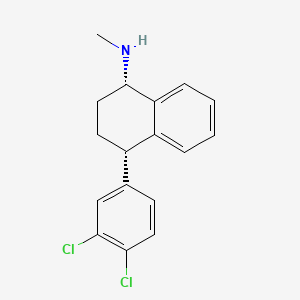
Sertraline
Overview
Description
Sertraline is a widely used antidepressant belonging to the class of selective serotonin reuptake inhibitors. It is primarily prescribed for the treatment of major depressive disorder, generalized anxiety disorder, panic disorder, social anxiety disorder, post-traumatic stress disorder, and obsessive-compulsive disorder . This compound works by increasing the levels of serotonin, a neurotransmitter, in the brain, which helps improve mood and reduce anxiety .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of sertraline involves several steps, starting from commercially available starting materials. One common synthetic route involves the reduction of a racemic tetralone precursor to yield a chiral alcohol, followed by oxidation to form an enantiopure ketone. This ketone is then subjected to reductive amination to produce this compound .
Industrial Production Methods: In industrial settings, the production of this compound often employs biocatalysis due to its high selectivity and efficiency. Enzymes such as ketoreductases are used to achieve the desired stereochemistry, and the process is optimized for large-scale production .
Chemical Reactions Analysis
Imine Formation and Reduction
-
Imine formation : A tetralone reacts with monomethylamine to form an imine intermediate.
-
Reduction : Hydrogenation using palladium catalysts reduces the imine to the tetrahydro-naphthalenamine core .
Resolution with Mandelic Acid
The diastereomeric salts of mandelic acid are resolved to isolate the desired (1S,4S)-enantiomer. This step ensures high enantiomeric purity (e.g., 99.8% in some methods) .
Optimized Process by Pfizer
Pfizer’s green chemistry approach streamlined the synthesis by:
-
Replacing multiple solvents (methylene chloride, THF, toluene) with ethanol.
-
Eliminating titanium tetrachloride and hazardous reagents.
-
Achieving a 2x higher yield with reduced raw material use (monomethylamine, tetralone) .
| Step | Key Reaction | Conditions/Solvents |
|---|---|---|
| Imine formation | Tetralone + monomethylamine → imine | Ethanol, reflux |
| Reduction | Hydrogenation of imine | Pd catalyst, H₂ gas |
| Resolution | Diastereomeric salt separation | Mandelic acid, pH adjustment |
Metabolic Pathways
This compound undergoes extensive hepatic metabolism via cytochrome P450 (CYP) enzymes and other pathways:
N-Demethylation
-
Primary pathway: CYP2B6 catalyzes the conversion of this compound to desmethylthis compound , a weaker active metabolite .
-
Secondary enzymes: CYP2C19, CYP3A4, and CYP2D6 contribute minimally .
Hydroxylation and Deamination
-
Hydroxylation: Forms α-hydroxy this compound via CYP3A4 and CYP2C19.
-
Deamination: Oxidative deamination by MAO-A/B or CYP3A4 generates α-hydroxy this compound ketone .
Glucuronidation
-
This compound undergoes glucuronidation via UGT1A3, UGT1A6, UGT2B4, and UGT2B7, forming this compound carbamoyl-O-glucuronide .
| Metabolic Step | Key Enzymes | Major Product |
|---|---|---|
| N-Demethylation | CYP2B6 (primary) | Desmethylthis compound |
| Hydroxylation | CYP3A4, CYP2C19 | α-Hydroxy this compound |
| Deamination | MAO-A/B, CYP3A4 | α-Hydroxy this compound ketone |
| Glucuronidation | UGT isoforms | This compound carbamoyl-O-glucuronide |
Drug-Drug Interactions
This compound modulates CYP enzymes, affecting co-administered drugs:
CYP2D6 Inhibition
-
This compound inhibits CYP2D6, increasing plasma concentrations of imipramine and desipramine (e.g., AUC fold-change: 1.25–2 at steady state) .
-
Weak inhibition of CYP2D6 substrates like timolol and propafenone (controversial, with conflicting in vitro and in vivo data) .
CYP3A4 Modulation
-
Inhibition by CYP3A4 inhibitors (e.g., ketoconazole) increases this compound levels, while inducers (e.g., carbamazepine) reduce them .
CYP2C19 Interactions
-
Proton pump inhibitors (e.g., esomeprazole) inhibit CYP2C19, raising this compound/desmethylthis compound ratios .
Repurposing as a Metabolic Inhibitor
This compound inhibits serine hydroxymethyltransferase (SHMT) enzymes, blocking serine/glycine synthesis in cancer cells. This interaction is exploited in combination therapies (e.g., with mitochondrial inhibitors like artemether) .
Structural Analysis
This compound’s structure (C₁₇H₁₇Cl₂N) includes a tetrahydronaphthalenamine core with a 3,4-dichlorophenyl substituent. Its stereochemical purity is critical for efficacy, achieved via resolution with mandelic acid .
Scientific Research Applications
Sertraline, commonly known by its brand name Zoloft, is a selective serotonin reuptake inhibitor (SSRI) antidepressant used to treat a variety of mental health conditions . Clinical studies indicate this compound's efficacy in improving cognition in depressed patients .
Scientific Research Applications
Approved Indications
this compound is approved for the management of several conditions :
- Major Depressive Disorder (MDD)
- Obsessive-Compulsive Disorder (OCD)
- Post-Traumatic Stress Disorder (PTSD)
- Panic Disorder (PD)
- Premenstrual Dysphoric Disorder (PMDD)
- Social Anxiety Disorder (SAD)
Off-Label Uses
this compound also has off-label uses, including :
- Prevention of post-stroke depression
- Generalized Anxiety Disorder (GAD)
- Fibromyalgia
- Premature ejaculation
- Migraine prophylaxis
- Diabetic neuropathy
- Neurocardiogenic syncope
Cancer Therapy
this compound has demonstrated promising anticancer effects in several cancer cell lines, both when used alone and in combination with other treatments . this compound can modulate efflux pumps in multidrug-resistant cellular models. In vivo studies have shown that combining this compound with Doxil® (pegylated liposomal doxorubicin) effectively reduces tumor growth and extends survival in tumor-bearing mice, suggesting this compound's potential as a chemosensitizer and multidrug resistance modulator in ovarian cancer .
Pharmacokinetics
this compound's advantages over older antidepressants include improved patient tolerability, a low risk of lethality in overdose, and no dependence potential . this compound is at least as well-tolerated as other SSRIs and may have a more favorable side effect profile. It also has a low potential for pharmacokinetic drug interactions, and, unlike fluoxetine, fluvoxamine, and paroxetine, this compound is not a potent inhibitor of any of the cytochrome P450 isoenzyme systems .
Case Studies and Research Findings
Efficacy in PTSD Treatment
A study examining this compound's efficacy and safety in treating PTSD showed significantly greater improvement than placebo on several primary outcome measures, including changes in CAPS-2 total severity score and Clinical Global Impression–Severity (CGI-S) . this compound effectively attenuates the behavioral syndrome occurring in animals after uncontrollable stress, which is considered an animal model of PTSD .
Meta-Analysis of Antidepressants
Meta-analyses have indicated a modest but statistically significant additional treatment effect for this compound and venlafaxine compared to fluoxetine in treating major depressive disorder .
This compound Quality of Life Battery
The this compound Quality of Life Battery measures health perceptions, energy/vitality, and cognitive functions .
Mechanism of Action
Sertraline exerts its effects by selectively inhibiting the reuptake of serotonin into presynaptic neurons. This inhibition increases the concentration of serotonin in the synaptic cleft, enhancing serotonergic neurotransmission. The primary molecular target of this compound is the serotonin transporter, which is responsible for the reuptake of serotonin . By blocking this transporter, this compound prolongs the action of serotonin, leading to improved mood and reduced anxiety .
Comparison with Similar Compounds
- Citalopram
- Escitalopram
- Fluoxetine
- Fluvoxamine
- Paroxetine
Comparison: Sertraline is unique among selective serotonin reuptake inhibitors due to its relatively mild side effect profile and its efficacy in treating a broad range of psychiatric disorders . Compared to other selective serotonin reuptake inhibitors, this compound has a lower incidence of weight gain and cognitive impairment . Additionally, it is often preferred for its favorable balance of efficacy and tolerability .
Biological Activity
Sertraline, a selective serotonin reuptake inhibitor (SSRI), is primarily known for its efficacy in treating major depressive disorder and anxiety disorders. However, recent studies have revealed a broader spectrum of biological activities, including antimicrobial properties and effects on biofilm formation. This article explores the biological activity of this compound through various research findings, case studies, and data tables.
This compound functions by inhibiting the reuptake of serotonin in the brain, which increases serotonin levels in the synaptic cleft. This mechanism is crucial for its antidepressant and anxiolytic effects. Additionally, this compound has been shown to influence other neurotransmitter systems, including norepinephrine and dopamine, contributing to its therapeutic effects.
Antimicrobial Activity
Recent research has highlighted this compound's antimicrobial properties against various pathogens:
- Listeria monocytogenes : A study demonstrated that this compound significantly reduces the viability of L. monocytogenes in a dose-dependent manner. The green fluorescence intensity decreased by 29.1%, 38.7%, and 46.9% at concentrations of 16, 32, and 64 µg/mL, respectively .
- Candida species : this compound has shown synergistic effects when combined with essential oils, leading to a significant reduction in biofilm formation. The minimum inhibitory concentration (MIC) values for this compound dropped dramatically when used in combination with Cinnamomum verum essential oil .
Case Studies and Trials
- Generalized Anxiety Disorder : In a 12-week trial, this compound exhibited significant efficacy compared to placebo in reducing anxiety symptoms as measured by the Hamilton Anxiety Scale. The mean score reduction was significantly greater in the this compound group (11.7) compared to placebo (8.0) .
- Major Depression : A comparative analysis indicated that this compound was more effective in clinical practice settings than in controlled trials, with higher response rates (87% vs. 73%) and better tolerability .
Tolerability and Safety
This compound is generally well-tolerated, with dropout rates due to adverse events being lower in clinical practice settings compared to clinical trials (9.4% vs. 13.2%) . Common side effects include gastrointestinal disturbances, insomnia, and sexual dysfunction.
Table 1: Summary of this compound's Biological Activities
Q & A
Basic Research Questions
Q. What experimental designs are optimal for studying sertraline’s pharmacokinetics in diverse populations?
To investigate pharmacokinetic variability, employ a longitudinal cohort design with stratified sampling based on covariates like BMI, age, and comorbidities. Use therapeutic drug monitoring (TDM) to measure serum concentration/dose ratios (C/D) and apply multiple linear regression to identify predictors (e.g., drug interactions, BMI). Ensure sample sizes are powered to detect clinically significant differences (e.g., ≥20% C/D variation) . Methodological Example :
- Data Collection: Retrospective TDM data from 373 patients, analyzed via SPSS 23.0.
- Key Covariates: BMI, co-administered drugs (e.g., olanzapine, lithium).
- Statistical Tools: ANOVA and multivariate regression to isolate confounding factors .
Q. How can researchers address conflicting results in this compound’s efficacy across depression subtypes?
Adopt a hypothesis-driven approach:
- Define subtypes using validated scales (e.g., Hamilton Depression Rating Scale) .
- Use randomized controlled trials (RCTs) with subgroup analysis powered for heterogeneity.
- Apply Bayesian meta-analysis to quantify uncertainty in contradictory datasets .
Q. What are the best practices for ensuring reproducibility in preclinical this compound studies?
Follow ARRIVE guidelines:
- Animal Models: Specify strain, sex, and rcTBI (repetitive concussive traumatic brain injury) protocols for depression-like behavior studies .
- Dosage Standardization: Report mg/kg, administration routes, and treatment duration.
- Blinding: Implement double-blinding for behavioral assessments .
Advanced Research Questions
Q. How does this compound modulate amino acid addiction in cancer cells, and what experimental models validate this?
- In Vitro: Use CRISPR-edited cell lines to knockout mTOR or glutamine transporters. Measure proliferation via MTT assays under this compound treatment.
- In Vivo: Xenograft models (e.g., murine leukemia) with PET imaging to track tumor metabolism. Validate findings via RNA-seq to identify pathways like PI3K/AKT .
Key Findings :
| Model Type | This compound Effect | Mechanism Identified | Reference |
|---|---|---|---|
| Mouse Xenografts | Tumor Growth ↓40% | Glutamine uptake inhibition | |
| Leukemia Cells | Apoptosis ↑2x | mTOR pathway suppression |
Q. How can researchers resolve contradictions in this compound’s neuroprotective vs. neurotoxic effects in TBI models?
- Experimental Design: Compare acute vs. chronic dosing in rcTBI models.
- Endpoints: Axonal injury (β-APP immunohistochemistry) and depression-like behavior (forced swim test).
- Contradiction Analysis: Use dose-response curves to identify therapeutic windows. Unpublished data suggest 10 mg/kg mitigates depression but has no effect on tau pathology .
Q. What computational methods predict this compound-drug interactions impacting CYP450 metabolism?
- In Silico Tools: Use Schrödinger’s Molecular Dynamics for CYP2B6/2C19 binding affinity simulations.
- Clinical Validation: Cross-reference with TDM data showing ↑this compound C/D with CYP inhibitors (e.g., fluoxetine) .
Q. Methodological Guidelines
Q. How to design a clinical study analyzing this compound’s impact on comorbid depression and chronic pain?
- Population: Recruit patients with fibromyalgia or diabetic neuropathy.
- Outcomes: Dual primary endpoints (e.g., HAM-D scores and pain VAS).
- Analysis: Mixed-effects models to adjust for baseline pain severity .
Q. What statistical approaches are robust for this compound meta-analyses with high heterogeneity?
- Fixed vs. Random Effects: Use I² statistic to quantify heterogeneity.
- Subgroup Analysis: Stratify by study quality (e.g., Cochrane Risk of Bias Tool).
- Sensitivity Testing: Exclude outlier studies and re-pool effects .
Q. Data Presentation Standards
- Tables: Include confidence intervals, p-values, and effect sizes.
- Reproducibility: Publish raw data in repositories like Zenodo, with detailed protocols for synthesis and HPLC methods .
Properties
IUPAC Name |
(1S,4S)-4-(3,4-dichlorophenyl)-N-methyl-1,2,3,4-tetrahydronaphthalen-1-amine;hydrochloride | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C17H17Cl2N.ClH/c1-20-17-9-7-12(13-4-2-3-5-14(13)17)11-6-8-15(18)16(19)10-11;/h2-6,8,10,12,17,20H,7,9H2,1H3;1H/t12-,17-;/m0./s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
BLFQGGGGFNSJKA-XHXSRVRCSA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CNC1CCC(C2=CC=CC=C12)C3=CC(=C(C=C3)Cl)Cl.Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
CN[C@H]1CC[C@H](C2=CC=CC=C12)C3=CC(=C(C=C3)Cl)Cl.Cl | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C17H18Cl3N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Related CAS |
79617-96-2 (Parent) | |
| Record name | Sertraline hydrochloride [USAN:USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0079559970 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
DSSTOX Substance ID |
DTXSID1040243 | |
| Record name | Sertraline hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1040243 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
342.7 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
CAS No. |
79559-97-0, 79617-89-3 | |
| Record name | Sertraline hydrochloride | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=79559-97-0 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | (±)-Sertraline hydrochloride | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=79617-89-3 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Sertraline hydrochloride [USAN:USP] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=0079559970 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Sertraline hydrochloride | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=758948 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Sertraline hydrochloride | |
| Source | DTP/NCI | |
| URL | https://dtp.cancer.gov/dtpstandard/servlet/dwindex?searchtype=NSC&outputformat=html&searchlist=746308 | |
| Description | The NCI Development Therapeutics Program (DTP) provides services and resources to the academic and private-sector research communities worldwide to facilitate the discovery and development of new cancer therapeutic agents. | |
| Explanation | Unless otherwise indicated, all text within NCI products is free of copyright and may be reused without our permission. Credit the National Cancer Institute as the source. | |
| Record name | Sertraline hydrochloride | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID1040243 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | 1-Naphthalenamine, 4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-, hydrochloride (1:1), (1S,4S) | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.111.337 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | SERTRALINE HYDROCHLORIDE | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/UTI8907Y6X | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















