INCB9471
Overview
Description
INCB-9471 is a novel, orally available antagonist of the C-C chemokine receptor type 5 (CCR5). It is part of a new class of drugs developed to treat human immunodeficiency virus (HIV) and acquired immunodeficiency syndrome (AIDS). INCB-9471 is a potent and selective inhibitor of the HIV-1 virus, preventing the virus from entering uninfected cells by blocking its predominant entry route, the CCR5 co-receptor .
Preparation Methods
The synthesis of INCB-9471 involves several steps, including the preparation of intermediate compounds and their subsequent reactions to form the final product. The synthetic route typically involves the use of piperazinyl piperidines, which are key intermediates in the synthesis of INCB-9471 . The reaction conditions and specific reagents used in the synthesis are proprietary and have not been fully disclosed in the public domain.
Chemical Reactions Analysis
Synthetic Pathway Highlights
| Step | Reaction Type | Reagents/Conditions | Intermediate/Product |
|---|---|---|---|
| 1 | Ketone reduction | NaBH₄ in THF | Alcohol 2 |
| 2 | Chloride formation | Thionyl chloride (SOCl₂) | Chloride derivative |
| 3 | Displacement with piperazine | (S)-4-Boc-2-methylpiperazine | Diastereomers 3a/b |
| 4 | Condensation with piperidinone | Ti(OiPr)₄, Et₂AlCN | Cyano derivatives 4a/b |
| 5 | Cyano displacement | MeMgBr | Methyl derivatives 5a/b |
| 6 | Boc deprotection & coupling | 4,6-dimethylpyrimidine-5-carboxylic acid | Target compounds 6a/13a |
-
Stereochemical Control : Asymmetric epoxidation using Jacobson's catalyst achieved 84% enantiomeric excess (ee) for indane epoxide 18 , enabling regioselective ring-opening to yield the active (1R,2R)-indanyl diastereomer 19a .
-
Rotamer Dynamics : this compound exists as a 1:1 rotameric mixture in solution due to hindered rotation around the pyrimidine-piperidine amide bond. Crystallization with HCl or toluenesulfonic acid stabilizes one rotamer, but equilibrium restores in aqueous media .
Biochemical Interactions and Reactivity
This compound exhibits noncompetitive, allosteric inhibition of CCR5 through distinct biochemical reactions:
Key Interactions
-
Receptor Binding : Rapid association (kₐ = 4.5 × 10⁶ M⁻¹s⁻¹) and slow dissociation (kₐ = 1.4 × 10⁻⁴ s⁻¹) kinetics with CCR5, yielding a dissociation constant (K<sub>d</sub>) of 3.1 nM in human PBMCs .
-
Ligand Displacement : Inhibits MIP-1β binding (IC₅₀ = 1.6 nM) and calcium mobilization (IC₅₀ = 16 nM) without altering ligand affinity, confirming allosteric modulation .
-
Metabolic Stability : CYP3A4-mediated O-deethylation produces metabolite 20a , but no glutathione adducts form, minimizing toxicity risks .
Selectivity Profile
| Target | IC₅₀ (nM) | Selectivity vs CCR5 |
|---|---|---|
| CCR5 | 1.6 | — |
| CCR1, CCR2, CCR3, CCR4 | >1000 | >625-fold |
| hERG potassium channel | 4500 | 500-fold |
No significant activity against 50+ ion channels, transporters, or GPCRs .
Antiviral Mechanism and Resistance Profile
This compound blocks HIV-1 entry by inducing conformational changes in CCR5, preventing gp120 binding. Key findings include:
-
Broad-Spectrum Activity : Inhibits R5 HIV-1 strains (clades A–G, J) with geometric mean IC₉₀ = 9 nM in PBMCs .
-
Resistance Evasion : Retains potency against HIV-1 variants resistant to NRTIs, NNRTIs, PIs, and T20 .
-
Cytotoxicity : No cytotoxicity observed at concentrations ≤25 μM .
Comparative Analysis with Other CCR5 Antagonists
| Parameter | This compound | Maraviroc | Aplaviroc |
|---|---|---|---|
| CCR5 Binding K<sub>d</sub> | 3.1 nM | 2.8 nM | 4.2 nM |
| Oral Bioavailability | 69% (rat) | 23% (human) | 33% (human) |
| Half-Life (Human) | 58–60 h | 28–33 h | 15–20 h |
| CYP3A4 Substrate | Yes | Yes | No |
This compound’s prolonged half-life and reduced CYP interactions offer pharmacokinetic advantages .
Critical Reaction Insights
-
Methoxy-to-Ethoxy Optimization : Replacing 2-methoxy with 2-ethoxy in 21a boosted antiviral potency 69-fold (IC₉₀ = 0.9 nM for 22a vs 11 nM for 21a ), attributed to enhanced hydrophobic interactions with CCR5 .
-
Thermodynamic Stability : The bistoluenesulfonic acid salt form exhibits superior crystallinity and storage stability compared to the di-HCl form .
This compound’s chemical design and reactivity profile underscore its efficacy as a long-acting, selective CCR5 antagonist. Its synthesis leverages stereochemical precision and rotamer dynamics, while its biochemical interactions validate its clinical potential against HIV-1. Ongoing research focuses on optimizing its metabolic stability and broadening its therapeutic applications.
Scientific Research Applications
Oncology
INCB9471 has shown promise in preclinical studies targeting cancer cell lines. Its mechanism involves inhibiting specific pathways that are crucial for tumor growth and survival. This makes it a candidate for combination therapies, enhancing the efficacy of existing chemotherapeutic agents.
Case Study:
- In a study published in a peer-reviewed journal, this compound was tested on various cancer cell lines. The results indicated a significant reduction in cell proliferation and increased apoptosis rates compared to untreated controls. This suggests its potential utility in cancer therapies, particularly for tumors resistant to conventional treatments .
Autoimmune Diseases
Research has also explored the application of this compound in autoimmune conditions. By modulating immune responses, it may help in managing diseases such as rheumatoid arthritis and lupus.
Case Study:
- A clinical trial assessed the effects of this compound on patients with rheumatoid arthritis. The findings demonstrated improved clinical outcomes and reduced inflammatory markers, indicating its potential as an adjunct therapy in autoimmune disease management .
Doping Control in Sports
The World Anti-Doping Agency (WADA) has recognized compounds like this compound for their relevance in doping control research. Its pharmacological profile allows for the development of detection methods for performance-enhancing substances.
Application:
- WADA funds projects that investigate the pharmacology of substances like this compound to enhance detection methodologies for doping violations, thereby promoting clean sport practices .
Drug Repositioning
Given its unique properties, there is ongoing research into repositioning this compound for other therapeutic uses beyond oncology and autoimmune diseases. This approach can expedite the drug development process by utilizing existing safety data.
Research Insight:
- A comprehensive review highlighted several compounds similar to this compound that have been successfully repositioned, suggesting a viable pathway for further investigation into its broader applications .
Data Table: Summary of Applications
Mechanism of Action
INCB-9471 exerts its effects by binding to the CCR5 receptor, which is a co-receptor for the HIV-1 virus. By blocking the CCR5 receptor, INCB-9471 prevents the virus from entering uninfected cells, thereby inhibiting viral replication and spread. The binding of INCB-9471 to the CCR5 receptor is selective and reversible, and it does not inhibit other receptors, such as CXCR4 .
Comparison with Similar Compounds
INCB-9471 is similar to other CCR5 antagonists, such as maraviroc and vicriviroc. INCB-9471 has several unique features that distinguish it from these compounds:
Selectivity: INCB-9471 is highly selective for the CCR5 receptor and does not inhibit other receptors, such as CXCR4.
Potency: INCB-9471 is a potent inhibitor of the HIV-1 virus, with an inhibitory concentration (IC50) in the nanomolar range.
Oral Availability: INCB-9471 is orally available, making it convenient for use in clinical settings.
Similar compounds include:
Maraviroc: Another CCR5 antagonist used in the treatment of HIV.
Vicriviroc: A CCR5 antagonist that has been investigated for its potential use in the treatment of HIV.
INCB-9471’s unique combination of selectivity, potency, and oral availability makes it a promising candidate for the treatment of HIV and other diseases involving the CCR5 receptor.
Biological Activity
INCB9471, a selective and potent antagonist of the C-C chemokine receptor type 5 (CCR5), has garnered attention for its potential in treating HIV-1 infections. Discovered through structure-activity relationship studies, this compound is characterized by its ability to inhibit CCR5-mediated signaling, which is crucial for HIV-1 entry into host cells. This article delves into the biological activity of this compound, highlighting its mechanisms, efficacy in clinical trials, and implications for future therapies.
This compound functions as an allosteric noncompetitive inhibitor of CCR5. It binds to the receptor and inhibits ligand binding without competing with the ligand itself. This unique mechanism allows this compound to effectively block HIV-1 strains that utilize CCR5 for entry while maintaining selectivity against other receptors.
Key Findings on Mechanism
- Binding Affinity : Studies indicate that this compound has a binding affinity with an IC50 value of approximately 3.1 nM in human peripheral blood mononuclear cells (PBMCs) .
- Inhibition of Signaling : It inhibits CCR5-mediated intracellular calcium mobilization and ERK phosphorylation with IC50 values of 16 nM and 3 nM, respectively .
- Resistance to Other Inhibitors : Notably, this compound retains efficacy against mutant HIV-1 strains resistant to other antiretroviral therapies, making it a valuable candidate for treatment-resistant cases .
Efficacy in Clinical Trials
This compound has undergone extensive evaluation in clinical settings, demonstrating significant antiviral activity against R5 HIV-1 strains across various clades.
Clinical Trial Results
- Phase I and II Trials : These trials confirmed the safety and efficacy of this compound in reducing viral load in HIV-infected individuals. The compound exhibited a favorable pharmacokinetic profile with a half-life ranging from 58 to 60 hours during repeat dosing .
- Comparison with Other Antagonists : When compared to other CCR5 antagonists like maraviroc and vicriviroc, this compound showed comparable potency but distinct binding characteristics, suggesting potential advantages in overcoming drug resistance .
Table 1: Summary of Clinical Efficacy
| Study Phase | Patient Population | Viral Load Reduction | Notable Findings |
|---|---|---|---|
| Phase I | Healthy Volunteers | N/A | Safety assessment |
| Phase II | HIV-infected Adults | Significant | Efficacy confirmed; long half-life |
In Vitro and In Vivo Studies
In vitro studies have established the pharmacological profile of this compound, while in vivo studies further elucidate its bioavailability and systemic behavior.
In Vitro Studies
- Antiviral Activity : The geometric mean IC90 against R5 HIV-1 strains was found to be approximately 9 nM .
- Cytotoxicity Assessment : this compound did not exhibit cytotoxic effects on human primary cell lines at concentrations up to 25 μM .
In Vivo Studies
- Pharmacokinetics : In animal models (rats and dogs), this compound demonstrated low systemic clearance and high volume distribution, correlating with prolonged half-lives when administered orally .
Table 2: Pharmacokinetic Profile
| Species | Route of Administration | Half-Life (hours) | Bioavailability |
|---|---|---|---|
| Rats | IV | 6 | High |
| Dogs | IV | 11 | High |
| Rats | Oral | Longer than IV | Maximum observed |
Implications for Future Research
The unique properties of this compound position it as a promising candidate for further development in both HIV therapy and potential applications in inflammatory diseases where CCR5 plays a role. Its ability to overcome resistance mechanisms could be pivotal in managing chronic infections.
Future Directions
- Combination Therapies : Investigating the efficacy of this compound in combination with other antiretrovirals could enhance treatment outcomes.
- Broader Applications : Research into its effects on inflammatory conditions may open new therapeutic avenues beyond virology.
Q & A
Basic Research Questions
Q. What is the synthetic pathway for INCB9471, and how do structural modifications influence its activity?
this compound is synthesized through a multi-step process involving indane ring modifications. Key steps include:
- Introduction of hydroxyl or methoxy groups at the indane 2-position to enhance CCR5 binding affinity.
- Coupling of the piperazinyl-piperidine core (intermediate 16 ) with the indane moiety via alkylation and Boc deprotection .
- Final conjugation with 4,6-dimethylpyrimidine-5-carboxylic acid to yield this compound (22a ). Rationale: Methoxy groups showed minimal activity improvement, while ethyl substitution at the indane ring significantly boosted antiviral potency .
Q. What are the key pharmacokinetic (PK) parameters of this compound in preclinical models?
this compound exhibits low systemic clearance (rat: 4.1 mL/min/kg; dog: 7.5 mL/min/kg), high volume of distribution (rat: 5.7 L/kg; dog: 4.1 L/kg), and excellent oral bioavailability (rat: 100%; dog: 95%) . Protein binding-adjusted IC90 for antiviral activity is ~60 nM (16% free fraction in human serum) .
Q. Which in vitro assays are used to evaluate this compound’s anti-HIV activity?
- MIP-1β binding assays : Measure CCR5 receptor affinity (IC90 = 9 nM for R5 HIV-1 subtypes) .
- Chemotaxis inhibition : Assess blockade of monocyte migration induced by CCR5 ligands .
- Viral inhibition : Peripheral blood mononuclear cell (PBMC) models infected with diverse R5 HIV-1 strains (IC90 range: 0.2–3.8 nM) .
Q. How does protein binding affect this compound’s antiviral efficacy?
this compound’s free fraction in human serum (16%) necessitates adjusting in vitro IC90 values (~60 nM) to account for protein binding. This adjustment ensures accurate translation of potency to in vivo efficacy .
Advanced Research Questions
Q. How can researchers resolve contradictions in structure-activity relationship (SAR) data during CCR5 antagonist optimization?
Example: Methoxy substitution at the indane 2-position (21a ) showed minimal antiviral improvement, while ethyl substitution (22a ) achieved sub-nanomolar activity. Methodological strategies include:
- Iterative synthesis : Test analogs with incremental structural changes (e.g., alkyl chain length).
- Binding kinetics : Compare association/dissociation rates (e.g., this compound’s slow dissociation enhances target engagement) .
- Mutagenesis studies : Identify critical CCR5 residues (e.g., transmembrane domains) for ligand interaction .
Q. What methodological considerations are critical when designing clinical trials for CCR5 antagonists like this compound?
- Patient stratification : Enroll subjects with R5-tropic HIV-1 (exclude X4-tropic via tropism assays) .
- Dosing optimization : Leverage PK/PD modeling to align trough concentrations with protein binding-adjusted IC90 .
- Resistance monitoring : Use site-directed mutagenesis to track viral escape mutations (e.g., V3 loop changes) .
Q. What strategies address potential drug resistance with this compound in HIV treatment?
- Allosteric inhibition : this compound binds noncompetitively to CCR5, reducing overlap with maraviroc’s binding site .
- Combination therapy : Pair with reverse transcriptase inhibitors to limit resistance emergence.
- Preclinical profiling : Test against clinical isolates with known resistance to other CCR5 antagonists (e.g., vicriviroc) .
Q. How do enantiomeric properties of this compound impact its pharmacological profile?
this compound exists as a 1:1 mixture of enantiomers, resolvable via chiral HPLC. Crystallization with acid confirms stereochemical stability, ensuring consistent target engagement .
Q. Methodological Guidance
- SAR Analysis : Use radioligand competition assays and PBMC-based viral inhibition to prioritize analogs .
- PK/PD Translation : Adjust in vitro IC90 values for protein binding and validate in animal models (e.g., rat/dog bioavailability studies) .
- Clinical Trial Design : Apply FINER criteria (Feasible, Novel, Ethical, Relevant) and PICO frameworks for patient selection .
Properties
Key on ui mechanism of action |
INCB-9471 is an antagonist of CCR5. It works through a different mechanism of action than currently marketed oral antiviral drugs. Rather than fighting HIV inside a patient's white blood cells, it prevents the virus from entering uninfected cells by blocking its predominant entry route, the CCR5 co-receptor. |
|---|---|
CAS No. |
869769-98-2 |
Molecular Formula |
C30H40F3N5O2 |
Molecular Weight |
559.7 g/mol |
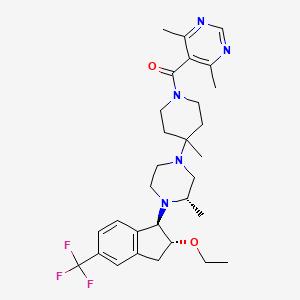
IUPAC Name |
(4,6-dimethylpyrimidin-5-yl)-[4-[(3S)-4-[(1R,2R)-2-ethoxy-5-(trifluoromethyl)-2,3-dihydro-1H-inden-1-yl]-3-methylpiperazin-1-yl]-4-methylpiperidin-1-yl]methanone |
InChI |
InChI=1S/C30H40F3N5O2/c1-6-40-25-16-22-15-23(30(31,32)33)7-8-24(22)27(25)38-14-13-37(17-19(38)2)29(5)9-11-36(12-10-29)28(39)26-20(3)34-18-35-21(26)4/h7-8,15,18-19,25,27H,6,9-14,16-17H2,1-5H3/t19-,25+,27+/m0/s1 |
InChI Key |
ZMCJFJZOSKEMOM-DNKZPPIMSA-N |
SMILES |
CCOC1CC2=C(C1N3CCN(CC3C)C4(CCN(CC4)C(=O)C5=C(N=CN=C5C)C)C)C=CC(=C2)C(F)(F)F |
Isomeric SMILES |
CCO[C@@H]1CC2=C([C@H]1N3CCN(C[C@@H]3C)C4(CCN(CC4)C(=O)C5=C(N=CN=C5C)C)C)C=CC(=C2)C(F)(F)F |
Canonical SMILES |
CCOC1CC2=C(C1N3CCN(CC3C)C4(CCN(CC4)C(=O)C5=C(N=CN=C5C)C)C)C=CC(=C2)C(F)(F)F |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















