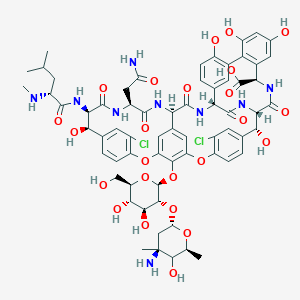
Vancomycin
Overview
Description
Vancomycin is a glycopeptide antibiotic primarily used to treat severe bacterial infections caused by Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus (MRSA) and Clostridium difficile . It was first isolated from a soil sample in Borneo in 1956 and has since become a critical tool in combating antibiotic-resistant infections .
Preparation Methods
Vancomycin is produced through the fermentation of the bacterium Amycolatopsis orientalis . The industrial production involves several steps:
Fermentation: Amycolatopsis orientalis is cultured in a nutrient-rich medium to produce this compound.
Extraction: The antibiotic is extracted from the fermentation broth using organic solvents.
Purification: High-performance liquid chromatography (HPLC) is commonly used to purify this compound.
Chemical Reactions Analysis
Biosynthetic Cross-Linking Reactions
Vancomycin’s bioactive structure arises from enzymatic oxidative cross-linking of a linear heptapeptide precursor. Three cytochrome P450 enzymes (OxyB, OxyA, OxyC) catalyze sequential aryl-ether and carbon-carbon bond formations:
- OxyB : Cross-links residues 4 (Tyr) and 6 (Hpg) via an aryl-ether bond.
- OxyA : Links residues 2 (Hpg) and 4 (Tyr) through a second aryl-ether bond.
- OxyC : Installs a strained carbon-carbon bond between residues 5 (Hpg) and 7 (Dpg), completing the rigid cup-shaped scaffold .
Mechanistic Hydrogen Bonding Interactions
This compound binds the D-Ala-D-Ala terminus of peptidoglycan precursors via five hydrogen bonds:
- Backbone carbonyl of residue 4 (Tyr) ↔ NH of D-Ala.
- Residue 5 (Hpg) NH ↔ D-Ala carbonyl.
- Residue 2 (Hpg) NH ↔ D-Ala carbonyl.
- Residue 6 (Hpg) NH ↔ D-Ala NH.
- Residue 7 (Dpg) NH ↔ D-Ala NH .
This interaction disrupts transglycosylation, weakening bacterial cell walls . Resistance arises when D-Ala-D-Lac replaces D-Ala-D-Ala, eliminating one hydrogen bond and introducing electrostatic repulsion .
Hydrophobic Substituents
Adding hydrophobic groups (e.g., quaternary ammonium moieties) enhances activity against this compound-resistant enterococci (VRE):
| Derivative | Modification Site | Effect on MIC (μg/mL) |
|---|---|---|
| QVA-1 | N-terminus | 0.5 (vs. 64 for this compound) |
| QVA-20 | C-terminus | 1.0 (vs. 128) |
Dual-Binding Analogs
Total synthesis enables modifications to the heptapeptide core for dual binding to D-Ala-D-Ala and D-Ala-D-Lac:
Degradation and Metabolic Pathways
This compound undergoes enzymatic and environmental degradation:
- In vitro metabolism : Rat liver microsomes catalyze demethylation and hydroxylation .
- Aquatic degradation : Hydrolysis under alkaline conditions cleaves glycosidic bonds, yielding aglycone fragments .
Table 2: Major Degradation Products
| Condition | Primary Product | Bioactivity Loss |
|---|---|---|
| Alkaline hydrolysis | Aglycone + vancosamine | Complete |
| Collision energy | CO loss from polypeptide | Partial |
Synthetic Conjugation Reactions
This compound’s N-methylleucine residue enables site-specific conjugation:
- Selenocysteine-mediated linking : Attaches antimicrobial peptides (e.g., CRAMP) via selenocysteine handles, enhancing potency against MRSA (MIC: 0.25 μg/mL vs. 2 μg/mL for this compound) .
Structural Analysis via NMR and MS
Nuclear magnetic resonance (NMR) and mass spectrometry (MS) elucidate this compound’s structure:
- 1H-13C HMBC : Confirmed α-L-vancosaminyl-(1→2)-O-β-D-glucosyl linkage .
- CD spectroscopy : Verified 3D structure consistency with reference standards .
Key Findings and Implications
- Biosynthesis : Enzymatic cross-linking by OxyB/OxyA/OxyC is indispensable for bioactivity .
- Resistance Mitigation : Hydrophobic and dual-binding analogs show promise against VRE .
- Degradation : Environmental stability limits ecological impact but necessitates controlled disposal .
This synthesis underscores this compound’s chemical versatility and ongoing efforts to combat resistance through innovative synthetic strategies.
Scientific Research Applications
Clinical Applications
FDA-Approved Indications:
Vancomycin is indicated for various infections, including:
- Clostridioides difficile-associated diarrhea
- Staphylococcal infections (including septicemia, skin and soft tissue infections)
- Endocarditis caused by enterococci and staphylococci
- Bone infections and lower respiratory tract infections
Off-Label Uses:
this compound is also utilized in several off-label scenarios:
- Catheter-related infections
- Bacterial meningitis
- Surgical prophylaxis
- Necrotizing skin and soft tissue infections
Research Findings
Recent studies have highlighted the efficacy of this compound in various clinical settings:
-
Topical Application in Surgical Site Infections:
A systematic review indicated that the local application of this compound powder significantly reduces the incidence of surgical site infections (SSIs) following joint arthroplasty. However, it was associated with an increased risk of complications such as delayed healing . -
Pediatric Applications:
Research has demonstrated this compound's immunomodulatory effects in pediatric patients with inflammatory bowel disease, suggesting its potential role beyond mere antibacterial action . -
Intrawound Use in Spine Surgery:
A meta-analysis involving 18 studies showed that intrawound application of this compound powder significantly lowers the odds of developing deep infections during spine surgeries compared to standard practices . The odds ratio for deep infections was found to be 0.23, indicating a substantial protective effect.
Data Table: Summary of this compound Applications
| Application Type | Description | Evidence Level |
|---|---|---|
| FDA-Approved Uses | Treats MRSA, endocarditis, bone infections | High |
| Off-Label Uses | Includes meningitis, surgical prophylaxis | Moderate |
| Topical Application | Reduces SSIs in joint surgeries | Systematic Review |
| Pediatric Immunomodulation | Affects TNF-alpha pathways in inflammatory conditions | Case Study |
| Intrawound Use in Spine Surgery | Lowers deep infection rates significantly | Meta-analysis |
Mechanism of Action
Vancomycin works by inhibiting cell wall synthesis in bacteria. It binds to the D-alanyl-D-alanine terminus of cell wall precursor units, preventing their incorporation into the cell wall and thus weakening the bacterial cell wall, leading to cell lysis and death . This mechanism makes it highly effective against Gram-positive bacteria .
Comparison with Similar Compounds
Vancomycin is often compared with other antibiotics, such as:
Linezolid: A synthetic oxazolidinone that inhibits protein synthesis.
Daptomycin: A lipopeptide that disrupts cell membrane function.
Tigecycline: A glycylcycline that inhibits protein synthesis.
Teicoplanin: Another glycopeptide with a similar mechanism of action.
This compound’s uniqueness lies in its ability to treat severe infections caused by antibiotic-resistant bacteria, making it a vital tool in modern medicine .
Biological Activity
Vancomycin is a glycopeptide antibiotic that plays a crucial role in the treatment of serious infections caused by Gram-positive bacteria, particularly those resistant to other antibiotics. This article delves into its biological activity, mechanisms of action, and recent research findings, including case studies and data tables.
This compound primarily exerts its antibacterial effect by inhibiting cell wall synthesis in bacteria. It binds to the D-alanyl-D-alanine terminus of cell wall precursor units, preventing their incorporation into the growing cell wall. This action leads to cell lysis and death, particularly in rapidly dividing bacteria. Additionally, this compound has been shown to alter the permeability of bacterial cell membranes and inhibit RNA synthesis, further contributing to its antimicrobial effects .
Antibacterial Spectrum
This compound is effective against a broad range of Gram-positive organisms, including:
- Staphylococcus aureus (including MRSA)
- Streptococcus pneumoniae
- Enterococcus faecium (including this compound-resistant strains)
- Clostridium difficile
Despite its effectiveness, this compound has limited activity against Gram-negative bacteria due to its large molecular size and inability to penetrate the outer membrane .
Recent Research Findings
Recent studies have focused on enhancing the efficacy of this compound through various modifications and combinations:
- Novel Derivatives : Research indicates that introducing quaternary ammonium cations into this compound can significantly improve its antibacterial activity. For instance, a derivative known as QAV-a1 showed higher survival rates in infected mice compared to standard this compound at higher doses .
- Conjugation with Cell-Penetrating Peptides (CPPs) : Studies demonstrated that conjugating this compound with CPPs like Tat (47–57) enhances its antimicrobial properties. The cytotoxicity of these conjugates was evaluated, showing varied effects on cell viability at different concentrations .
- Biofilm Activity : this compound's efficacy against biofilms is notably lower than its activity against planktonic cells. Modifications such as dipicolylamine functionalization have been explored to enhance its effectiveness against biofilm-associated infections .
Case Studies
Several case studies illustrate the clinical application of this compound:
- Case Study 1 : A patient with MRSA bacteremia was treated with this compound combined with an aminoglycoside, resulting in a successful outcome. The combination therapy demonstrated enhanced efficacy compared to monotherapy, highlighting the importance of synergistic effects in treatment .
- Case Study 2 : In a pediatric population, the emergence of resistance to this compound was monitored. Despite concerns about resistance patterns, no significant increase was observed over three decades, indicating sustained effectiveness against common pathogens .
Table 1: Efficacy of this compound Against Various Bacterial Strains
| Bacterial Strain | Minimum Inhibitory Concentration (MIC) | Resistance Status |
|---|---|---|
| Staphylococcus aureus (MRSA) | 1 µg/mL | Sensitive |
| Enterococcus faecium | 8 µg/mL | VanA phenotype |
| Streptococcus pneumoniae | 0.5 µg/mL | Sensitive |
| Clostridium difficile | 4 µg/mL | Sensitive |
Table 2: Comparative Survival Rates in In Vivo Studies
| Treatment | Dosage (mg/kg) | Survival Rate (%) |
|---|---|---|
| This compound | 40 | 33.3 |
| QAV-a1 | 40 | 100 |
Q & A
Basic Research Questions
Q. How should researchers design experiments to optimize vancomycin formulation parameters (e.g., drug entrapment efficiency)?
- Methodological Answer : Use the Box–Behnken experimental design (BBD) to systematically vary factors like polymer concentration and crosslinking agents. Analyze responses (e.g., drug release kinetics, entrapment efficiency) via ANOVA and response surface plots. This approach identifies optimal formulation parameters while minimizing experimental runs .
Q. What factors influence this compound blood concentration variability in critically ill patients?
- Methodological Answer : Conduct retrospective analyses using Spearman correlation and logistic regression to assess covariates like renal function (12-hour creatinine clearance, 12 h-CLCR), age, and APACHE II scores. Stratify dosing based on 12 h-CLCR thresholds to predict therapeutic concentrations .
Q. How can clinical trials evaluate this compound’s efficacy in reducing surgical site infections (SSIs)?
- Methodological Answer : Perform meta-analyses of randomized controlled trials (RCTs) with subgroup analyses to account for study heterogeneity (e.g., surgical type, dosing regimens). Use random-effects models to quantify SSI risk reduction, reporting odds ratios and confidence intervals .
Q. What are validated methods for monitoring this compound trough levels in diverse populations?
- Methodological Answer : Implement therapeutic drug monitoring (TDM) protocols using high-performance liquid chromatography (HPLC) or immunoassays. Adjust dosing based on population pharmacokinetic (PPK) models, particularly in patients with renal impairment or obesity .
Q. How do researchers address contradictions in this compound nephrotoxicity findings?
- Methodological Answer : Use advanced biostatistical models (e.g., generalized linear mixed models) to adjust for confounders like concurrent sepsis or chemotherapy. Compare cohorts with similar baseline risks and isolate this compound’s direct effects .
Advanced Research Questions
Q. What molecular pathways underlie this compound-induced nephrotoxicity?
- Methodological Answer : Apply stable isotope labeling by amino acids in cell culture (SILAC) to quantify proteomic changes in kidney cells. Perform pathway enrichment analysis to identify dysregulated processes (e.g., apoptosis, oxidative stress) and validate targets via CRISPR/Cas9 knockout models .
Q. How can PPK models improve this compound dosing in specific subpopulations (e.g., lower respiratory infections)?
- Methodological Answer : Develop PPK models using nonlinear mixed-effects modeling (NONMEM) with covariates like CLCR, albumin levels, and disease severity. Validate models through bootstrap and visual predictive checks to ensure robustness .
Q. What genetic mechanisms drive reduced this compound susceptibility in Staphylococcus aureus?
- Methodological Answer : Conduct whole-genome sequencing of this compound-intermediate (VISA) and heterogeneous VISA (hVISA) strains. Identify mutations in global regulators (e.g., walKR, graSR) and correlate with phenotypic changes (e.g., cell wall thickening) using transcriptomics and electron microscopy .
Q. How do multifaceted interventions improve guideline adherence for this compound dosing?
- Methodological Answer : Implement systematic reviews to evaluate educational, technological, and audit-focused interventions. Measure outcomes like trough target attainment and nephrotoxicity rates, using pre-post studies or interrupted time-series analyses .
Q. Can machine learning predict this compound resistance emergence in gut microbiota?
- Methodological Answer : Train algorithms on metagenomic datasets from patients receiving oral this compound. Feature selection should include antibiotic exposure duration, baseline microbiota diversity, and Enterococcus colonization dynamics. Validate models in prospective cohorts .
Q. Methodological Frameworks for Data Analysis
Q. How to resolve heterogeneity in this compound meta-analyses?
- Answer : Apply scoping review frameworks to map evidence gaps and sources of bias. Use sensitivity analyses to exclude low-quality studies and meta-regression to explore covariates (e.g., dosing regimens, pathogen susceptibility) .
Q. What statistical tools are optimal for analyzing this compound’s organ toxicity risks?
- Answer : Employ competing-risk regression models to account for simultaneous outcomes (e.g., sepsis, chemotherapy toxicity). Use mediation analysis to distinguish direct drug effects from confounding pathways .
Q. Data Sharing and Reproducibility
Q. How can researchers access clinical trial data for secondary this compound analyses?
- Answer : Submit proposals to repositories like the Antibacterial Resistance Leadership Group (ARLG). Include methodological rigor assessments and data-sharing agreements. Access protocols, statistical plans, and datasets for independent validation .
Q. What tools enhance reproducibility in this compound pharmacokinetic studies?
Properties
CAS No. |
1404-90-6 |
|---|---|
Molecular Formula |
C66H75Cl2N9O24 |
Molecular Weight |
1449.2 g/mol |
IUPAC Name |
48-[3-[(4S)-4-amino-5-hydroxy-4,6-dimethyloxan-2-yl]oxy-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-22-(2-amino-2-oxoethyl)-5,15-dichloro-2,18,32,35,37-pentahydroxy-19-[[4-methyl-2-(methylamino)pentanoyl]amino]-20,23,26,42,44-pentaoxo-7,13-dioxa-21,24,27,41,43-pentazaoctacyclo[26.14.2.23,6.214,17.18,12.129,33.010,25.034,39]pentaconta-3,5,8(48),9,11,14,16,29(45),30,32,34(39),35,37,46,49-pentadecaene-40-carboxylic acid |
InChI |
InChI=1S/C66H75Cl2N9O24/c1-23(2)12-34(71-5)58(88)76-49-51(83)26-7-10-38(32(67)14-26)97-40-16-28-17-41(55(40)101-65-56(54(86)53(85)42(22-78)99-65)100-44-21-66(4,70)57(87)24(3)96-44)98-39-11-8-27(15-33(39)68)52(84)50-63(93)75-48(64(94)95)31-18-29(79)19-37(81)45(31)30-13-25(6-9-36(30)80)46(60(90)77-50)74-61(91)47(28)73-59(89)35(20-43(69)82)72-62(49)92/h6-11,13-19,23-24,34-35,42,44,46-54,56-57,65,71,78-81,83-87H,12,20-22,70H2,1-5H3,(H2,69,82)(H,72,92)(H,73,89)(H,74,91)(H,75,93)(H,76,88)(H,77,90)(H,94,95)/t24?,34?,35?,42?,44?,46?,47?,48?,49?,50?,51?,52?,53?,54?,56?,57?,65?,66-/m0/s1 |
InChI Key |
MYPYJXKWCTUITO-BSRCFTEOSA-N |
SMILES |
CC1C(C(CC(O1)OC2C(C(C(OC2OC3=C4C=C5C=C3OC6=C(C=C(C=C6)C(C(C(=O)NC(C(=O)NC5C(=O)NC7C8=CC(=C(C=C8)O)C9=C(C=C(C=C9O)O)C(NC(=O)C(C(C1=CC(=C(O4)C=C1)Cl)O)NC7=O)C(=O)O)CC(=O)N)NC(=O)C(CC(C)C)NC)O)Cl)CO)O)O)(C)N)O |
Isomeric SMILES |
CC1C([C@@](CC(O1)OC2C(C(C(OC2OC3=C4C=C5C=C3OC6=C(C=C(C=C6)C(C(C(=O)NC(C(=O)NC5C(=O)NC7C8=CC(=C(C=C8)O)C9=C(C=C(C=C9O)O)C(NC(=O)C(C(C1=CC(=C(O4)C=C1)Cl)O)NC7=O)C(=O)O)CC(=O)N)NC(=O)C(CC(C)C)NC)O)Cl)CO)O)O)(C)N)O |
Canonical SMILES |
CC1C(C(CC(O1)OC2C(C(C(OC2OC3=C4C=C5C=C3OC6=C(C=C(C=C6)C(C(C(=O)NC(C(=O)NC5C(=O)NC7C8=CC(=C(C=C8)O)C9=C(C=C(C=C9O)O)C(NC(=O)C(C(C1=CC(=C(O4)C=C1)Cl)O)NC7=O)C(=O)O)CC(=O)N)NC(=O)C(CC(C)C)NC)O)Cl)CO)O)O)(C)N)O |
Appearance |
White to off-white solid powder |
Key on ui application |
Vancomycin is an antibiotic used to treat a number of bacterial infections. It is a member of the glycopeptide antibiotic class and is effective mostly against Gram-positive bacteria. |
boiling_point |
N/A |
melting_point |
N/A |
Key on ui other cas no. |
1404-90-6 |
physical_description |
Tan to brown solid; [HSDB] |
Pictograms |
Health Hazard |
Purity |
>98% (or refer to the Certificate of Analysis) |
Related CAS |
1404-93-9 (hydrochloride) 64685-75-2 (sulfate) |
shelf_life |
When reconstituted with sterile water for injection, vancomycin hydrochloride injection is stable for 2 weeks at room temperature; the manufacturers state that reconstituted injections may be stored for 96 hours at 2 - 8 °C without substantial loss of potency. When reconstituted as directed in 0.9% sodium chloride injection or 5% dextrose injection, solutions prepared from ADD-Vantage vials of the drug are stable for 24 hours at room temperature. Vancomycin solutions containing 5 mg/mL in 0.9% sodium chloride injection or 5% dextrose injection are reportedly stable for at least 17 days when stored at 24 °C in glass or PVC containers and for at least 63 days when stored at 5 °C or -10 °C in glass containers. Following reconstitution with sterile water for injection as directed, vancomycin solutions that have been further diluted to a concentration of 5 mg/mL in 5 - 30% dextrose injection are stable when stored in plastic syringes for 24 hours at 4 eg C and then subsequently for 2 hours at room temperature. Solutions are stable for two weeks at room temp or longer if refrigerated. /Hydrochloride/ |
solubility |
White solid; solubility in water: greater than 100 mg/mL; moderately soluble in methanol; insoluble in higher alcohols, acetone, ether; UV max absorption (water): 282 nm (e = 40, 1%, 1 cm) /Vancomycin hydrochloride/ |
source |
Synthetic |
Synonyms |
AB-Vancomycin Diatracin Hydrochloride, Vancomycin Sulfate, Vancomycin Vanco Azupharma VANCO-cell Vanco-saar Vancocin Vancocin HCl Vancocine Vancomicina Abbott Vancomicina Chiesi Vancomicina Combino Phar Vancomicina Norman Vancomycin Vancomycin Hexal Vancomycin Hydrochloride Vancomycin Lilly Vancomycin Phosphate (1:2) Vancomycin Phosphate (1:2), Decahydrate Vancomycin Sulfate Vancomycin-ratiopharm Vancomycine Dakota |
Origin of Product |
United States |
Retrosynthesis Analysis
AI-Powered Synthesis Planning: Our tool employs the Template_relevance Pistachio, Template_relevance Bkms_metabolic, Template_relevance Pistachio_ringbreaker, Template_relevance Reaxys, Template_relevance Reaxys_biocatalysis model, leveraging a vast database of chemical reactions to predict feasible synthetic routes.
One-Step Synthesis Focus: Specifically designed for one-step synthesis, it provides concise and direct routes for your target compounds, streamlining the synthesis process.
Accurate Predictions: Utilizing the extensive PISTACHIO, BKMS_METABOLIC, PISTACHIO_RINGBREAKER, REAXYS, REAXYS_BIOCATALYSIS database, our tool offers high-accuracy predictions, reflecting the latest in chemical research and data.
Strategy Settings
| Precursor scoring | Relevance Heuristic |
|---|---|
| Min. plausibility | 0.01 |
| Model | Template_relevance |
| Template Set | Pistachio/Bkms_metabolic/Pistachio_ringbreaker/Reaxys/Reaxys_biocatalysis |
| Top-N result to add to graph | 6 |
Feasible Synthetic Routes
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















