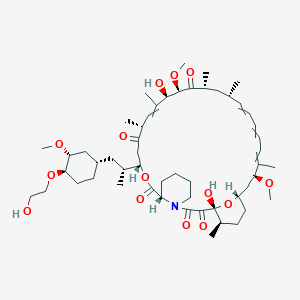
Everolimus
Overview
Description
Everolimus is a derivative of rapamycin, known for its immunosuppressive and anti-proliferative properties. It is primarily used to prevent organ rejection in transplant patients and to treat various types of cancer, including renal cell carcinoma and certain types of breast cancer . This compound works by inhibiting the mammalian target of rapamycin (mTOR), a key protein involved in cell growth and proliferation .
Preparation Methods
Synthetic Routes and Reaction Conditions: Everolimus is synthesized from rapamycin through a series of chemical reactions. The primary synthetic route involves the reaction of rapamycin with ethylene glycol mono trifluoromethanesulfonate under the influence of an organic base. This reaction produces an intermediate compound, which is then subjected to an acetal protection group removal reaction under acidic conditions to yield this compound .
Industrial Production Methods: The industrial production of this compound follows similar synthetic routes but is optimized for higher yields and cost-effectiveness. The process involves the use of inexpensive and readily available reagents, such as ethylene glycol mono trifluoromethanesulfonate, and includes steps for the recycling of unreacted rapamycin to ensure product quality and reduce costs .
Chemical Reactions Analysis
Types of Reactions: Everolimus undergoes various chemical reactions, including oxidation, reduction, and substitution. These reactions are essential for its metabolic processing and therapeutic action.
Common Reagents and Conditions:
Oxidation: Typically involves the use of oxidizing agents like hydrogen peroxide or potassium permanganate.
Reduction: Often carried out using reducing agents such as sodium borohydride or lithium aluminum hydride.
Substitution: Involves nucleophilic or electrophilic reagents under controlled conditions.
Major Products: The primary products formed from these reactions include various metabolites that are further processed in the body to exert their therapeutic effects .
Scientific Research Applications
Oncology Applications
Everolimus is primarily utilized in oncology for its antiproliferative properties. It has been approved by the United States Food and Drug Administration (FDA) for several cancer types, including:
- Renal Cell Carcinoma : this compound is indicated for advanced renal cell carcinoma after prior treatment with other therapies like sorafenib or sunitinib. In clinical trials, it demonstrated a median progression-free survival of 4.9 months compared to 1.9 months for placebo .
- Breast Cancer : In combination with letrozole, this compound has shown efficacy in treating hormone receptor-positive advanced breast cancer. A phase II trial revealed a notable improvement in progression-free survival .
- Gastric Cancer : A phase II study indicated that this compound could reduce the risk of disease progression in pretreated advanced gastric cancer patients, with a disease control rate significantly higher than placebo .
- Neuroendocrine Tumors : this compound is also approved for pancreatic neuroendocrine tumors, where it has shown a 65% decrease in tumor progression risk .
| Cancer Type | Indication | Median PFS (months) | Response Rate |
|---|---|---|---|
| Renal Cell Carcinoma | After prior therapy | 4.9 | 7% |
| Breast Cancer | With letrozole | Not specified | Not specified |
| Gastric Cancer | Advanced, previously treated | Not specified | 38.9% |
| Neuroendocrine Tumors | Advanced pancreatic neuroendocrine tumors | 12 | 65% decrease |
Transplantation Applications
This compound plays a crucial role in organ transplantation due to its immunosuppressive properties:
- Kidney Transplantation : It is used to prevent acute rejection in kidney transplant recipients, allowing for potential minimization or elimination of calcineurin inhibitors, which can have nephrotoxic effects .
- Other Organ Transplants : The drug has also been investigated for use in heart and liver transplants, showing promise in maintaining graft function and reducing rejection rates .
Other Therapeutic Uses
Beyond oncology and transplantation, this compound has been explored for various other conditions:
- Tuberous Sclerosis Complex : Approved for treating subependymal giant cell astrocytoma associated with tuberous sclerosis complex, this compound has shown efficacy in improving neurological function and survival rates in affected patients .
- Cardiovascular Applications : As a drug-eluting stent coating, this compound helps prevent restenosis by inhibiting smooth muscle cell proliferation .
Case Study 1: Advanced Renal Cell Carcinoma
A study involving patients with metastatic renal cell carcinoma treated with this compound showed an overall response rate of approximately 7%, with significant improvements in progression-free survival compared to placebo groups .
Case Study 2: Hormone Receptor-Positive Breast Cancer
In the MIRACLE trial, premenopausal women with advanced breast cancer receiving this compound plus letrozole exhibited enhanced outcomes compared to those on letrozole alone, demonstrating the potential of combination therapy in this setting .
Mechanism of Action
Everolimus exerts its effects by inhibiting the mTOR pathway, specifically targeting the mTORC1 protein complex. This inhibition disrupts cell growth, proliferation, and survival signals, leading to reduced tumor growth and immune response modulation. The molecular targets include various proteins involved in the PI3K/AKT/mTOR signaling pathway .
Comparison with Similar Compounds
Sirolimus (Rapamycin): The parent compound of everolimus, used for similar therapeutic purposes but with different pharmacokinetic properties.
Temsirolimus: Another mTOR inhibitor with a similar mechanism of action but used primarily for treating renal cell carcinoma.
Uniqueness of this compound: this compound is unique due to its enhanced oral bioavailability and specific targeting of the mTORC1 protein complex. This specificity allows for more effective inhibition of cell growth and proliferation, making it a valuable therapeutic agent in oncology and transplant medicine .
Biological Activity
Everolimus, a potent inhibitor of the mammalian target of rapamycin (mTOR), is an analog of rapamycin and has gained prominence in the treatment of various malignancies, including renal cell carcinoma and neuroendocrine tumors. This article delves into the biological activity of this compound, focusing on its mechanisms of action, pharmacodynamics, clinical efficacy, and safety profile, supported by data tables and relevant case studies.
This compound exerts its effects by binding to the FK506 binding protein-12 (FKBP-12), forming a complex that inhibits mTOR activity. This inhibition disrupts several downstream signaling pathways critical for cell growth and proliferation:
- Cell Cycle Regulation : By inhibiting mTOR, this compound blocks the progression of cells from the G1 phase to the S phase, leading to cell cycle arrest and apoptosis.
- Angiogenesis Inhibition : this compound reduces the expression of hypoxia-inducible factor (HIF) and vascular endothelial growth factor (VEGF), thereby inhibiting angiogenesis, which is crucial for tumor growth and metastasis .
- Metabolic Effects : The drug also impacts glucose uptake in cancer cells, further contributing to its anti-tumor effects .
Pharmacodynamics
The pharmacodynamic profile of this compound includes its effects on various biomarkers associated with mTOR signaling. A comprehensive study indicated that this compound effectively inhibited phosphorylation of ribosomal protein S6, a common marker for mTORC1 activity. The study also revealed that daily dosing at 10 mg provided more robust and prolonged inhibition compared to weekly dosing schedules .
Key Pharmacodynamic Findings
| Parameter | Daily Dosing (10 mg) | Weekly Dosing (50 mg) |
|---|---|---|
| S6 Phosphorylation Inhibition | Almost complete | Moderate |
| Proliferation Reduction | Significant | Moderate |
| Hyperphosphorylation of Akt | Observed in ~50% samples | Not maintained |
Clinical Efficacy
This compound has been evaluated in numerous clinical trials demonstrating its efficacy across various cancer types. Notably, it has shown significant activity in patients with renal cell carcinoma who are refractory to other therapies.
Case Studies
- Renal Cell Carcinoma : In a pivotal trial involving 410 patients with advanced renal cell carcinoma, this compound demonstrated a median progression-free survival (PFS) of 4.9 months compared to 1.9 months for placebo .
- Neuroendocrine Tumors : A study involving patients with advanced neuroendocrine tumors showed an overall response rate (ORR) of 32% when treated with this compound, highlighting its potential in this indication .
Safety Profile
While this compound is generally well-tolerated, it is associated with several adverse effects:
- Common Adverse Effects : Include stomatitis, infections (e.g., pneumonia), fatigue, and skin rash.
- Serious Adverse Events : Renal failure and proteinuria have been reported in clinical settings .
Table of Adverse Effects
| Adverse Effect | Incidence (%) |
|---|---|
| Stomatitis | 40 |
| Infections | 30 |
| Fatigue | 25 |
| Renal Failure | 10 |
Q & A
Basic Research Questions
Q. What are the key considerations for designing phase III clinical trials evaluating everolimus in hormone receptor-positive advanced breast cancer?
- Trials should randomize patients with prior nonsteroidal aromatase inhibitor (NSAI) resistance to this compound + exemestane vs. placebo + exemestane, using progression-free survival (PFS) as the primary endpoint. Stratification factors (e.g., visceral metastases, prior chemotherapy) ensure balanced cohorts. Interim analyses by independent committees are critical for early efficacy/safety assessments . Secondary endpoints include overall survival (OS) and objective response rates.
Q. How should researchers address adverse events (AEs) like stomatitis and hyperglycemia in this compound trials?
- Proactively monitor AEs using CTCAE criteria. Grade 3/4 stomatitis (8% incidence) may require dose reductions or topical therapies, while hyperglycemia (4%) necessitates glucose monitoring and antihyperglycemic agents. Preemptive patient education and AE management protocols improve trial adherence .
Q. What statistical methods are appropriate for analyzing PFS in this compound trials?
- Use stratified log-rank tests and Cox proportional hazards models to calculate hazard ratios (HRs) with 95% confidence intervals. Central vs. local PFS assessments may yield divergent results (e.g., HR 0.36 vs. 0.43 in BOLERO-2), highlighting the need for blinded independent review .
Advanced Research Questions
Q. How do PI3K/AKT/mTOR pathway alterations influence this compound efficacy across cancer types?
- In HER2+ breast cancer, PIK3CA mutations or PTEN loss correlate with improved PFS (HR 0.67 for hyperactive PI3K pathway). Conversely, wild-type PI3K tumors derive minimal benefit. Use next-generation sequencing (NGS) and immunohistochemistry to stratify patients by biomarker status in trials .
Q. What methodologies resolve contradictions in this compound efficacy between renal cell carcinoma (RCC) and pancreatic neuroendocrine tumors (pNET)?
- In pNET, this compound improves median PFS by 6.4 months (HR 0.35), whereas in RCC, nivolumab outperforms this compound in OS (25.0 vs. 19.6 months). Contextual factors like tumor microenvironment and prior therapies (e.g., antiangiogenic agents in RCC) require subgroup analyses and translational studies .
Q. How can pharmacokinetic (PK) data optimize this compound dosing in heterogeneous populations?
- Phase I PK studies show dose-proportional AUC but saturable Cmax beyond 20 mg/week. A 10 mg/day regimen achieves steady-state concentrations with manageable toxicity. Model-informed drug development (MIDD) tools, such as pharmacodynamic (PD) biomarkers (e.g., S6K1 inhibition), guide dose adjustments in organ dysfunction .
Q. What strategies validate exploratory biomarkers (e.g., chromogranin A) for this compound response prediction?
- In RADIANT-3, elevated baseline chromogranin A and neuron-specific enolase predicted poor OS. Validate such biomarkers through preplanned correlative analyses in prospective trials, adjusting for multiplicity. Cross-study consistency (e.g., BOLERO-1/-3) strengthens clinical utility .
Q. Methodological Frameworks
Q. How to structure a PICOT-compliant research question for this compound trials?
- P opulation: Postmenopausal women with HR+/HER2- advanced breast cancer; I ntervention: this compound + exemestane; C omparison: Placebo + exemestane; O utcome: PFS; T ime: 12-month follow-up. This framework ensures clarity and feasibility .
Q. What tools analyze this compound-induced transcriptomic changes in tumor samples?
- RNA-seq paired with pathway enrichment analysis (e.g., Gene Set Enrichment Analysis) identifies mTORC1-dependent gene signatures. Single-cell sequencing can dissect intratumoral heterogeneity and resistance mechanisms .
Q. How to design a crossover-adjusted OS analysis in this compound trials?
- Apply rank-preserving structural failure time models to account for placebo-to-everolimus crossover (85% in RADIANT-3). Sensitivity analyses (e.g., inverse probability weighting) mitigate confounding, though residual bias may persist .
Q. Data Interpretation & Reporting
Q. Why do central vs. local PFS assessments differ in this compound trials?
- Central review (blinded to treatment) reduces investigator bias, particularly in slow-progressing tumors like pNET. Discordance rates (e.g., 10.6 vs. 6.9 months in BOLERO-2) necessitate protocol-specified imaging schedules and adjudication processes .
Q. How to reconcile negative OS results despite significant PFS benefits?
- In BOLERO-2, crossover and subsequent therapies (e.g., CDK4/6 inhibitors) diluted OS signals. Pre-specified weighted analyses (e.g., RPSFTM) and post-hoc subgroup evaluations (e.g., biomarker-positive cohorts) clarify this compound' survival impact .
Q. Emerging Research Directions
Q. What trial designs evaluate this compound in CDK4/6 inhibitor-resistant HR+ breast cancer?
- Phase II single-arm studies with historical controls (e.g., fulvestrant monotherapy) can assess this compound + endocrine therapy. Endpoints include PFS at 6 months and ctDNA clearance rates .
Q. How to integrate this compound with immunotherapy in mTOR-altered tumors?
Properties
CAS No. |
159351-69-6 |
|---|---|
Molecular Formula |
C53H83NO14 |
Molecular Weight |
958.2 g/mol |
IUPAC Name |
(1R)-1,18-dihydroxy-12-[1-[4-(2-hydroxyethoxy)-3-methoxycyclohexyl]propan-2-yl]-19,30-dimethoxy-15,17,21,23,29,35-hexamethyl-11,36-dioxa-4-azatricyclo[30.3.1.04,9]hexatriaconta-16,24,26,28-tetraene-2,3,10,14,20-pentone |
InChI |
InChI=1S/C53H83NO14/c1-32-16-12-11-13-17-33(2)44(63-8)30-40-21-19-38(7)53(62,68-40)50(59)51(60)54-23-15-14-18-41(54)52(61)67-45(35(4)28-39-20-22-43(66-25-24-55)46(29-39)64-9)31-42(56)34(3)27-37(6)48(58)49(65-10)47(57)36(5)26-32/h11-13,16-17,27,32,34-36,38-41,43-46,48-49,55,58,62H,14-15,18-26,28-31H2,1-10H3/t32?,34?,35?,36?,38?,39?,40?,41?,43?,44?,45?,46?,48?,49?,53-/m1/s1 |
InChI Key |
HKVAMNSJSFKALM-VUDSBINYSA-N |
SMILES |
CC1CCC2CC(C(=CC=CC=CC(CC(C(=O)C(C(C(=CC(C(=O)CC(OC(=O)C3CCCCN3C(=O)C(=O)C1(O2)O)C(C)CC4CCC(C(C4)OC)OCCO)C)C)O)OC)C)C)C)OC |
Isomeric SMILES |
CC1CCC2CC(C(=CC=CC=CC(CC(C(=O)C(C(C(=CC(C(=O)CC(OC(=O)C3CCCCN3C(=O)C(=O)[C@@]1(O2)O)C(C)CC4CCC(C(C4)OC)OCCO)C)C)O)OC)C)C)C)OC |
Canonical SMILES |
CC1CCC2CC(C(=CC=CC=CC(CC(C(=O)C(C(C(=CC(C(=O)CC(OC(=O)C3CCCCN3C(=O)C(=O)C1(O2)O)C(C)CC4CCC(C(C4)OC)OCCO)C)C)O)OC)C)C)C)OC |
Appearance |
White to off-white solid powder |
Key on ui application |
Everolimus is currently used as an immunosuppressant to prevent rejection of organ transplants. In a similar fashion to other mTOR inhibitors Everolimus' effect is solely on the mTORC1 protein and not on the mTORC2 protein. |
boiling_point |
998.7±75.0 °C at 760 mmHg |
melting_point |
N/A |
Key on ui other cas no. |
159351-69-6 |
Pictograms |
Health Hazard |
Purity |
>98% (or refer to the Certificate of Analysis) |
shelf_life |
Stable under recommended storage conditions. |
solubility |
Soluble in DMSO, not in water |
storage |
−20°C |
Synonyms |
001, RAD 40-O-(2-hydroxyethyl)-rapamycin Afinitor Certican everolimus RAD 001 RAD, SDZ RAD001 SDZ RAD SDZ-RAD |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















