populations?
Properties
IUPAC Name |
2-(2-oxo-1-phenyl-5-pyridin-2-ylpyridin-3-yl)benzonitrile | |
|---|---|---|
| Details | Computed by Lexichem TK 2.7.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C23H15N3O/c24-15-17-8-4-5-11-20(17)21-14-18(22-12-6-7-13-25-22)16-26(23(21)27)19-9-2-1-3-10-19/h1-14,16H | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
PRMWGUBFXWROHD-UHFFFAOYSA-N | |
| Details | Computed by InChI 1.0.6 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
C1=CC=C(C=C1)N2C=C(C=C(C2=O)C3=CC=CC=C3C#N)C4=CC=CC=N4 | |
| Details | Computed by OEChem 2.3.0 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C23H15N3O | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID80191501 | |
| Record name | Perampanel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80191501 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
349.4 g/mol | |
| Details | Computed by PubChem 2.1 (PubChem release 2021.05.07) | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Practically insoluble in water. | |
| Details | From FDA label. | |
| Record name | Perampanel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08883 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
The exact mechanism of action of perampanel in seizures is not yet determined, but it is known that perampanel decreases neuronal excitation by non-competitive ihibition of the AMPA receptor. | |
| Record name | Perampanel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08883 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
CAS No. |
380917-97-5 | |
| Record name | Perampanel | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=380917-97-5 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Perampanel [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0380917975 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Perampanel | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08883 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Perampanel | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID80191501 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 2-(2-oxo-1-phenyl-5-pyridin-2-yl-1,2-dihydropyridin-3-yl)benzonitrile hydrate(4:3) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/information-on-chemicals | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | PERAMPANEL | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/H821664NPK | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Synthesis routes and methods I
Procedure details










Synthesis routes and methods II
Procedure details







Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.













