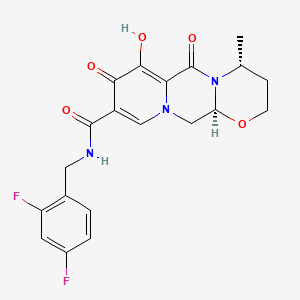
Dolutegravir
Overview
Description
Dolutegravir is an antiretroviral medication used in the treatment of HIV/AIDS. It is classified as an HIV integrase strand transfer inhibitor, which blocks the functioning of the HIV integrase enzyme necessary for viral replication. This compound was approved for medical use in the United States in 2013 and is included in the World Health Organization’s List of Essential Medicines .
Preparation Methods
Synthetic Routes and Reaction Conditions: The synthesis of Dolutegravir involves multiple steps, starting with the construction of a pyridone ring. This is followed by cyclization using 3-®-amino-1-butanol and several sequential chemical transformations. The process can be optimized using continuous flow chemistry, which significantly reduces reaction time and improves yield .
Industrial Production Methods: Industrial production of this compound often employs high-speed homogenization and probe sonication techniques to create nanosuspensions. These methods enhance the solubility and bioavailability of the drug, making it more effective in treating HIV-positive individuals .
Chemical Reactions Analysis
Hydrolysis
- Selective Monoester Hydrolysis :
Amidation
- Coupling Agents : Carbonyldiimidazole (CDI) facilitates amidation between carboxylic acid 4 and 2,4-difluorobenzylamine (10 ) .
- Yield Improvement : Flow conditions increased yield to 89% (vs. 33% in batch) by reducing side reactions .
Cyclization
- Reagent : (R)-3-Aminobutan-1-ol under acidic conditions (acetic acid) forms the oxazine ring .
- Byproduct Control : MgBr<sub>2</sub> promotes intramolecular cyclization, enhancing regioselectivity .
O-Debenzylation
Demethylation
- Conditions : LiBr in THF at 100°C (31-minute residence time in flow) .
- Challenge : Elevated temperatures (>120°C) lead to elimination byproducts (e.g., 20 ) .
Metabolic Reactions of this compound
DTG undergoes three primary metabolic pathways :
| Pathway | Enzyme Involved | Metabolite Formed | Activity |
|---|---|---|---|
| Glucuronidation | UGT1A1 | Ether glucuronide (M2) | Inactive |
| Oxidation | CYP3A4 | Hydroxylated derivatives | Minor activity |
| Glutathione Conjugation | Non-enzymatic | Defluorinated glutathione adduct | Inactive |
- Plasma Half-Life : 15.6 hours, with 64% excreted in feces and 31.6% in urine .
- Drug Interactions : Minimal CYP3A4 induction/inhibition, but UGT1A1 inhibitors (e.g., atazanavir) may increase DTG exposure .
Stability and Byproduct Management
- Thermal Degradation : Demethylation at >120°C produces elimination byproducts; optimized flow conditions mitigate this .
- Oxidative Byproducts : Controlled using inert atmospheres during amidation and cyclization .
This synthesis and metabolic profile underscores this compound’s role as a second-generation HIV integrase inhibitor with a high genetic barrier to resistance .
Scientific Research Applications
Efficacy in Treatment-Naive Patients
Dolutegravir has demonstrated significant efficacy in treatment-naive individuals. Clinical trials such as SPRING-2 and SINGLE have shown that this compound-based regimens achieve viral suppression rates exceeding 90% within 48 weeks. These trials reported that a substantial proportion of participants achieved an undetectable viral load, defined as HIV-1 RNA <50 copies/mL.
| Study | Population | Viral Suppression Rate | Duration |
|---|---|---|---|
| SPRING-2 | Treatment-naive adults | 90% | 48 weeks |
| SINGLE | Treatment-naive adults | 92% | 48 weeks |
Use in Treatment-Experienced Patients
This compound has also been effective in treatment-experienced patients, particularly those with resistance to other antiretrovirals. The VIKING studies assessed this compound's efficacy in patients with documented resistance to raltegravir or elvitegravir. Results indicated that a significant percentage of these patients achieved viral suppression after switching to this compound.
| Study | Population | Viral Suppression Rate | Duration |
|---|---|---|---|
| VIKING-1 | Raltegravir-resistant patients | 75% | 24 weeks |
| VIKING-3 | Integrase inhibitor-naïve patients | 69% | 24 weeks |
Pediatric Applications
Recent studies have explored this compound's potential for pediatric use. A study involving this compound-loaded chitosan nanoparticles aimed to enhance bioavailability for children infected with HIV. The formulation demonstrated improved pharmacokinetics compared to standard this compound, indicating promise for its use in younger populations.
Safety Profile and Adverse Effects
While this compound is generally well-tolerated, some adverse effects have been reported. Notably, psychiatric disorders have emerged as a concern in certain cases. A case report documented a patient experiencing severe psychiatric symptoms after switching to a this compound-based regimen, highlighting the need for careful monitoring.
Resistance and Long-Term Efficacy
Although this compound has a high genetic barrier to resistance, cases of emergent resistance have been documented, particularly in treatment-experienced patients with suboptimal adherence. A notable case from South Africa reported intermediate resistance to this compound after a patient failed to adhere to the prescribed regimen.
Case Study 1: Treatment-Experienced Patient
A 38-year-old woman with tuberculosis was switched to a regimen including this compound but failed to achieve viral suppression due to poor adherence and developed intermediate resistance.
Case Study 2: Psychiatric Symptoms
A 59-year-old woman developed severe psychiatric symptoms shortly after starting this compound, necessitating discontinuation of the drug and subsequent improvement within days.
Mechanism of Action
Dolutegravir inhibits the replication of HIV-1 by binding to the active site of the integrase enzyme. This prevents the strand transfer step of retroviral DNA integration into the host cell’s genome, which is essential for viral replication. By blocking this step, this compound effectively inhibits viral activity and reduces the viral load in the patient’s body .
Comparison with Similar Compounds
Raltegravir: The first integrase inhibitor approved for HIV treatment.
Elvitegravir: Another integrase inhibitor that requires pharmacologic boosting.
Bictegravir: A newer integrase inhibitor with similar efficacy to Dolutegravir.
Cabotegravir: Recently approved integrase inhibitor with a long-acting formulation
Uniqueness of this compound: this compound stands out due to its high efficacy, minimal drug interactions, and ability to be administered without pharmacologic boosting. It also retains activity against some strains of HIV that are resistant to other integrase inhibitors, making it a valuable option in HIV treatment .
Biological Activity
Dolutegravir (DTG) is a potent integrase strand transfer inhibitor (INSTI) that has been extensively studied for its efficacy in treating HIV-1 infection. This article delves into the biological activity of this compound, highlighting its pharmacological properties, resistance profiles, and clinical implications based on recent research findings.
This compound functions by inhibiting the integrase enzyme, which is crucial for the integration of viral DNA into the host genome. By binding to the integrase-DNA complex, this compound prevents the formation of the stable strand transfer complex necessary for viral replication. This mechanism is distinct from other antiretroviral drugs, allowing DTG to maintain efficacy against various resistant strains of HIV-1.
Pharmacokinetics
This compound exhibits favorable pharmacokinetic properties:
- Absorption : Rapidly absorbed with peak plasma concentrations reached within 2-3 hours post-ingestion.
- Half-life : Approximately 12 hours, permitting once-daily dosing without the need for pharmacokinetic enhancers.
- Metabolism : Primarily metabolized via hepatic glucuronidation (UGT1A1), with minimal renal excretion, making it suitable for patients with renal impairment .
Resistance Profile
This compound has demonstrated robust activity against various HIV-1 variants, including those resistant to other INSTIs like raltegravir. Key findings include:
- In vitro Activity : this compound retains activity against clinical isolates from patients who have failed raltegravir therapy. For instance, fold changes in susceptibility were observed as follows:
The data suggests that this compound maintains near wild-type levels of activity against isolates with Y143 or N155H mutations, while susceptibility diminishes with the presence of multiple mutations at the Q148 position .
Efficacy in Clinical Studies
Clinical trials have established this compound's efficacy in diverse populations:
- Phase III Trials : Demonstrated superior virologic suppression compared to other antiretrovirals like efavirenz and rilpivirine.
- Pediatric Studies : A study involving children aged 4 weeks to less than 6 years indicated that a dispersible tablet formulation of this compound was well-tolerated and effective .
Case Studies
Several case studies illustrate this compound's clinical application:
- Case Study A : A patient with extensive drug resistance achieved viral suppression after switching to a this compound-based regimen, highlighting its efficacy in treatment-experienced individuals.
- Case Study B : In a cohort study involving pregnant women, this compound was associated with favorable maternal and neonatal outcomes without significant adverse effects .
Safety Profile
This compound has a favorable safety profile:
- Common adverse effects include insomnia and headache, but these are generally mild and manageable.
- Concerns regarding potential neural tube defects in early pregnancy have led to recommendations for alternative therapies during conception planning .
Future Directions
Research is ongoing to explore modified formulations of this compound aimed at enhancing its bioavailability and prolonging its action. One notable advancement is the development of a prodrug variant that significantly increases half-life and reduces clearance rates, showing promise in preclinical models .
Q & A
Basic Research Questions
Q. What are the standard methodologies for assessing Dolutegravir's pharmacokinetic parameters in clinical studies?
- Methodological Answer : Pharmacokinetic studies typically employ serial blood sampling over 24 hours post-administration to measure parameters such as AUC (area under the curve), Cmax (maximum concentration), and trough levels. High-performance liquid chromatography (HPLC) or mass spectrometry is used for quantification. For example, studies in Black Southern African adults used this protocol to evaluate obesity's marginal impact on this compound exposure . Statistical analysis should include non-compartmental modeling to account for inter-individual variability.
Q. How are neuropsychiatric adverse effects (e.g., depression, insomnia) of this compound systematically evaluated?
- Methodological Answer : Meta-analyses of randomized controlled trials (RCTs) with Jadad scores ≥4 (indicating high quality) are standard. Studies like the neurotoxicity meta-analysis compared this compound with rilpivirine and efavirenz-based therapies, using standardized scales (e.g., PHQ-9 for depression) and adjusting for confounding variables such as prior psychiatric history . Subgroup analyses should exclude studies with efavirenz to isolate this compound-specific effects.
Q. What criteria guide this compound dosing in special populations (e.g., pregnant individuals, obese patients)?
- Methodological Answer : Dose adjustments are informed by pharmacokinetic studies measuring drug exposure in target populations. For example, a 24-hour pharmacokinetic study in obese patients (BMI ≥30 kg/m²) found no clinically significant exposure differences, supporting standard dosing . In pregnancy, prospective surveillance studies (e.g., Botswana’s Tsepamo study) monitor outcomes like neural tube defects (NTDs) to assess safety .
Advanced Research Questions
Q. How can researchers design multicohort studies to investigate this compound resistance in real-world settings?
- Methodological Answer : Collaborative designs (e.g., DTG RESIST study) integrate genotypic resistance tests and clinical data from observational cohorts. Key steps include:
- Cohort Selection : Enroll participants with virological failure (viral load >1,000 copies/mL) on this compound-based regimens.
- Data Harmonization : Standardize resistance mutation reporting (e.g., integrase mutations like R263K) and risk factors (e.g., NRTI resistance, subtype differences).
- Statistical Analysis : Use multivariable logistic regression to identify predictors (e.g., monotherapy, non-B subtypes) .
Q. How should contradictory findings on this compound-associated neurotoxicity be reconciled?
- Methodological Answer : Conduct sensitivity analyses to address heterogeneity. For example, the neurotoxicity meta-analysis found increased depression risk only when excluding efavirenz studies, highlighting the need to control for comparator therapies . Additional approaches:
- Mechanistic Studies : Explore this compound’s blood-brain barrier penetration via cerebrospinal fluid (CSF) pharmacokinetic sampling.
- Longitudinal Designs : Track neuropsychiatric symptoms over extended periods to distinguish transient vs. persistent effects.
Q. What strategies improve reproducibility in this compound pharmacokinetic studies across diverse populations?
- Methodological Answer : Implement protocol standardization:
- Sampling Intervals : Fixed timepoints (e.g., 0, 2, 4, 8, 24 hours post-dose).
- Analytical Validation : Cross-laboratory calibration of HPLC/mass spectrometry methods.
- Demographic Reporting : Include BMI, renal/hepatic function, and genetic factors (e.g., UGT1A1 polymorphisms affecting metabolism) .
Q. Contradiction Analysis
- Obesity vs. Dosing : While found no need for dose adjustment in obesity, prior hypotheses suggested altered drug distribution in adipose tissue. Researchers should validate findings in larger cohorts and diverse ethnicities .
- Subtype-Specific Resistance : highlights higher this compound resistance in non-B subtypes (e.g., CRF02_AG). This necessitates subtype-stratified analyses in clinical trials .
Properties
IUPAC Name |
(3S,7R)-N-[(2,4-difluorophenyl)methyl]-11-hydroxy-7-methyl-9,12-dioxo-4-oxa-1,8-diazatricyclo[8.4.0.03,8]tetradeca-10,13-diene-13-carboxamide | |
|---|---|---|
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI |
InChI=1S/C20H19F2N3O5/c1-10-4-5-30-15-9-24-8-13(17(26)18(27)16(24)20(29)25(10)15)19(28)23-7-11-2-3-12(21)6-14(11)22/h2-3,6,8,10,15,27H,4-5,7,9H2,1H3,(H,23,28)/t10-,15+/m1/s1 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
InChI Key |
RHWKPHLQXYSBKR-BMIGLBTASA-N | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Canonical SMILES |
CC1CCOC2N1C(=O)C3=C(C(=O)C(=CN3C2)C(=O)NCC4=C(C=C(C=C4)F)F)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Isomeric SMILES |
C[C@@H]1CCO[C@@H]2N1C(=O)C3=C(C(=O)C(=CN3C2)C(=O)NCC4=C(C=C(C=C4)F)F)O | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Molecular Formula |
C20H19F2N3O5 | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
DSSTOX Substance ID |
DTXSID90909356 | |
| Record name | Dolutegravir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90909356 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
Molecular Weight |
419.4 g/mol | |
| Source | PubChem | |
| URL | https://pubchem.ncbi.nlm.nih.gov | |
| Description | Data deposited in or computed by PubChem | |
Solubility |
Slightly soluble | |
| Record name | Dolutegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08930 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Mechanism of Action |
Dolutegravir is an HIV-1 antiviral agent. It inhibits HIV integrase by binding to the active site and blocking the strand transfer step of retroviral DNA integration in the host cell. The strand transfer step is essential in the HIV replication cycle and results in the inhibition of viral activity. Dolutegravir has a mean EC50 value of 0.5 nM (0.21 ng/mL) to 2.1 nM (0.85 ng/mL) in peripheral blood mononuclear cells (PBMCs) and MT-4 cells., Dolutegravir inhibits HIV integrase by binding to the integrase active site and blocking the strand transfer step of retroviral deoxyribonucleic acid (DNA) integration which is essential for the HIV replication cycle. Strand transfer biochemical assays using purified HIV-1 integrase and pre-processed substrate DNA resulted in IC50 values of 2.7 nM and 12.6 nM. | |
| Record name | Dolutegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08930 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Dolutegravir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8152 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
CAS No. |
1051375-16-6 | |
| Record name | Dolutegravir | |
| Source | CAS Common Chemistry | |
| URL | https://commonchemistry.cas.org/detail?cas_rn=1051375-16-6 | |
| Description | CAS Common Chemistry is an open community resource for accessing chemical information. Nearly 500,000 chemical substances from CAS REGISTRY cover areas of community interest, including common and frequently regulated chemicals, and those relevant to high school and undergraduate chemistry classes. This chemical information, curated by our expert scientists, is provided in alignment with our mission as a division of the American Chemical Society. | |
| Explanation | The data from CAS Common Chemistry is provided under a CC-BY-NC 4.0 license, unless otherwise stated. | |
| Record name | Dolutegravir [USAN:INN] | |
| Source | ChemIDplus | |
| URL | https://pubchem.ncbi.nlm.nih.gov/substance/?source=chemidplus&sourceid=1051375166 | |
| Description | ChemIDplus is a free, web search system that provides access to the structure and nomenclature authority files used for the identification of chemical substances cited in National Library of Medicine (NLM) databases, including the TOXNET system. | |
| Record name | Dolutegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08930 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
| Record name | Dolutegravir | |
| Source | EPA DSSTox | |
| URL | https://comptox.epa.gov/dashboard/DTXSID90909356 | |
| Description | DSSTox provides a high quality public chemistry resource for supporting improved predictive toxicology. | |
| Record name | (4R,12aS)-N-(2,4-difluorobenzyl)-7-hydroxy-4-methyl-6,8-dioxo-3,4,6,8,12,12a-hexahydro-2H-pyrido[1',2':4,5]pyrazino[2,1-b][1,3]oxazine-9-carboxamide | |
| Source | European Chemicals Agency (ECHA) | |
| URL | https://echa.europa.eu/substance-information/-/substanceinfo/100.237.735 | |
| Description | The European Chemicals Agency (ECHA) is an agency of the European Union which is the driving force among regulatory authorities in implementing the EU's groundbreaking chemicals legislation for the benefit of human health and the environment as well as for innovation and competitiveness. | |
| Explanation | Use of the information, documents and data from the ECHA website is subject to the terms and conditions of this Legal Notice, and subject to other binding limitations provided for under applicable law, the information, documents and data made available on the ECHA website may be reproduced, distributed and/or used, totally or in part, for non-commercial purposes provided that ECHA is acknowledged as the source: "Source: European Chemicals Agency, http://echa.europa.eu/". Such acknowledgement must be included in each copy of the material. ECHA permits and encourages organisations and individuals to create links to the ECHA website under the following cumulative conditions: Links can only be made to webpages that provide a link to the Legal Notice page. | |
| Record name | DOLUTEGRAVIR | |
| Source | FDA Global Substance Registration System (GSRS) | |
| URL | https://gsrs.ncats.nih.gov/ginas/app/beta/substances/DKO1W9H7M1 | |
| Description | The FDA Global Substance Registration System (GSRS) enables the efficient and accurate exchange of information on what substances are in regulated products. Instead of relying on names, which vary across regulatory domains, countries, and regions, the GSRS knowledge base makes it possible for substances to be defined by standardized, scientific descriptions. | |
| Explanation | Unless otherwise noted, the contents of the FDA website (www.fda.gov), both text and graphics, are not copyrighted. They are in the public domain and may be republished, reprinted and otherwise used freely by anyone without the need to obtain permission from FDA. Credit to the U.S. Food and Drug Administration as the source is appreciated but not required. | |
| Record name | Dolutegravir | |
| Source | Hazardous Substances Data Bank (HSDB) | |
| URL | https://pubchem.ncbi.nlm.nih.gov/source/hsdb/8152 | |
| Description | The Hazardous Substances Data Bank (HSDB) is a toxicology database that focuses on the toxicology of potentially hazardous chemicals. It provides information on human exposure, industrial hygiene, emergency handling procedures, environmental fate, regulatory requirements, nanomaterials, and related areas. The information in HSDB has been assessed by a Scientific Review Panel. | |
Melting Point |
190-193ºC | |
| Record name | Dolutegravir | |
| Source | DrugBank | |
| URL | https://www.drugbank.ca/drugs/DB08930 | |
| Description | The DrugBank database is a unique bioinformatics and cheminformatics resource that combines detailed drug (i.e. chemical, pharmacological and pharmaceutical) data with comprehensive drug target (i.e. sequence, structure, and pathway) information. | |
| Explanation | Creative Common's Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/legalcode) | |
Disclaimer and Information on In-Vitro Research Products
Please be aware that all articles and product information presented on BenchChem are intended solely for informational purposes. The products available for purchase on BenchChem are specifically designed for in-vitro studies, which are conducted outside of living organisms. In-vitro studies, derived from the Latin term "in glass," involve experiments performed in controlled laboratory settings using cells or tissues. It is important to note that these products are not categorized as medicines or drugs, and they have not received approval from the FDA for the prevention, treatment, or cure of any medical condition, ailment, or disease. We must emphasize that any form of bodily introduction of these products into humans or animals is strictly prohibited by law. It is essential to adhere to these guidelines to ensure compliance with legal and ethical standards in research and experimentation.















